Abstract
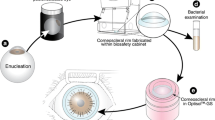
To evaluate the characteristics of microbiological contamination in donor corneas preserved for medium-term. A total of 82 donated corneas from June 1, 2014 to November 30, 2014 were retrospectively analyzed. The corneas were preserved in cornea chambers medium-term solution at 4–8 °C for keratoplasty. After removal of the central corneas for transplantation, the corneoscleral rims were put back into the medium for 1 month at room temperature (20–25 °C). The suspicious contaminated storage solutions indicated with transparency or color change were examined with bacteria and fungi cultivation for strain identification. The data collected included gender, age, procurement site and causes of death of donors, and follow-up of recipients. Statistical analysis was performed using Microsoft Excel and SPSS 24.0. Significance level was set at a P value < 0.05. The overall pathogen positive rate was 9.8% (n = 8), including 7 (87.5%) fungi and 1 (12.5%) bacteria. They were 2 (2.44%) Fusarium, 2 (2.44%) Chromomycosis, 1 (1.22%) Candida albicans, 1 (1.22%) Aspergillus versicolor, 1 (1.22%) Acremonium species, and 1 (1.22%) Enterococcus. 5 contaminated corneas were used for penetrating keratoplasty; although four out of five (80%) had not been given antifungal drugs during more than 6 months following-up period, none of the recipients was infected with a graft. Donor age (P = 0.839), gender (P = 0.062), procurement sites (P = 0.713) and cause of death (P = 0.711) had no statistically significant influence on the contamination rate. All donor corneas have a possibility of microbiological contamination. Strict tissue preservation protocol but not antifungal drugs following keratoplasty seems necessary to prevent graft infection.


Similar content being viewed by others
References
Borowsky CM, Wallau AD, Reetz A, Kwitko S, Rymer S, Locatelli CI (2008) Positive corneoscleral rim culture in penetrating keratoplasty at the Porto Alegre Clinical Hospital. Arq Bras Oftalmol 71:13–17
Chinese Medical Association Eye Branch Keratology Group (2006) Eye bank access standards. Chin J Ophthalmol 08:693 (in Chinese)
Chinese Medical Association Ophthalmology Branch Corneal Disease Group, Xie et al (2006) Corneal transplantation access standards. Chin J Ophthalmol 8:753 (in Chinese)
Davila JR, Mian SI (2016) Infectious keratitis after keratoplasty. Curr Opin Ophthalmol 27:358–366
Fan N, Sun J, Xie L et al (2016) Pathogenic bacteria and drug resistance analysis of 1896 conjunctival sac secretions. Lab Med Clin 15:2180–2182 (in Chinese)
Fuest M, Plum W, Salla S, Walter P, Hermel M (2016) Conjunctival and intraocular swabs for the microbiological assessment of donor corneas. Acta Ophthalmol 94:70–75
Gruenert AK, Rosenbaum K, Geerling G, Fuchsluger TA (2017) The influence of donor factors on corneal organ culture contamination. Acta Ophthalmol 7:733–740
Hong J (2009) Descemet’s stripping automated endothelium keratoplasty. Eye 03:204–207 (in Chinese)
Khouani M, Debellemaniere G, Malugani C et al (2014) Evaluation of microbial contamination of corneal transplants: one-year report from a French regional eye bank. Cornea 33:899–904
Kong Q, Zhai H, Cheng J et al (2016) Clinical characteristics of secondary graft fungal infection after corneal transplantation. Chin J Ophthalmol Vis Sci 18(12):751–756 (in Chinese)
Lin IH, Chang YS, Tseng SH, Huang YH (2016) A comparative, retrospective, observational study of the clinical and microbiological profiles of post-penetrating keratoplasty keratitis. Sci Rep 6:32751
Linke SJ, Fricke OH, Eddy M et al (2013) Risk factors for donor cornea contamination: retrospective analysis of 4546 procured corneas in a single eye bank. Cornea 32:141–148
Merchant A, Zacks CM, Wilhelmus K, Durand M, Dohlman CH (2001) Candidal endophthalmitis after keratoplasty. Cornea 20:226–229
Shang X, Zhang M (2010) Body and organ donation in Wuhan, China. Lancet 376:1033–1034
Shi W, Xie L (2014) The status quo and expectation of corneal research in China. Chin J Ophthalmol 50(9):641–645 (in Chinese)
Vislisel JM, Goins KM, Wagoner MD et al (2017) Incidence and outcomes of positive donor corneoscleral rim fungal cultures after keratoplasty. Ophthalmology 124:36–42
Xie L, Shi W (2007) Cornea disease, vol 4(1). People’s Health Publishing House, Beijing, pp 440–535 (in Chinese)
Xu J, Zhang H, Jiang J et al (2015) Security research of cardiac death donation corneas used as penetrating keratoplasty grafts. Int Eye Sci 02:317–319 (in Chinese)
Funding
The study was funded by the National Natural Science Foundation of China (Nos. 81200661, 81470606, 81570819), the Nature Science Foundation of Hubei Province (No. 2014CFB973), Top ten translational medical research projects from Tongji Hospital (No. 2016ZHYX20). Training Project of young medical pioneers in Wuhan city (No. 2015whzqnyxggrc10).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the ethical committee of Tongji Hospital. Human LNC were isolated and cultured as previously reported. Donor corneas were obtained from the Red Cross Eye Bank of Wuhan City, Tongji Hospital (Hubei, China) and managed in accordance with the Declaration of Helsinki.
Human and animal ethics rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Gg., Zhu, H., Ji, Cn. et al. Microbiological contamination in donor corneas preserved for medium-term. Cell Tissue Bank 20, 379–387 (2019). https://doi.org/10.1007/s10561-019-09776-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-019-09776-5