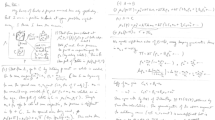Abstract
A pairwise preferential interaction model (PPIM), based on Kirkwood–Buff integrals, is developed to quantify and characterize the interactions between some of the functional groups commonly observed in peptides. The existing experimental data are analyzed to determine the preferential interaction (PI) parameters for different amino acid and small peptide systems in aqueous solutions. The PIs between the different functional groups present in the peptides are then isolated and quantified by assuming simple pairwise additivity. The PPIM approach provides consistent estimates for the pair interactions between the same functional groups obtained from different solute molecules. Furthermore, these interactions appear to be chemically intuitive. It is argued that this type of approach can provide valuable information concerning specific functional group correlations which could give rise to peptide aggregation.
Similar content being viewed by others
References
Ross C.A., Poirier M.A.: Nat. Med. 10, S10–S17 (2004)
Hetz C., Soto C.: Cell. Mol. Life Sci. 60, 133 (2003)
Cohen F.E., Kelly J.W.: Nature 426, 905 (2003)
McQuarrie D.A.: Statistical Mechanics. Harper & Row, New York (1976)
Hill T.L.: Introduction to Statistical Thermodynamics. Addison-Wesley, Reading (1960)
Ben-Naim A.: Statistical Thermodynamics for Chemists and Biochemists. Plenum Press, New York (1992)
Thirumalai D., Klimov D.K., Dima R.I.: Curr. Opin. Struct. Biol. 13, 146 (2003)
Tjernberg L., Hosia W., Bark N., Thyberg J., Johansson J.: J. Biol. Chem. 277, 43243 (2002)
Perutz M.F., Pope B.J., Owen D., Wanker E.E., Scherzinger E.: Proc. Natl. Acad. Sci. USA 99, 5596 (2002)
Wurth C., Guimard N.K., Hecht M.H.: J. Mol. Biol. 319, 1279 (2002)
Ben-Naim A.: Cell Biophys. 12, 255 (1988)
Marcus Y.: J. Chem. Soc. Fara. Trans. 86, 2215 (1990)
Matteoli E., Mansoori G.A.: Fluctuation Theory of Mixtures. Taylor & Francis, New York (1990)
Matteoli E., Lepori L.: J. Chem. Soc. Farad. Trans. 91, 431 (1995)
Zielkiewicz J.: Phys. Chem. Chem. Phys. 2, 2925 (2000)
Smith P.E.: Biophys. J. 91, 849 (2006)
Shulgin I.L., Ruckenstein E.: J. Phys. Chem. B 111, 3990 (2007)
Shimizu S., Boon C.L.: J. Chem. Phys. 121, 9147 (2004)
Chitra R., Smith P.E.: J. Phys. Chem. B 105, 11513 (2001)
Aburi M., Smith P.E.: J. Phys. Chem. B 108, 7382 (2004)
Baynes B.M., Trout B.L.: J. Phys. Chem. B 107, 14058 (2003)
Kang M., Smith P.E.: Fluid Phase Equilib. 256, 14 (2007)
Kirkwood J.G., Buff F.P.: J. Chem. Phys. 19, 774 (1951)
Chou P.Y., Fasman G.D.: Trends Biochem. Sci. 2, 128 (1977)
Matteoli E., Lepori L.: J. Chem. Phys. 80, 2856 (1984)
Chitra R., Smith P.E.: J. Phys. Chem. B 106, 1491 (2002)
Liu H.Q., Ruckenstein E.: J. Phys. Chem. B 102, 1005 (1998)
Rosgen J., Pettitt B.M., Perkyns J., Bolen D.W.: J. Phys. Chem. B 108, 2048 (2004)
Rosgen J., Pettitt B.M., Bolen D.W.: Biochemistry. 43, 14472 (2004)
Smith E.R.B., Smith P.K.: J. Biol. Chem. 117, 209 (1937)
Kuramochi H., Noritomi H., Hoshino D., Nagahama K.: J. Chem. Eng. Data 42, 470 (1997)
Smith E.R.B., Smith P.K.: J. Biol. Chem. 135, 273 (1940)
Zielkiewicz J.: Int. DATA Ser. Sel. Data Mixtures, Ser. A 28, 299 (2000)
Savage J.J., Wood R.H.: J. Solut. Chem. 5, 733 (1976)
Plyasunov A.V., Shock E.L., Wood R.H.: J. Chem. Eng. Data 48, 1463 (2003)
Sedlbauer J., Jakubu P.: Ind. Eng. Chem. Res. 47, 5048 (2008)
Gmehling J.: Pure Appl. Chem. 75, 875 (2003)
Smith P.E., Mazo R.M.: J. Phys. Chem. B 112, 7875 (2008)
Newman K.E.: Chem. Soc. Rev. 23, 31 (1994)
Kusalik P.G., Patey G.N.: J. Chem. Phys. 86, 5110 (1987)
Lei H.X., Smith P.E.: Biophys. J. 85, 3513 (2003)
de la Paz M.L., Goldie K., Zurdo J., Lacroix E., Dobson C.M., Hoenger A., Serrano L.: Proc. Natl. Acad. Sci. USA 99, 16052 (2002)
Chiti F., Stefani M., Taddei N., Ramponi G., Dobson C.M.: Nature 424, 805 (2003)
H. Tsai H., Zanuy D., Haspel N., Gunasekaran K., Ma B.Y., Tsai C.J., Nussinov R.: Biophys. J. 87, 146 (2004)
Rousseau F., Serrano L., Schymkowitz J.W.H.: J. Mol. Biol. 355, 1037 (2006)
Rousseau F., Schymkowitz J., Serrano L.: Curr. Opin. Struct. Biol. 16, 118 (2006)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kang, M., Smith, P.E. A Pairwise Preferential Interaction Model for Understanding Peptide Aggregation. Int J Thermophys 31, 793–804 (2010). https://doi.org/10.1007/s10765-009-0694-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10765-009-0694-z