Abstract
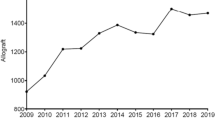
The use of musculoskeletal allografts has become increasingly popular among surgeons. The purpose of this review is to highlight the procurment and delievery process of fresh osteochondral allografts in the United States. The four distributors of fresh osteochondral allografts in the United States were contacted. Surveys containing quantitative and qualitative sections concerning the procurement and processing of osteochondral allograft tissue were obtained. Our results showed an average of 13 ± 4.24 years of experience with osteochondral allografts. The average donor age ranged from 13.5 ± 3 to 37.5 ± 5 years, with an average age of 27 ± 2.83 years. All donors were between ages 12 and 45 years old. The percentage of screened donors that were accepted for allograft transplant was consistent at 70–75% for 3 out of the 4 tissue banks. The percentage of grafts that expire without implantation ranged from 20% to 29%. Maximum shipping time varied between 24 and 96 hours. Each tissue bank used its own proprietary storage medium. The time from donor death to the harvest of allograft tissue was < 24 hours. The most commonly requested osteochondral allograft tissue for all banks was the medial femoral condyle. The market share of fresh allografts is as follows: Joint Restoration Foundation (JRF) 59.9%, Muskuloskeletal Transplant Foundation (MTF) 15.3%, LifeNet Health (LN) 14.5%, and Regeneration Technology Incorporated (RTI) 10.2%, with approximately 4700 fresh allografts distributed in 2018. This compiled data from the four tissue banks that supply fresh osteochondral allograft in the United States provides important background information for patients and orthopaedic surgeons.

Similar content being viewed by others
References
Alford JW, Cole BJ (2005) Cartilage restoration, part 2: techniques, outcomes, and future directions. Am J Sports Med 33:443–460
Allen RT et al (2005) Analysis of stored osteochondral allografts at the time of surgical implantation. Am J Sports Med 33:1479–1484
Årøen A et al (2004) Articular cartilage lesions in 993 consecutive knee arthroscopies. Am J Sports Med 32:211–215
Curl WW et al (1997) Cartilage injuries: a review of 31,516 knee arthroscopies. Arthroscopy. https://doi.org/10.1016/s0749-8063(97)90124-9
Czitrom AA, Keating S, Gross AE (1990) The viability of articular cartilage in fresh osteochondral allografts after clinical transplantation. J Bone Jt Surg 72:574–581
Dozin B et al (2005) Comparative evaluation of autologous chondrocyte implantation and mosaicplasty. Clin J Sport Med 15:220–226
Enneking WF, Campanacci DA (2001) Retrieved human allografts. J Bone Jt Surg Am 83:971–986
Enneking WF, Mindell ER (1991) Observations on massive retrieved human allografts. J Bone Jt Surg Ser A. https://doi.org/10.2106/00004623-199173080-00002
Garrity JT, Stoker AM, Sims HJ, Cook JL (2012) Improved osteochondral allograft preservation using serum-free media at body temperature. Am J Sports Med 40:2542–2548
Gross AE et al (2008) Fresh osteochondral allografts for posttraumatic knee defects: long-term followup. Clin Orthop Relat Res 466:1863–1870
Hangody LR et al (2012) Osteochondral allograft transplantation from a living donor. Arthrosc J Arthrosc Relat Surg 28:1180–1183
Hunt HE, Sadr K, Deyoung AJ, Gortz S, Bugbee WD (2014) The role of immunologic response in fresh osteochondral allografting of the knee. Am J Sports Med. https://doi.org/10.1177/0363546513518733
Kreuz PC et al (2011) Repair of focal cartilage defects with scaffold-assisted autologous chondrocyte grafts. Am J Sports Med 39:1697–1706
LaPrade RF, Botker J, Herzog M, Agel J (2009) Refrigerated osteoarticular allografts to treat articular cartilage defects of the femoral condyles. J Bone Jt Surg Am 91:805–811
McCormick F et al (2014) Trends in the surgical treatment of articular cartilage lesions in the united states: an analysis of a large private-payer database over a period of 8 years. Arthrosc J Arthrosc Relat Surg 30:222–226
Mohr J et al (2016) Disinfection of human musculoskeletal allografts in tissue banking: a systematic review. Cell Tissue Bank. https://doi.org/10.1007/s10561-016-9584-3
Montgomery SR et al (2013) Trends in the surgical treatment of articular cartilage defects of the knee in the United States. Knee Surg Sport Traumatol Arthrosc 22:2070–2075
Nielsen ES, McCauley JC, Pulido PA, Bugbee WD (2017) Return to sport and recreational activity after osteochondral allograft transplantation in the knee. Am J Sports Med 45:1608–1614
Pallante AL et al (2012a) The in vivo performance of osteochondral allografts in the goat is diminished with extended storage and decreased cartilage cellularity. Am J Sports Med 40:1814–1823
Pallante AL et al (2012b) Treatment of articular cartilage defects in the goat with frozen versus fresh osteochondral allografts. J Bone Jt Surg 94:1984–1995
Pitsillides AA, Beier F (2011) Cartilage biology in osteoarthritis–lessons from developmental biology. Nat Rev Rheumatol 7:654–663
Sadr KN, Pulido PA, McCauley JC, Bugbee WD (2016) Osteochondral allograft transplantation in patients with osteochondritis dissecans of the knee. Am J Sports Med 44:2870–2875
Teng MS, Yuen AS, Kim HT (2008) Enhancing osteochondral allograft viability: effects of storage media composition. Clin Orthop Relat Res 466:1804–1809
Thomas RJ, Hourd PC, Williams DJ (2008) Application of process quality engineering techniques to improve the understanding of the in vitro processing of stem cells for therapeutic use. J Biotechnol 136:148–155
Tírico LEP, McCauley JC, Pulido PA, Bugbee WD (2018) Lesion size does not predict outcomes in fresh osteochondral allograft transplantation. Am J Sports Med 46:900–907
Tran-Khanh N, Chevrier A, Lascau-Coman V, Hoemann CD, Buschmann MD (2010) Young adult chondrocytes proliferate rapidly and produce a cartilaginous tissue at the gel-media interface in agarose cultures. Connect Tissue Res 51:216–223
Vaishnav S, Vangsness CT (2009) New techniques in allograft tissue processing. Clin Sports Med 28:127–141
Vangsness CT et al (2003) Allograft transplantation in the knee: tissue regulation, procurement, processing, and sterilization. Am J Sports Med 31:474–481
Vangsness CT, Wagner PP, Moore TM, Roberts MR (2006) Overview of safety issues concerning the preparation and processing of soft-tissue allografts. Arthrosc J Arthrosc Relat Surg 22:1351–1358
Vannini F et al (2013) Osteochondral allografts in the ankle joint. Cartilage 4:204–213
Zouzias IC, Bugbee WD (2016) Osteochondral allograft transplantation in the knee. Sports Med Arthrosc 24:79–84
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Goodfriend, B., Essilfie, A.A., Jones, I.A. et al. Fresh osteochondral grafting in the United States: the current status of tissue banking processing. Cell Tissue Bank 20, 331–337 (2019). https://doi.org/10.1007/s10561-019-09768-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-019-09768-5