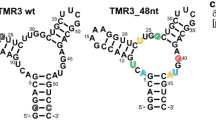
Abstract
Ligand binding RNAs such as artificially created RNA-aptamers are structurally highly diverse. Therefore, they represent important model systems for investigating RNA-folding, RNA-dynamics and the molecular recognition of chemically very different ligands, ranging from small molecules to whole cells. High-resolution structures of RNA-aptamers in complex with their cognate ligands often reveal unexpected tertiary structure elements. Recent studies on different classes of aptamers binding the nucleotide triphosphate GTP as a ligand showed that these systems not only differ widely in binding affinity but also in their ligand binding modes and structural complexity. We initiated the NMR-based structure determination of the high-affinity binding GTP-aptamer 9-12 in order to gain further insights into the diversity of ligand binding modes and structural variability of those aptamers. Here, we report 1H, 13C and 15N resonance assignments for the GTP 9-12-aptamer bound to GTP as the prerequisite for the structure determination by solution NMR.


Similar content being viewed by others
References
Carothers JM, Oestreich SC, Davis JH, Szostak JW (2004) Informational complexity and functional activity of RNA structures. J Am Chem Soc 126:5130–5137. https://doi.org/10.1021/ja031504a
Carothers JM, Oestreich SC, Szostak JW (2006a) Aptamers selected for higher-affinity binding are not more specific for the target ligand. J Am Chem Soc 128:7929–7937. https://doi.org/10.1021/ja060952q
Carothers JM, Davis JH, Chou JJ, Szostak JW (2006b) Solution structure of an informationally complex high-affinity RNA aptamer to GTP. RNA 12:567–579. https://doi.org/10.1261/rna.2251306
Dallmann A, Simon B, Duszczyk MM, Kooshapur H, Pardi A, Bermel W, Sattler M (2013) Efficient detection of hydrogen bonds in dynamic regions of RNA by sensitivity-optimized NMR pulse sequences. Angew Chem Int Ed Engl 52:10487–10490. https://doi.org/10.1002/anie.201304391
Davis JH, Szostak JW (2002) Isolation of high-affinity GTP aptamers from partially structured RNA libraries. Proc Natl Acad Sci USA 99:11616–11621. https://doi.org/10.1073/pnas.182095699
Dingley AJ, Grzesiek S (1998) Direct observation of hydrogen bonds in nucleic acid base pairs by internucleotide 2JNN couplings. J Am Chem Soc 120:8293–8297. https://doi.org/10.1021/ja981513x
Duchardt-Ferner E, Weigand JE, Ohlenschläger O, Schmidtke SR, Suess B, Wöhnert J (2010) Highly modular structure and ligand binding by conformational capture in a minimalistic riboswitch. Angew Chem Int Ed Engl 49:6216–6219. https://doi.org/10.1002/anie.201001339
Ebrahimi M, Rossi P, Rogers C, Harbison GS (2001) Dependence of 13C NMR chemical shifts on conformations of RNA nucleosides and nucleotides. J Magn Reson 150:1–9. https://doi.org/10.1006/jmre.2001.2314
Ellington AD, Szostak JW (1990) In vitro selection of RNA molecules that bind specific ligands. Nature 346:818–822. https://doi.org/10.1038/346818a0
Ferré-D’Amaré AR, Doudna JA (1996) Use of cis- and trans-ribozymes to remove 5′ and 3′ heterogeneities from milligrams of in vitro transcribed RNA. Nucleic Acids Res 24:977–978
Fiala R, Sklenář V (2007) 13C-detected NMR experiments for measuring chemical shifts and coupling constants in nucleic acid bases. J Biomol NMR 39:153–163. https://doi.org/10.1007/s10858-007-9184-4
Homann M, Göringer HU (1999) Combinatorial selection of high affinity RNA ligands to live African trypanosomes. Nucleic Acids Res 27:2006–2014. https://doi.org/10.1093/nar/27.9.2006
Jones CP, Ferré-D’Amaré AR (2017) Long-range interactions in riboswitch control of gene expression. Annu Rev Biophys 46:455–481. https://doi.org/10.1146/annurev-biophys-070816-034042
Juen MA, Wunderlich CH, Nußbaumer F, Tollinger M, Kontaxis G, Konrat R, Hansen DF, Kreutz C (2016) Excited states of nucleic acids probed by proton relaxation dispersion NMR spectroscopy. Angew Chem Int Ed Engl 55:12008–12012. https://doi.org/10.1002/anie.201605870
Keller R (2004) The computer aided resonance assignment tutorial. CANTINA Verlag, Goldau
Legault P, Farmer BT II, Mueller L, Pardi A (1994) Through-bond correlation of adenine protons in a 13C-labeled ribozyme. J Am Chem Soc 116:2203–2204. https://doi.org/10.1021/ja00084a094
Majumdar A, Kettani A, Skripkin E (1999) Observation and measurement of internucleotide 2JNN coupling constants between 15N nuclei with widely separated chemical shifts. J Biomol NMR 14:67–70. https://doi.org/10.1023/A:1008335502416
Milligan JF, Uhlenbeck OC (1989) Synthesis of small RNAs using T7 RNA polymerase. Methods Enzymol 180:51–62. https://doi.org/10.1016/0076-6879(89)80091-6
Nasiri AH, Wurm JP, Immer C, Weickhmann AK, Wöhnert J (2016) An intermolecular G-quadruplex as the basis for GTP recognition in the class V-GTP aptamer. RNA 22:1750–1759. https://doi.org/10.1261/rna.058909.116
Neuner S, Santner T, Kreutz C, Micura R (2015) The “speedy” synthesis of atom-specific 15N imino/amido-labeled RNA. Chemistry 21:11634–11643. https://doi.org/10.1002/chem.201501275
Ohlenschläger O, Wöhnert J, Bucci E, Seitz S, Häfner S, Ramachandran R, Zell R, Görlach M (2004) The structure of the stemloop D subdomain of coxsackievirus B3 cloverleaf RNA and its interaction with the proteinase 3C. Structure 12:237–248. https://doi.org/10.1016/j.str.2004.01.014
Ohlenschläger O, Haumann S, Ramachandran R, Görlach M (2008) Conformational signatures of 13C chemical shifts in RNA ribose. J Biomol NMR 42:139–142. https://doi.org/10.1007/s10858-008-9271-1
Pardi A, Nikonowicz EP (1992) Simple procedure for resonance assignment of the sugar protons in 13C-labeled RNAs. J Am Chem Soc 114:9202–9203. https://doi.org/10.1021/ja00049a070
Piotto M, Saudek V, Sklenář V (1992) Gradient-tailored excitation for single-quantum NMR spectroscopy of aqueous solutions. J Biomol NMR 2:661–665. https://doi.org/10.1007/BF02192855
Simon B, Zanier K, Sattler M (2001) A TROSY relayed HCCH-COSY experiment for correlating adenine H2/H8 resonances in uniformly 13C-labeled RNA molecules. J Biomol NMR 20:173–176. https://doi.org/10.1023/A:1011214914452
Sklenář V, Peterson RD, Rejante MR, Feigon J (1993) Two-and three-dimensional HCN experiments for correlating base and sugar resonances in 15N,13C-labeled RNA oligonucleotides. J Biomol NMR 3:721–727. https://doi.org/10.1007/BF00198375
Sklenář V, Peterson RD, Rejante MR, Feigon J (1994) Correlation of nucleotide base and sugar protons in a 15N-labeled HIV-1 RNA oligonucleotide by 1H-15N HSQC experiments. J Biomol NMR 4:117–122. https://doi.org/10.1007/BF00178339
Sklenář V, Dieckmann T, Butcher SE, Feigon J (1996) Through-bond correlation of imino and aromatic resonances in 13C-,15N-labeled RNA via heteronuclear TOCSY. J Biomol NMR 7:83–87. https://doi.org/10.1007/BF00190460
Tuerk C, Gold L (1990) Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 249:505–510. https://doi.org/10.1126/science.2200121
Wenter P, Reymond L, Auweter SD, Allain FH-T, Pitsch S (2006) Short, synthetic and selectively 13C-labeled RNA sequences for the NMR structure determination of protein-RNA complexes. Nucleic Acids Res 34:e79. https://doi.org/10.1093/nar/gkl427
Wöhnert J, Ramachandran R, Görlach M, Brown LR (1999) Triple-resonance experiments for correlation of H5 and exchangeable base hydrogens in 13C,15N-labeled RNA. J Magn Reson 139:430–433. https://doi.org/10.1006/jmre.1999.1797
Wöhnert J, Görlach M, Schwalbe H (2003) Triple resonance experiments for the simultaneous correlation of H6/H5 and exchangeable protons of pyrimidine nucleotides in 13C,15N-labeled RNA applicable to larger RNA molecules. J Biomol NMR 26:79–83. https://doi.org/10.1023/A:1023040520291
Wolter AC, Weickhmann AK, Nasiri AH, Hantke K, Ohlenschlager O, Wunderlich CH, Kreutz C, Duchardt-Ferner E, Wöhnert J (2017) A stably protonated adenine nucleotide with a highly shifted pKa value stabilizes the tertiary structure of a GTP-binding RNA aptamer. Angew Chem Int Ed Engl 56:401–404. https://doi.org/10.1002/anie.201609184
Wunderlich CH, Spitzer R, Santner T, Fauster K, Tollinger M, Kreutz C (2012) Synthesis of (6-13C) pyrimidine nucleotides as spin-labels for RNA dynamics. J Am Chem Soc 134:7558–7569. https://doi.org/10.1021/ja302148g
Acknowledgements
We are grateful to Christian Richter and Manfred Strupf for maintenance of the NMR facility and to Kerstin Yacoub for support in sample preparation. This work was supported by the Center for Biomolecular Magnetic Resonance (BMRZ) of the Goethe University Frankfurt, the Deutsche Forschungsgemeinschaft (DFG) (SFB 902 ‘‘Molecular principles of RNA-based regulation’’ B10) and the Austrian Science Fund (P28725 and P30370). Robbin Schnieders is recipient of a stipend of the Fonds der Chemischen Industrie.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wolter, A.C., Pianu, A., Kremser, J. et al. NMR resonance assignments for the GTP-binding RNA aptamer 9-12 in complex with GTP. Biomol NMR Assign 13, 281–286 (2019). https://doi.org/10.1007/s12104-019-09892-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12104-019-09892-z