Abstract
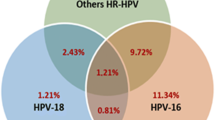
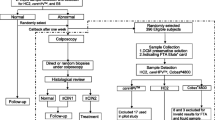
We compared the detection rate of cervical neoplasias between a liquid-based cytology (LBC) method using SurePath and the conventional method. We also studied the feasibility of human papillomavirus (HPV) typing by linear array assay. Cytological specimens from 1551 Japanese women were prepared using the conventional and SurePath methods; the cytological and histological results from biopsy samples were compared. HPV typing using an HPV linear array assay was carried out on residual specimens using the SurePath method. The cytodiagnostic results showed a concordance rate of 85.3% (κ = 0.46) between the two methods. The sensitivity of lesions histopathologically diagnosed as CIN1 or above was not significantly different between the two methods (P = 0.575–1.000). The receiver operating characteristic curve analysis of the detectability in CIN2 or above revealed no significant difference between the two methods (P = 0.096). Among the 44 patients who underwent HPV typing using a linear array assay, 33 samples were eligible for HPV testing and were stored at ambient temperature. In conclusion, the SurePath and conventional methods have equivalent abilities for detecting cervical lesions. After preparation for cytological diagnosis, use of the remaining cells from the SurePath specimens to perform HPV typing using the linear array method could be feasible.
Similar content being viewed by others
References
Doyle B, O’Farrell C, Mahoney E, Turner L, Magee D, Gibbons D. Liquid-based cytology improves productivity in cervical cytology screening. Cytopathology 2006; 17: 60–4.
Kirschner B, Simonsen K, Junge J. Comparison of conventional Papanicolaou smear and SurePath liquid-based cytology in the Copenhagen population screening programme for cervical cancer. Cytopathology 2006; 17: 187–94.
Williams AR. Liquid-based cytology and conventional smears compared over two 12-month periods. Cytopathology 2006; 17: 82–5.
Ronco G, Cuzick J, Pierotti P et al. Accuracy of liquid based versus conventional cytology: overall results of new technologies for cervical cancer screening: randomised controlled trial. BMJ 2007; 335: 28.
Strander B, Andersson-Ellstrom A, Milsom I, Radberg T, Ryd W. Liquid-based cytology versus conventional Papanicolaou smear in an organized screening program: a prospective randomized study. Cancer 2007; 111: 285–91.
Bergeron C, Bishop J, Lemarie A et al. Accuracy of thin-layer cytology in patients undergoing cervical cone biopsy. Acta Cytol 2001; 45: 519–24.
Hessling JJ, Raso DS, Schiffer B, Callicott J Jr, Husain M, Taylor D. Effectiveness of thin-layer preparations vs. conventional Pap smears in a blinded, split-sample study. Extended cytologic evaluation. J Reprod Med 2001; 46: 880–6.
Schledermann D, Ejersbo D, Hoelund B. Improvement of diagnostic accuracy and screening conditions with liquid-based cytology. Diagn Cytopathol 2006; 34: 780–5.
Ferenczy A, Robitaille J, Franco E, Arseneau J, Richart RM, Wright TC. Conventional cervical cytologic smears vs. ThinPrep smears. A paired comparison study on cervical cytology. Acta Cytol 1996; 40: 1136–42.
Sherman ME, Mendoza M, Lee KR et al. Performance of liquid-based, thin-layer cervical cytology: correlation with reference diagnoses and human papillomavirus testing. Mod Pathol 1998; 11: 837–43.
Arbyn M, Bergeron C, Klinkhamer P, Martin-Hirsch P, Siebers AG, Bulten J. Liquid compared with conventional cervical cytology: a systematic review and meta-analysis. Obstet Gynecol 2008; 111: 167–77.
Davey E, Barratt A, Irwig L et al. Effect of study design and quality on unsatisfactory rates, cytology classifications, and accuracy in liquid-based versus conventional cervical cytology: a systematic review. Lancet 2006; 367: 122–32.
Masumoto N, Fujii T, Ishikawa M et al. Papanicolaou tests and molecular analyses using new fluid-based specimen collection technology in 3000 Japanese women. Br J Cancer 2003; 88: 1883–8.
van Hamont D, van Ham MA, Bakkers JM, Massuger LF, Melchers WJ. Evaluation of the SPF10-INNO LiPA human papillomavirus (HPV) genotyping test and the roche linear array HPV genotyping test. J Clin Microbiol 2006; 44: 3122–9.
Jamison J, Wilson RT, Carson J. The evaluation of human papillomavirus genotyping in cervical liquid-based cytology specimens; using the Roche Linear Array HPV genotyping assay. Cytopathology 2009; 20: 242–8.
Benevolo M, Vocaturo A, Mottolese M et al. Clinical role of p16INK4a expression in liquid-based cervical cytology: correlation with HPV testing and histologic diagnosis. Am J Clin Pathol 2008; 129: 606–12.
Halfon P, Benmoura D, Agostini A et al. Relevance of HPV mRNA detection in a population of ASCUS plus women using the NucliSENS EasyQ((R)) HPV assay. J Clin Virol 2009; 47: 177–81.
Lee JK, Kim MK, Song SH et al. Comparison of human papillomavirus detection and typing by hybrid capture 2, linear array, DNA chip, and cycle sequencing in cervical swab samples. Int J Gynecol Cancer 2009; 19: 266–72.
Hardie A, Moore C, Patnick J et al. High-risk HPV detection in specimens collected in SurePath preservative fluid: comparison of ambient and refrigerated storage. Cytopathology 2009; 20: 235–41.
Nayar R, Solomon D. The Bethesda System for Reporting Cervical Cytology — Atlas, 2nd edn. New York: Springer-Verlag, 2004. Available from URL: http://nih.techriver.net/, and Bethesda interobserver reproducibility project.
Taylor S, Kuhn L, Dupree W, Denny L, De Souza M, Wright TC Jr. Direct comparison of liquid-based and conventional cytology in a South African screening trial. IntJ Cancer 2006; 118: 957–62.
Sykes PH, Harker DY, Miller A et al. A randomised comparison of SurePath liquid-based cytology and conventional smear cytology in a colposcopy clinic setting. BJOG 2008; 115: 1375–81.
Obwegeser JH, Brack S. Does liquid-based technology really improve detection of cervical neoplasia? A prospective, randomized trial comparing the ThinPrep Pap Test with the conventional Pap Test, including follow-up of HSIL cases. Acta Cytol 2001; 45: 709–14.
Siebers AG, Klinkhamer PJ, Grefte JM et al. Comparison of liquid-based cytology with conventional cytology for detection of cervical cancer precursors: a randomized controlled trial. JAMA 2009; 302: 1757–64.
Beerman H, van Dorst EB, Kuenen-Boumeester V, Hogendoorn PC. Superior performance of liquid-based versus conventional cytology in a population-based cervical cancer screening program. Gynecol Oncol 2009; 112: 572–6.
Davey E, d’Assuncao J, Irwig L et al. Accuracy of reading liquid based cytology slides using the ThinPrep Imager compared with conventional cytology: prospective study. BMJ 2007; 335: 31.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflicts of interest
The authors have no conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Taoka, H., Yamamoto, Y., Sakurai, N. et al. Comparison of conventional and liquid-based cytology, and human papillomavirus testing using SurePath preparation in Japan. Hum Cell 23, 126–133 (2010). https://doi.org/10.1111/j.1749-0774.2010.00093.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1749-0774.2010.00093.x