Abstract
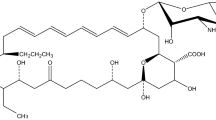
The heterologous production of iso-migrastatin (iso-MGS) was successfully demonstrated in an engineered S. lividans SB11002 strain, which was derived from S. lividans K4-114, following introduction of pBS11001, which harbored the entire mgs biosynthetic gene cluster. However, under similar fermentation conditions, the iso-MGS titer in the engineered strain was significantly lower than that in the native producer — Streptomyces platensis NRRL 18993. To circumvent the problem of low iso-MGS titers and to expand the utility of this heterologous system for iso-MGS biosynthesis and engineering, systematic optimization of the fermentation medium was carried out. The effects of major components in the cultivation medium, including carbon, organic and inorganic nitrogen sources, were investigated using a single factor optimization method. As a result, sucrose and yeast extract were determined to be the best carbon and organic nitrogen sources, resulting in optimized iso-MGS production. Conversely, all other inorganic nitrogen sources evaluated produced various levels of inhibition of iso-MGS production. The final optimized R2YE production medium produced iso-MGS with a titer of 86.5 mg/L, about 3.6-fold higher than that in the original R2YE medium, and 1.5 fold higher than that found within the native S. platensis NRRL 18993 producer.
Similar content being viewed by others
References
Newman, D. J., G. M. Cragg, and K. M. Snader (2003) Natural products as sources of new drugs over the period 1981–2002. J. Nat. Prod. 66: 1022–1037.
Bode, H. B. and R. Muller (2005) The impact of bacterial genomics on natural product research. Angew. Chem. Int. Ed. 44: 6828–6846.
Wendt-Pienkowski, E., Y. Huang, J. Zhang, B. Li, H. Jiang, H. Kwon, C. R. Hutchinson, and B. Shen (2005) Cloning, sequencing, analysis, and heterologous expression of the fredericamycin biosynthetic gene cluster from Streptomyces griseus. J. Am. Chem. Soc. 127: 16442–16452.
Sosio, M., F. Giusino, C. Cappellano, E. Bossi, A. M. Puglia, and S. Donadio (2000) Artificial chromosomes for antibiotic-producing actinomycetes. Nat. Biotechnol. 18: 343–345.
Martinez, A., S. J. Kolvek, C. L. T. Yip, J. Hopke, K. A. Brown, I. A. MacNeil, and M. S. Osburne (2004) Genetically modified bacterial strains and novel bacterial artificial chromosome shuttle vectors for constructing environmental libraries and detecting heterologous natural products in multiple expression hosts. Appl. Env. Microb. 70: 2452–2463.
Zhang, H. R., Y. Wang, and B. A. Pfeifer (2008) Bacterial hosts for natural product production. Mol. Pharmacol. 5: 212–225.
Ju, J., S. K. Lim, H. Jiang, and B. Shen (2005) Migrastatin and dorrigocins are shunt metabolites of iso-migrastain. J. Am. Chem. Soc. 127: 1622–1623.
Shan, D., L. Chen, J. T. Njardarson, C. Gaul, X. Ma, S. J. Danishefsky, and X. Y. Huang (2005) Synthetic analogues of migrastatin that inhibit mammary tumor metastasis in mice. Proc. Natl. Acad. Sci. 102: 3772–3776.
Metaferia, B. B., L. Chen, H. L. Baker, X. Y. Huang, and C. A. Bewley (2007) Synthetic macrolides that inhibit breast cancer cell migration in vitro. J. Am. Chem. Soc. 129: 2434–2435
Reymond, S. and J. Cossy (2008) Migrastatin and analogues: New anti-metastatic agents. Comptes Rendus Chim. 11: 1447–1462.
Ju, J., S. R. Rajski, S. K. Lim, J. W. Seo, N. R. Peters, F. Hoffmann, and B. Shen (2009) Lactimidomycin, iso-migrastatin and related glutarimide-containing 12-membered macrolides are extremely potent inhibitors of cell migration. J. Am. Chem. Soc. 131: 1370–1371.
Nakae, K., Y. Nishimura, S. Ohba, and Y. Akamatsu (2006) Migrastatin acts as a muscarinic acetylcholine receptor antagonist. J. Antibiot. 59: 685–692.
Takemoto, Y., E. Tashiro, and M. Imoto (2006) Suppression of multidrug resistance by migrastatin. J. Antibiot. 59: 435–438.
Lim, S. K., J. Ju, E. Zazopoulos, H. Jiang, J. W. Seo, Y. Chen, Z. Feng, S. R. Rajski, C. M. Farnet, and B. Shen (2009) iso-Migrastatin, migrastatin, and dorrigocin production in Streptomyces platensis NRRL 18993 is governed by single biosynthetic machinery featuring an acyltransferase-less Type I polyketide synthase. J. Biol. Chem. 284: 29746–29756.
Feng, Z., L. Wang, S. R. Rajski, Z. Xu, M. F. Coeffet-LeGal, and B. Shen (2009) Engineered production of iso-migrastatin in heterologous hosts. Bioorg. Med. Chem. 17: 2147–2153.
Ju, J., S. K. Lim, H. Jiang, J. W. Seo, and B. Shen (2005) Isomigrastatin congeners from Streptomyces platensis and generation of a glutarimide polyketide library featuring the dorrigocin, lactimidomycin, migrastatin, and NK 30424 scaffolds. J. Am. Chem. Soc. 127: 11930–11931.
Kieser, T., M. J. Bibb, M. J. Buttner, K. F. Chater, and D. A. Hopwood (2000) Practical Streptomyces genetics: A Laboratory Manual. 2nd Ed. The John Innes Foundation, Norwich, U.K.
Ju, J., S. K. Lim, H. Jiang, J. W. Seo, Y. Her, and B. Shen (2006) Thermolysis of Iso-migrastatin and its congeners via [3,3]-sigmatropic rearrangment: A new route to the synthesis of migrastatin and its analogues. Org. Lett. 8: 5865–5868.
Buese, M., A. Kopmann, H. Diekmann, and M. Thoma (1999) Oxygen, pH value, and carbon sources induced changes of the mode of oscillation in synchronous continuous culture of Saccharomyces cerevisiae. Biotechnol. Bioeng. 63: 410–417.
Nakamura, M. and B. L. Pitsch (1961) Effect of size of inocula on growth of Shigella sonnei in a chemically defined medium. Can. J. Microbiol. 7: 848–849.
Olano, C., F. Lombó, C. Méndez, and A. S. José (2008) Improving production of bioactive secondary metabolites in actinomycetes by metabolic engineering. Metab. Eng. 10: 281–292.
Galm, U. and B. Shen (2006) Expression of biosynthetic gene cluster in heterologous hosts for natural product production and combinatorial biosynthesis. Expert Opin. Drug Discov. 1: 409–437.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, X., Yang, D., Zhu, X. et al. Iso-migrastatin titer improvement in the engineered Streptomyces lividans SB11002 strain by optimization of fermentation conditions. Biotechnol Bioproc E 15, 664–669 (2010). https://doi.org/10.1007/s12257-009-3129-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-009-3129-6