Abstract
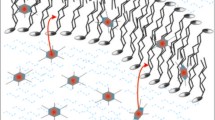
This study validated the use of small unilamellar vesicles (SUVs) made of 1-palmitoyl-2-oleoylphosphatidylcholine with 1 mol % spin label of 1-palmitoyl-2-(16-doxylstearoyl)phosphatidylcholine (16-PC) as an oxygen-sensitive analyte to study cellular respiration. In the analyte, the hydrocarbon environment surrounds the nitroxide moiety of 16-PC. This ensures high oxygen concentration and oxygen diffusion at the location of the nitroxide as well as isolation of the nitroxide moiety from cellular reductants and paramagnetic ions that might interfere with spin-label oximetry measurements. The saturation-recovery electron paramagnetic resonance approach was applied in the analysis since this approach is the most direct method to carry out oximetric studies. It was shown that this display (spin–lattice relaxation rate) is linear in oxygen partial pressure up to 100 % air (159 mmHg). Experiments using a neuronal cell line in suspension were carried out at X-band for closed-chamber geometry. Oxygen consumption rates showed a linear dependence on the number of cells. Other significant benefits of the analyte are: the fast effective rotational diffusion and slow translational diffusion of the spin-probe is favorable for the measurements, and there is no cross-reactivity between oxygen and paramagnetic ions in the lipid bilayer.





Similar content being viewed by others
References
J.M. Backer, V.G. Budker, S.I. Eremenko, Y.N. Molin, Biochim. Biophys. Acta 460, 152–156 (1977)
C.-S. Lai, L.E. Hopwood, J.S. Hyde, S. Lukiewicz, Proc. Natl. Acad. Sci. USA 79, 1166–1170 (1982)
A. Ligeza, A. Wisniewska, W.K. Subczynski, Curr. Topics in Biophys. 16, 92–98 (1992)
A. Kusumi, W.K. Subczynski, J.S. Hyde, Proc. Natl. Acad. Sci. USA 79, 1854–1858 (1982)
J.-J. Yin, J.S. Hyde, Z. Phys. Chem. 153, 57–65 (1987)
W.K. Subczynski, J.S. Hyde, A. Kusumi, Proc. Natl. Acad. Sci. USA 86, 4474–4478 (1989)
W.K. Subczynski, J.S. Hyde, Biochim. Biophys. Acta 643, 283–291 (1981)
C. Altenbach, W. Froncisz, J.S. Hyde, W.L. Hubbell, Biophys. J. 56, 1183–1191 (1989)
W. Froncisz, C.-S. Lai, J.S. Hyde, Proc. Natl. Acad. Sci. USA 82, 411–415 (1985)
H.S. Mchaourab, J.S. Hyde, J. Magn. Reson. 101, 178–184 (1993)
H.S. Mchaourab, J.S. Hyde, J.B. Feix, Biochemistry 33, 6691–6699 (1994)
J.S. Hyde, W.K. Subczynski, J. Magn. Reson. 56, (1984)
W.K. Subczynski, J.S. Hyde, Biophys. J. 45, 743–748 (1984)
J.S. Hyde, W.K. Subczynski, in Biological Magnetic Resonance, vol. 8, ed. by L.J. Berliner, J. Reuben (Plenum Press, New York, 1989), pp. 399–425
D.A. Windrem, W.Z. Plachy, Biochim. Biophys. Acta 600, 655–665 (1980)
C.E. St Denis, C.J. Fell, Can. J. Chem. Eng. 49, 885 (1971)
Y.N. Molin, K.M. Salikhov, K.I. Zamaraev, Spin Exchange (Springer-Verlag, New York, 1980), pp. 111–115
M.-K. Ahn, J. Magn. Reson. 22, 289–293 (1976)
W.K. Subczynski, L.E. Hopwood, J.S. Hyde, J. Gen. Physiol. 100, 69–87 (1992)
W.F. Linke, Solubilities. Inorganic and Metal Organic Compounds II, 4th edn. (American Chemical Society, Washington, DC, 1965), pp. 1233–1236
L. Mainali, M. Raguz, W.K. Subczynski, J. Phys. Chem. B 117, 8994–9003 (2013)
J.-J. Yin, W.K. Subczynski, Biophys. J. 71, 832–839 (1996)
W.K. Subczynski, J. Widomska, A. Wisniewska, A. Kusumi, Methods in Molecular Biology, Lipid Rafts, vol. 398 (Humana Press, Totowa, 2007), pp. 143–157
M.L. Hitchman, Measurement of Dissolved Oxygen (John Wiley & Sons Inc, New York, 1978)
H.M. Swartz, K. Chen, M. Pals, M. Sentjurc, P.D. Morse 2nd, Magn. Reson. Med. 3, 169–174 (1986)
W.K. Subczynski, H.M. Swartz, in Biomedical ESR Part A: Free Radicals, Metals, Medicine, and Physiology, Biological Magnetic Resonance, vol. 23, ed. by S.S. Eaton, G.R. Eaton, L.J. Berliner (Kluwer, Boston, 2005), pp. 229–282
M. Raguz, L. Mainali, J. Widomska, W.K. Subczynski, Chem. Phys. Lipids 164, 819–829 (2011)
H. Träuble, H. Eibl, Proc. Natl. Acad. Sci. USA 71, 214–219 (1974)
D. Papahadjopoulos, Biochim. Biophys. Acta 163, 240–254 (1968)
M. Egret-Charlier, A. Sanson, M. Ptak, FEBS Lett. 89, 313–316 (1978)
A. Sanson, M. Ptak, J.L. Rigaud, C.M. Gary-Bobo, Chem. Phys. Lipids 17, 435–444 (1976)
A. Kusumi, W.K. Subczynski, J.S. Hyde, Fed. Proc. 41, 1394 (1982)
A. Ligeza, H. Swartz, W.K. Subczynski, Curr. Topics in Biophys. 18, 29–38 (1994)
K.J. Liu, M.W. Grinstaff, J. Jiang, K.S. Suslick, H.M. Swartz, W. Wang, Biophys. J. 67, 896–901 (1994)
W. Froncisz, T. Oles, J.S. Hyde, Rev. Sci. Instrum. 57, 1095–1099 (1986)
J.W. Sidabras, R.R. Mett, W. Froncisz, T.G. Camenisch, J.R. Anderson, J.S. Hyde, Rev. Sci. Instrum. 78, 034701 (2007)
J.S. Hyde, J.-J. Yin, W.K. Subczynski, T.G. Camenisch, J.J. Ratke, W. Froncisz, J. Phys. Chem. B 108, 9524–9529 (2004)
W.K. Subczynski, L. Mainali, T.G. Camenisch, W. Froncisz, J.S. Hyde, J. Magn. Reson. 209, 142–148 (2011)
L. Mainali, J.W. Sidabras, T.G. Camenisch, J.J. Ratke, M. Raguz, J.S. Hyde, W.K. Subczynski, Appl. Magn. Reson. 45, 1343–1358 (2014)
L. Mainali, M. Raguz, T.G. Camenisch, J.S. Hyde, W.K. Subczynski, J. Magn. Reson. 212, 86–94 (2011)
L. Mainali, J.S. Hyde, W.K. Subczynski, J. Magn. Reson. 226, 35–44 (2013)
W. Froncisz, T.G. Camenisch, J.J. Ratke, J.R. Anderson, W.K. Subczynski, R.A. Strangeway, J.W. Sidabras, J.S. Hyde, J. Magn. Reson. 193, 297–304 (2008)
J.-J. Yin, J.S. Hyde, J. Chem. Phys. 91, 6029–6035 (1989)
Acknowledgments
This work was supported by Grants EY015526, EB002052, EB001980, and NS081936 from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mainali, L., Vasquez-Vivar, J., Hyde, J.S. et al. Spin-Labeled Small Unilamellar Vesicles with the T 1-Sensitive Saturation-Recovery EPR Display as an Oxygen-Sensitive Analyte for Measurement of Cellular Respiration. Appl Magn Reson 46, 885–895 (2015). https://doi.org/10.1007/s00723-015-0684-1
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00723-015-0684-1