Abstract
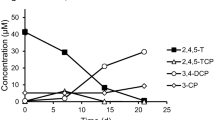
The biotransformation of hexachlorocyclohexane isomers (HCH) by two Dehalococcoides mccartyi strains (195 and BTF08) and an enrichment culture was investigated and compared to conversion by the obligate anaerobic strain Clostridium pasteurianum strain DSMZ 525. The D. mccartyi strains preferentially transformed γ-HCH over α-HCH and δ-HCH isomers while β-HCH biotransformation was not significant. In case of the enrichment culture, γ-HCH was preferentially transformed over the δ-HCH, β-HCH and α-HCH isomers. Major observed metabolites in both cases were tetrachlorocyclohexene and as end products monochlorobenzene (MCB) and benzene. Dechlorination of the γ-HCH isomer was linked to an increase in cell numbers for strain 195. γ-HCH transformation was linked to considerable carbon stable isotope fractionation with the enrichment factor εc = − 5.5 ± 0.8‰ for D. mccartyi strain 195, εc = − 3.1 ± 0.4‰ for the enrichment culture and εc = − 4.1 ± 0.6‰ for co-metabolic transformation by C. pasteurianum.



Similar content being viewed by others
References
Adrian L, Hansen SK, Fung JM, Görisch H, Zinder SH (2007) Growth of Dehalococcoides strains with chlorophenols as electron acceptors. Environ Sci Technol 41:2318–2323
Bachmann A, De Bruin W, Jumelet J, Rijnaarts H, Zehnder A (1988) Aerobic biomineralization of alpha-hexachlorocyclohexane in contaminated soil. Appl Environ Microbiol 54:548–554
Badea SL, Vogt C, Weber S, Danet AF, Richnow HH (2009) Stable isotope fractionation of gamma-hexachlorocyclohexane (Lindane) during reductive dechlorination by two strains of sulfate-reducing bacteria. Environ Sci Technol 43:3155–3161
Badea SL, Vogt C, Gehre M, Fischer A, Danet AF, Richnow HH (2011) Development of an enantiomer-specific stable carbon isotope analysis (ESIA) method for assessing the fate of α-hexachlorocyclohexane in the environment. Rapid Commun Mass Spectrom 25:1363–1372
Bashir S, Fischer A, Nijenhuis I, Richnow H-H (2013) Enantioselective carbon stable isotope fractionation of hexachlorocyclohexane during aerobic biodegradation by Sphingobium spp. Environ Sci Technol 47:11432–11439
Bashir S, Hitzfeld KL, Gehre M, Richnow HH, Fischer A (2015) Evaluating degradation of hexachlorcyclohexane (HCH) isomers within a contaminated aquifer using compound-specific stable carbon isotope analysis (CSIA). Water Res 71:187–196
Bashir S, Hassan W, Niazi NK, Bibi I, Ahmad N (2017) Current approaches for the assessment of in situ remediation of xenobiotics. In: Hashmi M, Kumar V, Varma A (eds) Xenobiotics in the soil environment, vol. 49. Springer, Cham, pp 171–196
Boyle AW, Häggblom MM, Young LY (1999) Dehalogenation of lindane (γ-hexachlorocyclohexane) by anaerobic bacteria from marine sediments and by sulfate-reducing bacteria. FEMS Microbiol Ecol 29:379–387
Bradley AE, Shoenfelt JL, Durda JL (2016) Carcinogenicity and mode of action evaluation for alphahexachlorocyclohexane: implications for human health risk assessment. Regul Toxicol Pharmacol 76:152–173
Buser H-R, Mueller MD (1995) Isomer and enantioselective degradation of hexachlorocyclohexane isomers in sewage sludge under anaerobic conditions. Environ Sci Technol 29:664–672
Cichocka D, Nikolausz M, Haest PJ, Nijenhuis I (2010) Tetrachloroethene conversion to ethene by a Dehalococcoides-containing enrichment culture from Bitterfeld. FEMS Microbiol Ecol 72:297–310
Coplen TB (2011) Guidelines and recommended terms for expression of stable-isotope-ratio and gas-ratio measurement results. Rapid Commun Mass Spectrom 25:2538–2560
Doesburg V, Eekert MH, Middeldorp PJ, Balk M, Schraa G, Stams AJ (2005) Reductive dechlorination of β-hexachlorocyclohexane (β-HCH) by a Dehalobacter species in coculture with a Sedimentibacter sp. FEMS Microbiol Ecol 54:87–95
Elango V, Kurtz HD Jr, Anderson C, Freedman DL (2011) Use of γ-hexachlorocyclohexane as a terminal electron acceptor by an anaerobic enrichment culture. J Hazard Mater 197:204–210
Elsner M (2010) Stable isotope fractionation to investigate natural transformation mechanisms of organic contaminants: principles, prospects and limitations. J Environ Monit 12:2005–2031
Garg N, Lata P, Jit S, Sangwan N, Singh AK, Dwivedi V, Niharika N, Kaur J, Saxena A, Dua A (2016) Laboratory and field scale bioremediation of hexachlorocyclohexane (HCH) contaminated soils by means of bioaugmentation and biostimulation. Biodegradation 27:179–193
Heritage A, MacRae I (1977) Degradation of lindane by cell-free preparations of Clostridium sphenoides. Appl Environ Microbiol 34:222–224
Jagnow G, Haider K, Ellwardt PCHR (1977) Anaerobic dechlorination and degradation of hexachlorocyclohexane isomers by anaerobic and facultative anaerobic bacteria. Arch Microbiol 115:285–292
Kaufhold T, Schmidt M, Cichocka D, Nikolausz M, Nijenhuis I (2013) Dehalogenation of diverse halogenated substrates by a highly enriched Dehalococcoides-containing culture derived from the contaminated mega-site in Bitterfeld. FEMS Microbiol Ecol 83:176–188
Krajmalnik-Brown R, Sung Y, Ritalahti KM, Saunders FM, Loffler FE (2007) Environmental distribution of the trichloroethene reductive dehalogenase gene (tceA) suggests lateral gene transfer among Dehalococcoides. FEMS Microbiol Ecol 59:206–214. https://doi.org/10.1111/j.1574-6941.2006.00243.x
Kumar M, Chaudhary P, Dwivedi M, Kumar R, Paul D, Jain RK, Garg SK, Kumar A (2005) Enhanced biodegradation of β-and δ-hexachlorocyclohexane in the presence of α-and γ-isomers in contaminated soils. Environ Sci Technol 39:4005–4011
Lal R, Pandey G, Sharma P, Kumari K, Malhotra S, Pandey R, Raina V, Kohler HPE, Holliger C, Jackson C, Oakeshott JG (2010) Biochemistry of microbial degradation of hexachlorocyclohexane and prospects for bioremediation. Microbiol Mol Biol Rev 74:58–80
Liu Y, Bashir S, Stollberg R, Trabitzsch R, Weiß H, Paschke H, Nijenhuis I, Richnow H-H (2017) Compound specific and enantioselective stable isotope analysis as tools to monitor transformation of hexachlorocyclohexane (HCH) in a complex aquifer system. Environ Sci Technol 51:8909–8916
Löffler FE, Ritalahti KM, Zinder SH (2013) Dehalococcoides and reductive dechlorination of chlorinated solvents. In: Stroo H, Leeson A, Ward C (eds) Bioaugmentation for groundwater remediation. SERDP ESTCP Environmental Remediation Technology. Springer, New York, pp 39–88
Macrae IC, Raghu K, Bautista EM (1969) Anaerobic degradation of the insecticide lindane by clostridium sp.. Nature 221(5183):859–860
Maymo-Gatell X, Chien Y, Gossett JM, Zinder SH (1997) Isolation of a bacterium that reductively dechlorinates tetrachloroethene to ethene. Science 276(5318):1568–1571
Meckenstock RU, Morasch B, Griebler C, Richnow HH (2004) Stable isotope fractionation analysis as a tool to monitor biodegradation in contaminated acquifers. J Contam Hydrol 75:215–255
Mehboob F, Langenhoff AA, Schraa G, Stams AJ (2013) Anaerobic degradation of lindane and other HCH isomers. In: Malik A, Grohmann E, Alves M (eds) Management of microbial resources in the environment. Springer, Dordrecht, pp 495–521
Middeldorp PJM, Jaspers M, Zehnder AJB, Schraa G (1996) Biotransformation of alpha-, beta-, gamma-, and delta-hexachlorocyclohexane under methanogenic conditions. Environ Sci Technol 30:2345–2349
Nagasawa S, Kikuchi R, Nagata Y, Takagi M, Matsuo M (1993) Aerobic mineralization of [gamma]-HCH by Pseudomonas paucimobilis UT26. Chemosphere 26:1719–1728
Nijenhuis I, Nikolausz M, Köth A, Felföldi T, Weiss H, Drangmeister J, Großmann J, Kästner M, Richnow H-H (2007) Assessment of the natural attenuation of chlorinated ethenes in an anaerobic contaminated aquifer in the Bitterfeld/Wolfen area using stable isotope techniques, microcosm studies and molecular biomarkers. Chemosphere 67:300–311
Ohisa N, Yamaguchi M, Kurihara N (1980) Lindane degradation by cell-free extracts of Clostridium rectum. Arch Microbiol 125:221–225
Phillips TM, Seech AG, Lee H, Trevors JT (2005) Biodegradation of hexachlorocyclohexane (HCH) by microorganisms. Biodegradation 16:363–392
Popp P, Brüggemann L, Keil P, Thuß U, Weiß H (2000) Chlorobenzenes and hexachlorocyclohexanes (HCHs) in the atmosphere of Bitterfeld and Leipzig (Germany). Chemosphere 41:849–855
Pöritz M, Goris T, Wubet T, Tarkka MT, Buscot F, Nijenhuis I, Lechner U, Adrian L (2013) Genome sequences of two dehalogenation specialists—Dehalococcoides mccartyi strains BTF08 and DCMB5 enriched from the highly polluted Bitterfeld region. FEMS Microbiol Lett 343:101–104. https://doi.org/10.1111/1574-6968.12160
Raina V, Suar M, Singh A, Prakash O, Dadhwal M, Gupta SK, Dogra C, Lawlor K, Lal S, van der Meer JR (2008) Enhanced biodegradation of hexachlorocyclohexane (HCH) in contaminated soils via inoculation with Sphingobium indicum B90A. Biodegradation 19:27–40
Ricking M, Schwarzbauer J (2008) HCH residues in point-source contaminated samples of the Teltow Canal in Berlin, Germany. Environ Chem Lett 6:83–89
Sahu SK, Patnaik K, Bhuyan S, Sreedharan B, Kurihara N, Adhya T, Sethunathan N (1995) Mineralization of α-, γ-, and β-isomers of hexachlorocyclohexane by a soil bacterium under aerobic conditions. J Agric Food Chem 43:833–837
Schmidt M, Wolfram D, Birkigt J, Ahlheim J, Paschke H, Richnow H-H, Nijenhuis I (2014) Iron oxides stimulate microbial monochlorobenzene in situ transformation in constructed wetlands and laboratory systems. Sci Total Environ 472:185–193
Seshadri R, Adrian L, Fouts DE, Eisen JA, Phillippy AM, Methe BA, Ward NL, Nelson WC, Deboy RT, Khouri HM (2005) Genome sequence of the PCE-dechlorinating bacterium Dehalococcoides ethenogenes. Science 307:105–108
Van Eekert MH, Van Ras NJ, Mentink GH, Rijnaarts HH, Stams AJ, Field JA, Schraa G (1998) Anaerobic transformation of β-hexachlorocyclohexane by methanogenic granular sludge and soil microflora. Environ Sci Technol 32(21):3299–3304
Vijgen J, Abhilash P, Li YF, Lal R, Forter M, Torres J, Singh N, Yunus M, Tian C, Schäffer A (2011) Hexachlorocyclohexane (HCH) as new Stockholm Convention POPs-a global perspective on the management of Lindane and its waste isomers. Environ Sci Pollut Res 18:152–162
Walker K, Vallero DA, Lewis RG (1999) Factors influencing the distribution of lindane and other hexachlorocyclohexanes in the environment. Environ Sci Technol 33:4373–4378
Willett KL, Ulrich EM, Hites RA (1998) Differential toxicity and environmental fates of hexachlorocyclohexane isomers. Environ Sci Technol 32:2197–2207
Zhang N, Bashir S, Qin J, Schindelka J, Fischer A, Nijenhuis I, Herrmann H, Wick LY, Richnow HH (2014) Compound specific stable isotope analysis (CSIA) to characterize transformation mechanisms of α-hexachlorocyclohexane. J Hazard Mater 280:750–757
Zinder S (1998) Methanogens. In: Burlage RS, Atlas R, Stahl D, Geesy G, Sayler G (eds) Techniques in microbial ecology. Oxford University Press, New York, pp 113–136
Acknowledgements
We thank to Ursula Gunther, Stephanie Hinke and Falk Bratfisch for the technical support. Safdar Bashir was supported by University of Agriculture Faisalabad, Pakistan and the Helmholtz Impulse and Networking Fund through Helmholtz Interdisciplinary Graduate School for Environmental Research (HIGRADE). This study was further funded by the Bundesministerium für Bildung und Forschung (Project INTIME 02WU1221), Germany, and Ministry of Science and Technology, Israel (BMBF-MOST).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bashir, S., Kuntze, K., Vogt, C. et al. Anaerobic biotransformation of hexachlorocyclohexane isomers by Dehalococcoides species and an enrichment culture. Biodegradation 29, 409–418 (2018). https://doi.org/10.1007/s10532-018-9838-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-018-9838-9