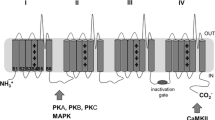
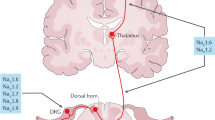
Summary
Drugs that block voltage-gated sodium channels are efficacious in the management of neuropathic pain. Accordingly, this class of ion channels has been a major focus of analgesic research both in academia and in the pharmaceutical/biotechnology industry. In this article, we review the history of the use of sodium channel blockers, describe the current status of sodium channel drug discovery, highlight the challenges and hurdles to attain sodium channel subtype selectivity, and review the potential usefulness of selective sodium channel blockers in neuropathic pain.
Article PDF
Similar content being viewed by others
References
Mitchell SW. Gunshot Wounds and Other Injuries of Nerves. Philadelphia: Lippincott, 1864.
Rivera MA, Aufderheide AC, Cartmell LW, Torres CM, Langsjoen O. Antiquity of coca-leaf chewing in the south central Andes: a 3,000 year archaeological record of coca-leaf chewing from northern Chile. J Psychoactive Drugs 2005;37: 455–458.
Fairley HB. [Anesthesia in the Inca empire]. Rev Esp Anestesiol Reanim 2007;54: 556–562.
Gaedcke F. Ueber das Erythroxylin, dargestellt aus den Blattern des in Sudamerika cultivirten Strauches Erythroxylon Coca Lam. Archiv der Pharmazie 1855;132: 141–150.
Ruetsch YA, Boni T, Borgeat A. From cocaine to ropivacaine: the history of local anesthetic drugs. Curr Top Med Chem 2001;1: 175–182.
Jones AE. Sigmund Freud: Life and Work. London: Hogarth Press, 1953.
Cocaine Chemistry and New Local Anesthetics. Cal West Med 1926;25:232-233.
Taylor IB, Marks BW, Edmonds G. Intravenous administration of procaine hydrochloride during general anesthesia. Arch Surg 1949;59: 714–723.
Phillips OC, Lyons WB, Harris LC, Nelson AT, Graff TD, Frazier TM. Intravenous lidocaine as an adjunct to general anesthesia: a clinical evaluation. Anesth Analg 1960;39: 317–322.
Bigelow N, Harrison I. General analgesic effects of procaine. J Pharmacol Exp Ther 1944;81: 368–373.
Lemoyne J. [Use of dimethyl-dithio-hydantoin in treatment of certain facial neuralgias.]. Ann Otolaryngol 1950;67: 626–628.
Kugelberg E, Lindblom U. The mechanism of the pain in trigeminal neuralgia. J Neurol Neurosurg Psychiatry 1959;22: 36–43.
Blom S. Trigeminal neuralgia: its treatment with a new anticonvulsant drug (G-32883). Lancet 1962;1: 839–840.
Vaughan Williams EM. Classification of antidysrhythmic drugs. Pharmacol Ther [B] 1975;1: 115–138.
Desanctis RW. Electrical Conversion of Ventricular Tachycardia. Jama 1965;191: 632–636.
Carden NL, Steinhaus JE. Lidocaine in cardiac resuscitation from ventricular fibrillation. Circ Res 1956;4: 680–683.
Vaughan Williams EM. A classification of antiarrhythmic actions reassessed after a decade of new drugs. J Clin Pharmacol 1984; 24: 129–147.
Loeser JD, Ward AA, Jr., White LE, Jr. Chronic deafferentation of human spinal cord neurons. J Neurosurg 1968;29: 48–50.
Burchiel KJ, Wyler AR. Ectopic action potential generation in peripheral trigeminal axons. Exp Neurol 1978;62: 269–281.
Boas RA, Covino BG, Shahnarian A. Analgesic responses to i.v. lignocaine. Br J Anaesth 1982;54: 501–505.
Petersen P, Kastrup J. Dercum’s disease (adiposis dolorosa). Treatment of the severe pain with intravenous lidocaine. Pain 1987;28: 77–80.
Kastrup J, Petersen P, Dejgard A, Angelo HR, Hilsted J. Intravenous lidocaine infusion—a new treatment of chronic painful diabetic neuropathy? Pain 1987;28: 69–75.
Lindstrom P, Lindblom U. The analgesic effect of tocainide in trigeminal neuralgia. Pain 1987;28: 45–50.
Dejgard A, Petersen P, Kastrup J. Mexiletine for treatment of chronic painful diabetic neuropathy. Lancet 1988;1: 9–11.
Watson CP, Evans RJ, Reed K, Merskey H, Goldsmith L, Warsh J. Amitriptyline versus placebo in postherpetic neuralgia. Neurology 1982;32: 671–673.
Leijon G, Boivie J. Central post-stroke pain—a controlled trial of amitriptyline and carbamazepine. Pain 1989;36: 27–36.
Watson CP, Vernich L, Chipman M, Reed K. Nortriptyline versus amitriptyline in postherpetic neuralgia: a randomized trial. Neurology 1998;51: 1166–1171.
Pancrazio JJ, Kamatchi GL, Roscoe AK, Lynch C, 3rd. Inhibition of neuronal Na+ channels by antidepressant drugs. J Pharmacol Exp Ther 1998;284: 208–214.
Barber MJ, Starmer CF, Grant AO. Blockade of cardiac sodium channels by amitriptyline and diphenylhydantoin. Evidence for two use-dependent binding sites. Circ Res 1991;69: 677–696.
Dick IE, Brochu RM, Purohit Y, Kaczorowski GJ, Martin WJ, Priest BT. Sodium channel blockade may contribute to the analgesic efficacy of antidepressants. J Pain 2007;8: 315–324.
Govrin-Lippmann R, Devor M. Ongoing activity in severed nerves: source and variation with time. Brain Res 1978;159: 406–410.
Bennett GJ, Xie YK. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 1988;33: 87–107.
Seltzer Z, Dubner R, Shir Y. A novel behavioral model of neuropathic pain disorders produced in rats by partial sciatic nerve injury. Pain 1990;43: 205–218.
Kim SH, Chung JM. An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain 1992;50: 355–363.
Kajander KC, Wakisaka S, Bennett GJ. Spontaneous discharge originates in the dorsal root ganglion at the onset of a painful peripheral neuropathy in the rat. Neurosci Lett 1992;138: 225–228.
Devor M, Wall PD, Catalan N. Systemic lidocaine silences ectopic neuroma and DRG discharge without blocking nerve conduction. Pain 1992;48: 261–268.
Ambrosio AF, Soares-Da-Silva P, Carvalho CM, Carvalho AP. Mechanisms of action of carbamazepine and its derivatives, oxcarbazepine, BIA 2-093, and BIA 2-024. Neurochem Res 2002; 27: 121–130.
Tecoma ES. Oxcarbazepine. Epilepsia 1999;40 Suppl 5: S37–46.
Dogra S, Beydoun S, Mazzola J, Hopwood M, Wan Y. Oxcarbazepine in painful diabetic neuropathy: a randomized, placebo-controlled study. Eur J Pain 2005;9: 543–554.
Grosskopf J, Mazzola J, Wan Y, Hopwood M. A randomized, placebo-controlled study of oxcarbazepine in painful diabetic neuropathy. Acta Neurol Scand 2006;114: 177–180.
Errington AC, Stohr T, Heers C, Lees G. The investigational anticonvulsant lacosamide selectively enhances slow inactivation of voltage-gated sodium channels. Mol Pharmacol 2008;73: 157–169.
Wallace MS, Rowbotham M, Bennett GJ, Jensen TS, Pladna R, Quessy S. A multicenter, double-blind, randomized, placebo-controlled crossover evaluation of a short course of 4030W92 in patients with chronic neuropathic pain. J Pain 2002;3: 227–233.
Hagen NA, du Souich P, Lapointe B, et al. Tetrodotoxin for moderate to severe cancer pain: a randomized, double blind, parallel design multicenter study. J Pain Symptom Manage 2008; 35: 420–429.
Bach FW, Jensen TS, Kastrup J, Stigsby B, Dejgard A. The effect of intravenous lidocaine on nociceptive processing in diabetic neuropathy. Pain 1990;40: 29–34.
Ragsdale DS, McPhee JC, Scheuer T, Catterall WA. Common molecular determinants of local anesthetic, antiarrhythmic, and anticonvulsant block of voltage-gated Na+ channels. Proc Natl Acad Sci U S A 1996;93: 9270–9275.
Wagner LE, 2nd, Eaton M, Sabnis SS, Gingrich KJ. Meperidine and lidocaine block of recombinant voltage-dependent Na+ channels: evidence that meperidine is a local anesthetic. Anesthesiology 1999;91: 1481–1490.
Wagner LE, 2nd, Gingrich KJ, Kulli JC, Yang J. Ketamine blockade of voltage-gated sodium channels: evidence for a shared receptor site with local anesthetics. Anesthesiology 2001;95: 1406–1413.
Wang GK, Russell C, Wang SY. State-dependent block of voltage-gated Na+ channels by amitriptyline via the local anesthetic receptor and its implication for neuropathic pain. Pain 2004: 110: 166–174.
Yarov-Yarovoy V, Brown J, Shatp EM, Clare JJ, Scheuer T, Catterall WA. Molecular determinants of voltage-dependent gating and binding of pore-blocking drugs in transmembrane segment IIIS6 of the Na(+) channel alpha subunit. J Biol Chem 2001;276: 20–27.
Starmer CF, Grant AO, Strauss HC. Mechanisms of use-dependent block of sodium channels in excitable membranes by local anesthetics. Biophys J 1984;46: 15–27.
Schwarz W, Palade PT, Hille B. Local anesthetics. Effect of pH on use-dependent block of sodium channels in frog muscle. Biophys J 1977;20: 343–368.
Courtney KR. Mechanism of frequency-dependent inhibition of sodium currents in frog myelinated nerve by the lidocaine derivative GEA. J Pharmacol Exp Ther 1975;195: 225–236.
Hille B. Local anesthetics: hydrophilic and hydrophobic pathways for the drug-receptor reaction. J Gen Physiol 1977;69: 497–515.
Hondeghem LM, Katzung BG. Antiarrhythmic agents: the modulated receptor mechanism of action of sodium and calcium channel-blocking drugs. Annu Rev Pharmacol Toxicol 1984: 24: 387–423.
Catterall WA, Goldin AL, Waxman SG. International Union of Pharmacology. XXXIX. Compendium of voltage-gated ion channels: sodium channels. Pharmacol Rev 2003;55: 575–578.
Catterall WA, Goldin AL, Waxman SG. International Union of Pharmacology. XLVII. Nomenclature and Structure-Function Relationships of Voltage-Gated Sodium Channels. Pharmacol Rev 2005;57: 397–409.
Catterall WA, Cestele S, Yarov-Yarovoy V, Yu FH, Konoki K, Scheuer T. Voltage-gated ion channels and gating modifier toxins. Toxicon 2007;49: 124–141.
Bosmans F, Tytgat J. Voltage-gated sodium channel modulation by scorpion alpha-toxins. Toxicon 2007;49: 142–158.
Wang J, Ou SW, Wang YJ, Kameyama M, Kameyama A, Zong ZH. Analysis of four novel variants of Nav1.5/SCN5A cloned from the brain. Neurosci Res 2009.
Ragsdale DS. How do mutant Navl.l sodium channels cause epilepsy? Brain Res Rev 2008;58: 149–159.
Vahedi K, Depienne C, Le Fort D, et al. Elicited repetitive daily blindness: a new phenotype associated with hemiplegic migraine and SCN1A mutations. Neurology 2009;72: 1178–1183.
Scalmani P, Rusconi R, Armatura E, et al. Effects in neocortical neurons of mutations of the Na(v)1.2 Na+ channel causing benign familial neonatal-infantile seizures. J Neurosci 2006;26: 10100–10109.
Colosimo E, Gambardella A, Mantegazza M, et al. Electroclinical features of a family with simple febrile seizures and temporal lobe epilepsy associated with SCN1A loss-of-function mutation. Epilepsia 2007;48: 1691–1696.
Fujiwara T. Clinical spectrum of mutations in SCN1A gene: severe myoclonic epilepsy in infancy and related epilepsies. Epilepsy Res 2006;70 Suppl 1: S223–230.
Fujiwara T, Sugawara T, Mazald-Miyazaki E, et al. Mutations of sodium channel alpha subunit type 1 (SCN1A) in intractable childhood epilepsies with frequent generalized tonic-clonic seizures. Brain 2003;126: 531–546.
Lossin C. A catalog of SCN1A variants. Brain Dev 2009;31: 114–130.
Lossin C, Rhodes TH, Desai RR, et al. Epilepsy-associated dysfunction in the voltage-gated neuronal sodium channel SCN1A. J Neurosci 2003: 23: 11289–11295.
Mantegazza M, Gambardella A, Rusconi R, et al. Identification of an Nav1.1 sodium channel (SCN1A) loss-of-function mutation associated with familial simple febrile seizures. Proc Natl Acad Sci U S A 2005;102: 18177–18182.
Meisler MH, Kearney JA. Sodium channel mutations in epilepsy and other neurological disorders. J Clin Invest 2005;115: 2010–2017.
Sugawara T, Mazald-Miyazaki E, Fukushima K, et al. Frequent mutations of SCN1A in severe myoclonic epilepsy in infancy. Neurology 2002;58: 1122–1124.
Vanoye CG, Lossin C, Rhodes TH, George AL, Jr. Single-channel properties of human NaV1.1 and mechanism of channel dysfunction in SCN1A-associated epilepsy. J Gen Physiol 2006;127: 1–14.
Jurkat-Rott K, Lehmann-Horn F. Genotype-phenotype correlation and therapeutic rationale in hyperkalemic periodic paralysis. Neurotherapeutics 2007;4: 216–224.
Lehmann-Horn F, Jurkat-Rott K, Rudel R. Periodic paralysis: understanding channelopathies. Curr Neurol Neurosci Rep 2002; 2: 61–69.
Sternberg D, Maisonobe T, Jurkat-Rott K, et al. Hypokalaemic periodic paralysis type 2 caused by mutations at codon 672 in the muscle sodium channel gene SCN4A. Brain 2001: 124: 1091–1099.
Rogart RB, Cribbs LL, Muglia LK, Kephart DD, Kaiser MW. Molecular cloning of a putative tetrodotoxin-resistant rat heart Na+ channel isoform. Proc Natl Acad Sci U S A 1989: 86: 8170–8174.
Remme CA, Wilde AA, Bezzina CR. Cardiac sodium channel overlap syndromes: different faces of SCN5A mutations. Trends Cardiovasc Med 2008;18: 78–87.
Zimmer T, Surber R. SCN5A channelopathies—an update on mutations and mechanisms. Prog Biophys Mol Biol 2008: 98: 120–136.
McKinney BC, Chow CY, Meisler MH, Murphy GG. Exaggerated emotional behavior in mice heterozygous null for the sodium channel Scn8a (Nav1.6). Genes Brain Behav 2008;7: 629–638.
Chen YH, Dale TJ, Romanos MA, Whitaker WR, Xie XM, Clare JJ. Cloning, distribution and functional analysis of the type III sodium channel from human brain. Eur J Neurosci 2000;12: 4281–4289.
Black JA, Liu S, Tanaka M, Cummins TR, Waxman SG. Changes in the expression of tetrodotoxin-sensitive sodium channels within dorsal root ganglia neurons in inflammatory pain. Pain 2004;108: 237–247.
Dib-Hajj SD, Fjell J, Cummins TR, et al. Plasticity of sodium channel expression in DRG neurons in the chronic constriction injury model of neuropathic pain. Pain 1999;83: 591–600.
Waxman SG, Kocsis JD, Black JA. Type III sodium channel mRNA is expressed in embryonic but not adult spinal sensory neurons, and is reexpressed following axotomy. J Neurophysiol 1994;72: 466–470.
Cummins TR, Waxman SG. Downregulation of tetrodotoxin-resistant sodium currents and upregulation of a rapidly repriming tetrodotoxin-sensitive sodium current in small spinal sensory neurons after nerve injury. J Neurosci 1997;17: 3503–3514.
Fukuoka T, Kobayashi K, Yamanaka H, Obata K, Dai Y, Noguchi K. Comparative study of the distribution of the alpha-subunits of voltage-gated sodium channels in normal and axotomized rat dorsal root ganglion neurons. J Comp Neurol 2008;510: 188–206.
Zhao P, Waxman SG, Hains BC. Sodium channel expression in the ventral posterolateral nucleus of the thalamus after peripheral nerve injury. Mol Pain 2006;2: 27.
Guo F, Yu N, Cai JQ, et al. Voltage-gated sodium channel Nav1.1, Nav1.3 and beta1 subunit were up-regulated in the hippocampus of spontaneously epileptic rat. Brain Res Bull 2008; 75: 179–187.
Bartolomei F, Gastaldi M, Massacrier A, Planells R, Nicolas S, Cau P. Changes in the mRNAs encoding subtypes I, II and III sodium channel alpha subunits following kainate-induced seizures in rat brain. J Neurocytol 1997;26: 667–678.
Nassar MA, Baker MD, Levato A, et al. Nerve injury induces robust allodynia and ectopic discharges in Nav1.3 null mutant mice. Mol Pain 2006;2: 33.
Hains BC, Saab CY, Klein JP, Craner MJ, Waxman SG. Altered sodium channel expression in second-order spinal sensory neurons contributes to pain after peripheral nerve injury. J Neurosci 2004;24: 4832–4839.
Lindia JA, Kohler MG, Martin WJ, Abbadie C. Relationship between sodium channel NaV1.3 expression and neuropathic pain behavior in rats. Pain 2005;117: 145–153.
Sangameswaran L, Fish LM, Koch BD, et al. A novel tetrodotoxin-sensitive, voltage-gated sodium channel expressed in rat and human dorsal root ganglia. J Biol Chem 1997;272: 14805–14809.
Toledo-Aral JJ, Moss BL, He ZJ, et al. Identification of PN1, a predominant voltage-dependent sodium channel expressed principally in peripheral neurons. Proc Natl Acad Sci U S A 1997; 94: 1527–1532.
Thakor DK, Lin A, Matsuka Y, et al. Increased peripheral nerve excitability and local NaV1.8 mRNA up-regulation in painful neuropathy. Mol Pain 2009;5: 14.
Kretschmer T, Happel LT, England JD, et al. Accumulation of PN1 and PN3 sodium channels in painful human neuroma-evidence from immunocytochemistry. Acta Neurochir (Wien) 2002; 144: 803–810; discussion 810.
Cummins TR, Howe JR, Waxman SG. Slow closed-state inactivation: a novel mechanism underlying ramp currents in cells expressing the hNE/PN1 sodium channel. J Neurosci 1998;18: 9607–9619.
Nassar MA, Stirling LC, Forlani G, et al. Nociceptor-specific gene deletion reveals a major role for Nav1.7 (PN1) in acute and inflammatory pain. Proc Natl Acad Sci U S A 2004;101: 12706–12711.
Nassar MA, Levato A, Stirling LC, Wood JN. Neuropathic pain develops normally in mice lacking both Nav1.7 and Nav1.8. Mol Pain 2005;1: 24.
Djouhri L, Fang X, Okuse K, Wood JN, Berry CM, Lawson SN. The TTX-resistant sodium channel Nav1.8 (SNS/PN3): expression and correlation with membrane properties in rat nociceptive primary afferent neurons. J Physiol 2003;550: 739–752.
Devor M. Ectopic discharge in Abeta afferents as a source of neuropathic pain. Exp Brain Res 2009;196: 115–128.
Fertleman CR, Baker MD, Parker KA, et al. SCN9A mutations in paroxysmal extreme pain disorder: allelic variants underlie distinct channel defects and phenotypes. Neuron 2006;52: 767–774.
Yang Y, Wang Y, Li S, et al. Mutations in SCN9A, encoding a sodium channel alpha subunit, in patients with primary erythermalgia. J Med Genet 2004;41: 171–174.
Cox JJ, Reimann F, Nicholas AK, et al. An SCN9A channelopathy causes congenital inability to experience pain. Nature 2006; 444: 894–898.
Goldberg YP, MacFarlane J, MacDonald ML, et al. Loss-of-function mutations in the Nav1.7 gene underlie congenital indifference to pain in multiple human populations. Clin Genet 2007; 71: 311–319.
Nilsen KB, Nicholas AK, Woods CG, Mellgren SI, Nebuchennykh M, Aasly J. Two novel SCN9A mutations causing insensitivity to pain. Pain 2009;143: 155–158.
Ahmad S, Dahllund L, Eriksson AB, et al. A stop codon mutation in SCN9A causes lack of pain sensation. Hum Mol Genet 2007; 16: 2114–2121.
Drenth JP, Waxman SG. Mutations in sodium-channel gene SCN9A cause a spectrum of human genetic pain disorders. J Clin Invest 2007;117: 3603–3609.
Cheng X, Dib-Hajj SD, Tyrrell L, Waxman SG. Mutation I136V alters electrophysiological properties of the Na(v)1.7 channel in a family with onset of erythromelalgia in the second decade. Mol Pain 2008;4: 1.
Choi JS, Dib-Hajj SD, Waxman SG. Inherited erythermalgia: limb pain from an S4 charge-neutral Na channelopathy. Neurology 2006;67: 1563–1567.
Cummins TR, Dib-Hajj SD, Waxman SG. Electrophysiological properties of mutant Nav1.7 sodium channels in a painful inherited neuropathy. J Neurosci 2004;24: 8232–8236.
Dib-Hajj SD, Rush AM, Cummins TR, et al. Gain-of-function mutation in Nav1.7 in familial erythromelalgia induces bursting of sensory neurons. Brain 2005;128: 1847–1854.
Han C, Rush AM, Dib-Hajj SD, et al. Sporadic onset of erythermalgia: a gain-of-function mutation in Nav1.7. Ann Neurol 2006;59: 553–558.
Harty TP, Dib-Hajj SD, Tyrrell L, et al. Na(V)1.7 mutant A863P in erythromelalgia: effects of altered activation and steady-state inactivation on excitability of nociceptive dorsal root ganglion neurons. J Neurosci 2006;26: 12566–12575.
Lampert A, Dib-Hajj SD, Tyrrell L, Waxman SG. Size matters: Erythromelalgia mutation S241T in Nav1.7 alters channel gating. J Biol Chem 2006;281: 36029–36035.
Sheets PL, Jackson JO, 2nd, Waxman SG, Dib-Hajj SD, Cummins TR. A Nav1.7 channel mutation associated with hereditary erythromelalgia contributes to neuronal hyperexcitability and displays reduced lidocaine sensitivity. J Physiol 2007;581: 1019–1031.
Dib-Hajj SD, Yang Y, Waxman SG. Genetics and molecular pathophysiology of Na(v)1.7-related pain syndromes. Adv Genet 2008;63: 85–110.
Jarecki BW, Sheets PL, Jackson IIJO, Cummins TR. Paroxysmal Extreme Pain Disorder mutations within the D3/S4-S5 Linker of Nav1.7 cause moderate destabilization of fast-inactivation. J Physiol 2008:jphysiol.2008.154906.
Zimmermann K, Leffler A, Babes A, et al. Sensory neuron sodium channel Nav1.8 is essential for pain at low temperatures. Nature 2007;447: 855–858.
Abrahamsen B, Zhao J, Asante CO, et al. The Cell and Molecular Basis of Mechanical, Cold, and Inflammatory Pain. Science 2008; 321: 702–705.
Okuse K, Chaplan SR, McMahon SB, et al. Regulation of expression of the sensory neuron-specific sodium channel SNS in inflammatory and neuropathic pain. Mol Cell Neurosci 1997;10: 196–207.
Decosterd I, Ji RR, Abdi S, Tate S, Woolf CJ. The pattern of expression of the voltage-gated sodium channels Na(v)1.8 and Na(v)1.9 does not change in uninjured primary sensory neurons in experimental neuropathic pain models. Pain 2002;96: 269–277.
Novakovic SD, Tzoumaka E, McGivern JG, et al. Distribution of the tetrodotoxin-resistant sodium channel PN3 in rat sensory neurons in normal and neuropathic conditions. J Neurosci 1998;18: 2174–2187.
Dong XW, Goregoaker S, Engler H, et al. Small interfering RNA-mediated selective knockdown of Na(V)1.8 tetrodotoxin-resistant sodium channel reverses mechanical allodynia in neuropathic rats. Neuroscience 2007;146: 812–821.
Joshi SK, Mikusa JP, Hernandez G, et al. Involvement of the TTX-resistant sodium channel Nav 1.8 in inflammatory and neuropathic, but not post-operative, pain states. Pain 2006; 123: 75–82.
Lai J, Gold MS, Kim CS, et al. Inhibition of neuropathic pain by decreased expression of the tetrodotoxin-resistant sodium channel, NaV1.8. Pain 2002;95: 143–152.
Roza C, Laird JM, Souslova V, Wood JN, Cervero F. The tetrodotoxin-resistant Na+ channel Nav1.8 is essential for the expression of spontaneous activity in damaged sensory axons of mice. J Physiol 2003;550: 921–926.
Cummins TR, Dib-Hajj SD, Black JA, Akopian AN, Wood JN, Waxman SG. A novel persistent tetrodotoxin-resistant sodium current in SNS-null and wild-type small primary sensory neurons. J Neurosci 1999;19: RC43.
Dib-Hajj SD, Tyrrell L, Cummins TR, Black JA, Wood PM, Waxman SG. Two tetrodotoxin-resistant sodium channels in human dorsal root ganglion neurons. FEBS Lett 1999;462: 117–120.
Dib-Hajj S, Black JA, Cummins TR, Waxman SG. NaN/Nav1.9: a sodium channel with unique properties. Trends Neurosci 2002; 25: 253–259.
Dib-Hajj SD, Tyrrell L, Black JA, Waxman SG. NaN, a novel voltage-gated Na channel, is expressed preferentially in peripheral sensory neurons and down-regulated after axotomy. Proc Natl Acad Sci U S A 1998;95: 8963–8968.
Tate S, Benn S, Hick C, et al. Two sodium channels contribute to the TTX-R sodium current in primary sensory neurons. Nat Neurosci 1998;1: 653–655.
Porreca F, Lai J, Bian D, et al. A comparison of the potential role of the tetrodotoxin-insensitive sodium channels, PN3/SNS and NaN/SNS2, in rat models of chronic pain. Proc Natl Acad Sci U S A 1999;96: 7640–7644.
Priest BT, Murphy BA, Lindia JA, et al. Contribution of the tetrodotoxjn-resistant voltage-gated sodium channel NaV1.9 to sensory transmission and nociceptive behavior. Proc Natl Acad Sci U S A 2005;102: 9382–9387.
Smith ESJ, Momin A. Persistent pain: the contribution of NaV1.9. J Physiol 2008:jphysiol.2008.152520.
Jarvis MF, Honore P, Shieh CC, et al. A-803467, a potent and selective Nav1.8 sodium channel blocker, attenuates neuropathic and inflammatory pain in the rat. Proc Natl Acad Sci U S A 2007; 104: 8520–8525.
Kort ME, Drizin I, Gregg RJ, et al. Discovery and biological evaluation of 5-aryl-2-furfuramides, potent and selective blockers of the Nav1.8 sodium channel with efficacy in models of neuropathic and inflammatory pain. J Med Chem 2008: 51: 407–416.
McGaraughty S, Chu KL, Scanio MJ, Kort ME, Faltynek CR, Jarvis MF. A selective Nav1.8 sodium channel blocker, A-803467 [5-(4-chlorophenyl-N-(3,5-dimethoxyphenyl)furan-2-carboxamide], attenuates spinal neuronal activity in neuropathic rats. J Pharmacol Exp Ther 2008;324: 1204–1211.
Felix JP, Williams BS, Priest BT, et al. Functional assay of voltage-gated sodium channels using membrane potential-sensitive dyes. Assay Drug Dev Technol 2004;2: 260–268.
Hoyt SB, London C, Gorin D, et al. Discovery of a novel class of benzazepinone Na(v)1.7 blockers: potential treatments for neuropathic pain. Bioorg Med Chem Lett 2007;17: 4630–4634.
Hoyt SB, London C, Ok H, et al. Benzazepinone Na(v)1.7 blockers: Potential treatments for neuropathic pain. Bioorg Med Chem Lett 2007.
Williams BS, Felix JP, Priest BT, et al. Characterization of a new class of potent inhibitors of the voltage-gated sodium channel Nav1.7. Biochemistry 2007;46: 14693–14703.
London C, Hoyt SB, Parsons WH, et al. Imidazopyridines: a novel class of hNav1.7 channel blockers. Bioorg Med Chem Lett 2008;18: 1696–1701.
Liu CJ, Priest BT, Bugianesi RM, et al. A high-capacity membrane potential FRET-based assay for NaV1.8 channels. Assay Drag Dev Technol 2006;4: 37–48.
Trivedi S, Dekermendjian K, Julien R, et al. Cellular HTS Assays for Pharmacological Characterization of Na(V)1.7 Modulators. Assay Drug Dev Technol 2007.
Vickery RG, Amagasu SM, Chang R, et al. Comparison of the pharmacological properties of rat Na(V)1.8 with rat Na(V)1.2a and human Na(V)1.5 voltage-gated sodium channel subtypes using a membrane potential sensitive dye and FLIPR. Receptors Channels 2004;10: 11–23.
Benjamin ER, Pruthi F, Olanrewaju S, et al. State-dependent compound inhibition of Nav1.2 sodium channels using the FLIPR Vm dye: on-target and off-target effects of diverse pharmacological agents. J Biomol Screen 2006;11: 29–39.
Bhattacharya A, Wang Q, Wu N, Chaplan SR, Wickenden AD. Assay dependent activity of the sodium channel gating modifier protoxin-I: implications for sodium channel drug discovery. In: Experimental Biology; 2009; New Orleans, LA, 2009.
Middleton RE, Warren VA, Kraus RL, et al. Two tarantula peptides inhibit activation of multiple sodium channels. Biochemistry 2002;41: 14734–14747.
Castle N, Printzenhoff D, Zellmer S, Antonio B, Wickenden A, Silvia C. Sodium channel inhibitor drug discovery using automated high throughput electrophysiology platforms. Comb Chem High Throughput Screen 2009;12: 107–122.
Schmalhofer W, Calhoun J, Burrows R, et al. ProTx-II, a selective inhibitor of NaV1.7 sodium channels, blocks action potential propagation in nociceptors. Mol Pharmacol 2008:mol.108.047670.
Escoubas P, Bosmans F. Spider peptide toxins as leads for drug development. Expert Opinion on Drug Discovery 2007;2: 823–835.
Xiao Y, Bingham JP, Zhu W, Moczydlowski E, Liang S, Cummins TR. Tarantula huwentoxin-IV inhibits neuronal sodium channels by binding to receptor site 4 and trapping the domain ii voltage sensor in the closed configuration. J Biol Chem 2008; 283: 27300–27313.
Bosmans F, Martin-Eauclaire MF, Swartz KJ. Deconstructing voltage sensor function and pharmacology in sodium channels. Nature 2008;456: 202–208.
Sokolov S, Kraus RL, Scheuer T, Catterall WA. Inhibition of Sodium Channel Gating by Trapping the Domain II Voltage Sensor with Protoxin II. Mol Pharmacol 2008;73: 1020–1028.
Smith JJ, Cummins TR, Alphy S, Blumenthal KM. Molecular Interactions of the Gating Modifier Toxin ProTx-II with Nav1.5: Implied existence of a novel toxin binding site coupled to activation. J Biol Chem 2007;282: 12687–12697.
Dib-Hajj SD, Binshtok AM, Cummins TR, Jarvis MF, Samad T, Zimmermann K. Voltage-gated sodium channels in pain states: Role in pathophysiology and targets for treatment. Brain Res Rev 2009: 60: 65–83.
Waxman SG. Channel, neuronal and clinical function in sodium channelopathies: from genotype to phenotype. Nat Neurosci 2007;10: 405–409.
Cummins TR, Rush AM. Voltage-gated sodium channel blockers for the treatment of neuropathic pain. Expert Rev Neurother 2007;7: 1597–1612.
Cummins TR, Sheets PL, Waxman SG. The roles of sodium channels in nociception: Implications for mechanisms of pain. Pain 2007;131: 243–257.
Wood JN, Boorman J. Voltage-gated sodium channel blockers; target validation and therapeutic potential. Curr Top Med Chem 2005;5: 529–537.
Wood JN, Boorman JP, Okuse K, Baker MD. Voltage-gated sodium channels and pain pathways. J Neurobiol 2004;61: 55–71.
England S. Voltage-gated sodium channels: the, search for subtype-selective analgesics. Expert Opinion on Investigational Drugs 2008;17: 1849–1864.
Momin A, Wood JN. Sensory neuron voltage-gated sodium channels as analgesic drag targets. Curr Opin Neurobiol 2008: 18: 383–388.
Priest BT, Kaczorowski GJ. Blocking sodium channels to treat neuropathic pain. Expert Opin Ther Targets 2007;11: 291–306.
Hargus NJ, Patel MK. Voltage-gated Na+ channels in neuropathic pain. Expert Opinion on Investigational Drugs 2007;16: 635–646.
Catterall WA. From ionic currents to molecular mechanisms: the structure and function of voltage-gated sodium channels. Neuron 2000;26: 13–25.
Kalso E, Tasmuth T, Neuvonen PJ. Amitriptyline effectively relieves neuropathic pain following treatment of breast cancer. Pain 1996;64: 293–302.
Kautio AL, Haanpaa M, Saarto T, Kalso E. Amitriptyline in the treatment of chemotherapy-induced neuropathic symptoms. J Pain Symptom Manage 2008;35: 31–39.
Campbell FG, Graham JG, Zilkha KJ. Clinical trial of carbazepine (tegretol) in trigeminal neuralgia. J Neurol Neurosurg Psychiatry 1966;29: 265–267.
Killian JM, Fromm GH. Carbamazepine in the treatment of neuralgia. Use of side effects. Arch Neurol 1968;19: 129–136.
Rull JA, Quibrera R, Gonzalez-Millan H, Lozano Castaneda O. Symptomatic treatment of peripheral diabetic neuropathy with carbamazepine (Tegretol): double blind crossover trial. Diabetologia 1969;5: 215–218.
Harke H, Gretenkort P, Ladleif HU, Rahman S, Harke O. The response of neuropathic pain and pain in complex regional pain syndrome 1 to carbamazepine and sustained-release morphine in patients pretreated with spinal cord stimulation: a double-blinded randomized study. Anesth Analg 2001;92: 488–495.
Kishore-Kumar R, Max MB, Schafer SC, et al. Desipramine relieves postherpetic neuralgia. Clin Pharmacol Ther 1990;47: 305–312.
Max MB, Kishore-Kumar R, Schafer SC, et al. Efficacy of desipramine in painful diabetic neuropathy: a placebo-controlled trial. Pain 1991;45: 3–9; discussion 1–2.
Goldstein DJ, Lu Y, Detke MJ, Lee TC, Iyengar S. Duloxetine vs. placebo in patients with painful diabetic neuropathy. Pain 2005; 116: 109–118.
Raskin J, Pritchett YL, Wang F, et al. A double-blind, randomized multicenter trial comparing duloxetine with placebo in the management of diabetic peripheral neuropathic pain. Pain Med 2005;6: 346–356.
Wernicke IF, Pritchett YL, D’Souza DN, et al. A randomized controlled trial of duloxetine in diabetic peripheral neuropathic pain. Neurology 2006;67: 1411–1420.
Rauck RL, Shaibani A, Biton V, Simpson J, Koch B. Lacosamide in painful diabetic peripheral neuropathy: a phase 2 double-blind placebo-controlled study. Clin J Pain 2007;23: 150–158.
Shaibani A, Fares S, Selam IL, et al. Lacosamide in Painful Diabetic Neuropathy: An 18-Week Double-Blind Placebo-Controlled Trial. J Pain 2009.
Wymer JP, Simpson J, Sen D, Bongardt S. Efficacy and safety of lacosamide in diabetic neuropathic pain: an 18-week double-blind placebo-controlled trial of fixed-dose regimens. Clin J Pain 2009; 25: 376–385.
Zakrzewska JM, Chaudhry Z, Nurmikko TJ, Patton DW, Mullens EL. Lamotrigine (lamictal) in refractory trigeminal neuralgia: results from a double-blind placebo controlled crossover trial. Pain 1997;73: 223–230.
McCleane G. 200 mg daily of lamotrigine has no analgesic effect in neuropathic pain: a randomised, double-blind, placebo controlled trial. Pain 1999: 83: 105–107.
Eisenberg E, Lurie Y, Braker C, Daoud D, Ishay A. Lamotrigine reduces painful diabetic neuropathy: a randomized, controlled study. Neurology 2001;57: 505–509.
Vestergaard K, Andersen G, Gottrup H, Kristensen BT, Jensen TS. Lamotrigine for central poststroke pain: a randomized controlled trial. Neurology 2001;56: 184–190.
Finnerup NB, Sindrup SH, Bach FW, Johannesen IL, Jensen TS. Lamotrigine in spinal cord injury pain: a randomized controlled trial. Pain 2002: 96: 375–383.
Simpson DM, McArthur JC, Olney R, et al. Lamotrigine for HIV-associated painful sensory neuropathies: a placebo-controlled trial. Neurology 2003;60: 1508–1514.
Rao RD, Flynn PJ, Sloan JA, et al. Efficacy of lamotrigine in the management of chemotherapy-induced peripheral neuropathy: a phase 3 randomized, double-blind, placebo-controlled trial, N01C3. Cancer 2008;112: 2802–2808.
Kastrup I, Bach FW, Petersen P, et al. Lidocaine treatment of painful diabetic neuropathy and endogenous opioid peptides in plasma. Clin J Pain 1989: 5: 239–244.
Ellemann K, Sjogren P, Banning AM, Jensen TS, Smith T, Geertsen P. Trial of intravenous lidocaine on painful neuropathy in cancer patients. Clin J Pain 1989: 5: 291–294.
Rowbotham MC, Reisner-Keller LA, Fields HL. Both intravenous lidocaine and morphine reduce the pain of postherpetic neuralgia. Neurology 1991: 41: 1024–1028.
Bruera E, Ripamonti C, Brenneis C, Macmillan K, Hanson J. A randomized double-blind crossover trial of intravenous lidocaine in the treatment of neuropathic cancer pain. J Pain Symptom Manage 1992;7: 138–140.
Medrik-Goldberg T, Lifschitz D, Pud D, Adler R, Eisenberg E. Intravenous lidocaine, amantadine, and placebo in the treatment of sciatica: a double-blind, randomized, controlled study. Reg Anesth Pain Med 1999;24: 534–540.
Wallace MS, Ridgeway BM, Leung AY, Gerayli A, Yaksh TL. Concentration-effect relationship of intravenous lidocaine on the allodynia of complex regional pain syndrome types I and II. Anesthesiology 2000;92: 75–83.
Attal N, Gaude V, Brasseur L, et al. Intravenous lidocaine in central pain: a double-blind, placebo-controlled, psychophysical study. Neurology 2000;54: 564–574.
Finnerup NB, Biering-Sorensen F, Johannesen IL, et al. Intravenous lidocaine relieves spinal cord injury pain: a randomized controlled trial. Anesthesiology 2005;102: 1023–1030.
Tremont-Lukats IW, Hutson PR, Backonja MM. A randomized, double-masked, placebo-controlled pilot trial of extended IV lidocaine infusion for relief of ongoing neuropathic pain. Clin J Pain 2006: 22: 266–271.
Backonja M, Gombar KA. Response of central pain syndromes to intravenous lidocaine. I Pain Symptom Manage 1992;7: 172–178.
Attal N, Rouaud J, Brasseur L, Chauvin M, Bouhassira D. Systemic lidocaine in pain due to peripheral nerve injury and predictors of response. Neurology 2004;62: 218–225.
Sakurai M, Kanazawa I. Positive symptoms in multiple sclerosis: their treatment with sodium channel blockers, lidocaine and mexiletine. J Neurol Sci 1999;162: 162–168.
Chabal C, Jacobson L, Russell LC, Burchiel KJ. Pain responses to perineuromal injection of normal saline, gallamine, and lidocaine in humans. Pain 1989;36: 321–325.
Chabal C, Jacobson L, Mariano A, Chaney E, Britell CW. The use of oral mexiletine for the treatment of pain after peripheral nerve injury. Anesthesiology 1992;76: 513–517.
Stracke H, Meyer UE, Schumacher HE, Federlin K. Mexiletine in the treatment of diabetic neuropathy. Diabetes Care 1992;15: 1550–1555.
Chiou-Tan FY, Tuel SM, Johnson JC, Priebe MM, Hirsh DD, Strayer JR. Effect of mexiletine on spinal cord injury dysesthetic pain. Am J Phys Med Rehabil 1996;75: 84–87.
Oskarsson P, Ljunggren JG, Lins PE. Efficacy and safety of mexiletine in the treatment of painful diabetic neuropathy. The Mexiletine Study Group. Diabetes Care 1997;20: 1594–1597.
Wright JM, Oki JC, Graves L, 3rd. Mexiletine in the symptomatic treatment of diabetic peripheral neuropathy. Ann Pharmacother 1997;31: 29–34.
Wallace MS, Magnuson S, Ridgeway B. Efficacy of oral mexiletine for neuropathic pain with allodynia: a double-blind, placebo-controlled, crossover study. Reg Anesth Pain Med 2000;25: 459–467.
Saudek CD, Werns S, Reidenberg MM. Phenytoin in the treatment of diabetic symmetrical polyneuropathy. Clin Pharmacol Ther 1977;22: 196–199.
Chadda VS, Mathur MS. Double blind study of the effects of diphenylhydantoin sodium on diabetic neuropathy. I Assoc Physicians India 1978: 26: 403–406.
Galer BS, Twilling LL, Harle J, Cluff RS, Friedman E, Rowbotham MC. Lack of efficacy of riluzole in the treatment of peripheral neuropathic pain conditions. Neurology 2000;55: 971–975.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhattacharya, A., Wickenden, A.D. & Chaplan, S.R. Sodium channel blockers for the treatment of neuropathic pain. Neurotherapeutics 6, 663–678 (2009). https://doi.org/10.1016/j.nurt.2009.08.001
Issue Date:
DOI: https://doi.org/10.1016/j.nurt.2009.08.001