Summary
A means to avoid the pharmacokinetic problems affecting the anti-epileptic drugs may be their direct intracerebroventricular (ICV) or intracerebral delivery. This approach may achieve a greater drug concentration at the epileptogenic area while minimizing it in other brain or systemic areas, and thus it could be an interesting therapeutic alternative in drug-resistant epilepsies. The objective of this article is to review a series of experiments, ranging from actute ICV injection to continuous intracerebral infusion of anti-epileptic drugs or grafting of neurotransmitter producing cells, in experimental models, especially in the kindling model of epilepsy in the rat.
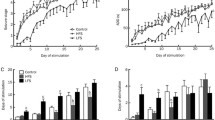
Acute ICV injection of phenytoin, phenobarbital or carbamacepine is able to diminish the intensity of kindling seizures, but it is also associated with a high neurologic toxicity, especially phenobarbital. Continuous ICV infusion of anti-epileptic drugs can effectively control seizures, but neurologic toxicity is not improved compared with systemic delivery. However, systemic toxicity may be improved, as in the case of valproic acid, whose continuous ICV infusion results in very low plasmatic or hepatic drug concentrations. Continuous intracerebral infusion at the epileptogenic area was studied as an alternative to minimize neurologic toxicity. Thus, intra-amygdalar infusion of gamma-aminobutyric acid (GABA) controls seizures with minimal neurotoxicity in amygdala-kindled rats. Similarly, continuous infusion of GABA into the dorsomedian nucleus of the thalamus improves seizure spread, while not affecting the local epileptogenic activity at the amygdala. Grafting of GABA releasing cells may reduce kindling parameter severity without behavioral side effects.
We may conclude that ICV or intracerebral delivery of anti-epileptic drugs or neurotransmitters may be a useful technique to modulate epilepsy.
Article PDF
Similar content being viewed by others
References
Sander JW. The epidemiology of epilepsy revisited. Curr Opin Neurol 2003;16: 165–170.
Regesta G, Tanganelli P. Clinical aspects and biological bases of drug-resistant epilepsies. Epilepsy Res 1999;34: 109–122.
Löscher W, Potschka H. Role of multidrug transporters in pharmacoresistance to antiepileptic drugs. J Pharmacol Exp Ther 2002; 301: 7–14.
Tamargo RJ, Brem H. Drug delivery to the central nervous system: a review. Neurosurg Quart 1992;2: 259–279.
Temsamani J, Scherrmann JM, Rees AR, Kaczorek M. Brain drug delivery technologies: novel approaches for transporting therapeutics. Pharm Sci Technolo Today 2000;3: 155–162.
Jefferys JG. Models and mechanisms of experimental epilepsies. Epilepsia 2003;44(suppl 12): 44–50.
Sarkisian MR. Overview of the current animal models for human seizure and epileptic disorders. Epilepsy Behav 2001;2: 201–216.
Pitkanen A, Schwartzkroin PA, Moshe SL. Models of seizures and epilepsy. New York: Elsevier; 2005.
McIntyre DC, Poulter MO, Gilby K. Kindling: some old and some new. Epilepsy Res 2002;50: 79–92.
Löscher W, Schmidt D. Which animal models should be used in the search for new antiepileptic drugs? A proposal based on experimental and clinical considerations Epilepsy Res 1988;2: 145–181.
Delgado JMR, Sevillano M. Evolution of repeated hippocampal seizures in the cat. Electroenceph Clin Neurophysiol 1961;13: 722–733.
Goddard GV, McIntyre DC, Leech CK. A permanent change in brain function resulting from daily electrical stimulation. Exp Neurol 1969;25: 295–330.
Racine RJ. Modification of seizure activity by electrical stimulation. II. Motor seizure. Electroencephalogr Clin Neurophysiol 1972;32: 281–294.
Hönack D, Löscher W. Kindling increases the sensitivity of rats to adverse effects of certain antiepileptic drugs. Epilepsia 1995;36: 763–771.
Racine RJ, Bumham WM, Gartner JG, Levitan D. Rates of motor seizure development in rats subjected to electrical brain stimulation: strain and inter-stimulation interval effects. Electroencephalogr Clin Neurophysiol 1973;35: 553–556.
Barcia JA, Rubio P, Alós M, Serralta A, Belda V. Anticonvulsant and neurotoxic effects of intracerebroventricular injection of phenytoin, phenobarbital and carbamazepine in an amygdala-kindling model of epilepsy in the rat. Epilepsy Res 1999;33: 159–167.
Serralta A, Barcia JA, Ortiz P, et al. Effect of intracerebroventricular continuous infusion of valproic acid versus single i.p. and i.c.v. injections in the amygdale kindling epilepsy model. Epilepsy Res 2006;70: 15–26.
Bourne JA. Intracerebral microdialysis: 30 years as a tool for the neuroscientist. Clin Exp Pharmacol Physiol 2003;30: 16–24.
Raghavan R, Brady ML, Rodríguez-Ponce MI, Hartlep A, Pedain C, Sampson JH. Convection-enhanced delivery of therapeutics for brain disease, and its optimization. Neurosurg Focus 2006;20(4): E12.
Wang PP, Frazier J, Brem H. Local drug delivery to the brain. Adv Drug Deliv Rev 2002;54: 987–1013.
Vogel TW, Bruce JN. Local delivery methods into the CNS. In: Proctor MR, Black PM, Minimally Invasive Neurosurgery. Totowa, New Jersey: Humana Press; 2005: 297–316.
Levy R, Lang AE, Dostrovsky JO, et al. Lidocaine and muscimol microinjections in subthalamic nucleus reverse Parkinsonian symptoms. Brain 2001;124: 2105–2118.
Malpeli JG. Reversible inactivation of subcortical sites by drug injection. J Neurosci Methods 1999;86: 119–128.
Fisher RS, Ho J. Potential new methods for antiepileptic drug delivery. CNS Drugs 2002;16: 579–593.
Fukuda H, Brailowsky S, Menini C, et al. Anticonvulsant effect of intracortical, chronic infusion of GABA in kindled rats: focal seizures upon withdrawal. Exp Neurol 1987;98: 120–129.
Mimajafi-Zadeh J, Mortazavi M, Fathollahi Y, Alasvand Zarasvand M, Reza Palizvan M. Effect of transient hippocampal inhibition on amygdaloid kindled seizures and amygdaloid kindling rate. Brain Res 2002;954: 220–226.
Stein AG, Eder HG, Blum DE, Drachev A, Fisher RS. An automated drug delivery system for focal epilepsy. Epilepsy Res 2000;39: 103–114.
Finnerty GT, Jefferys JG. Investigation of the neuronal aggregate generating seizures in the rat tetanus toxin model of epilepsy. J Neurophysiol 2002;88: 2919–2927.
Nilsen KE, Cock HR. Focal treatment for refractory epilepsy: hope for the future? Brain Res Brain Res Rev 2004;44(2–3): 141–53.
Schramm J, Clusmann H. The surgery of epilepsy. Neurosurgery 2008;62(suppl 2): 463–481.
Olsen RW, Avoli M. GABA and epileptogenesis. Epilepsia 1997; 38: 399–407.
Löscher W. Basic aspects of epilepsy. Curr Opin Neurol Neurosurg 1993;6: 223–232.
Eder HG, Stein A, Fisher RS. Interictal and ictal activity in the rat cobalt/pilocarpine model of epilepsy decreased by local perfusion of diazepam. Epilepsy Res 1997;29: 17–24.
Eder HG, Jones DB, Fisher RS. Local perfusion of diazepam attenuates interictal and ictal events in the bicuculline model of epilepsy in rats. Epilepsia 1997;38: 516–521.
Morimoto K, Fahnestock M, Racine RJ. Kindling and status epilepticus models of epilepsy: rewiring the brain. Prog Neurobiol 2004;73: 1–60.
Smith DC, Krahl SE, Browning RA, Barea EJ. Rapid cessation of focally induced generalized seizures in rats through microinfusion of lidocaine hydrochloride into the focus. Epilepsia 1993;34: 43–53.
Uemura S, Ienaga K, Higashiura K, Kimura H. Effects of intraamygdaloid injection of taurine and valyltaurine on amygdaloid kindled seizure in rats. Jpn J Psychiatry Neurol 1991;45: 383–385.
Wada Y, Nakamura M, Hasegawa H, Yamaguchi N. Intra-hippocampal injection of 8-hydroxy-2-(di-n-propylamino)tetralin (8-OH-DPAT) inhibits partial and generalized seizures induced by kindling stimulation in cats. Neurosci Lett 1993;159: 179–182.
Attwell PJ, Koumentaki A, Abdul-Ghani AS, Croucher MJ, Bradford HF. Specific group II metabotropic glutamate receptor activation inhibits the development of kindled epilepsy in rats. Brain Res 1998;787: 286–291.
Croucher MJ, Bradford HF, Sunter DC, Watkins JC. Inhibition of the development of electrical kindling of the prepyriform cortex by daily focal injections of excitatory amino acid antagonists. Eur J Pharmacol 1988;152: 29–38.
Pourgholami MH, Rostampour M, Mimajafi-Zadeh J, Palizvan MR. Intra-amygdala infusion of 2-chloroadenosine suppresses amygdala-kindled seizures. Brain Res 1997;775: 37–42.
Schwabe K, Ebert U, Löscher W. The central piriform cortex: anatomical connections and anticonvulsant effect of GABA elevation in the kindling model. Neuroscience 2004;126: 727–741.
Schwabe K, Ebert U, Löscher W. Bilateral microinjections of vigabatrin in the central piriform cortex retard amygdala kindling in rats. Neuroscience 2004;129: 425–429.
Tamargo RJ, Rossell LA, Kossoff EH, Tyler BM, Ewend MG, Aryanpur JJ. The intracerebral administration of phenytoin using controlled-release polymers reduces experimental seizures in rats. Epilepsy Res 2002;48: 145–155.
Weaver DF, Sankar R. Basic principles of medicinal chemistry. In: Engel J, Pedley TA, Aicardi J, et al. eds. Epilepsy: A comprehensive textbook, 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2008: 1447–1455.
Will BE, Toniolo G, Brailowsky S. Unilateral infusion of GABA and saline into the nucleus basalis of rats: 1. Effects on motor function and brain morphology. Behav Brain Res 1988;27: 123–129.
Barcia JA, Castillo A, Gutiérrez R, Gallego JM, Ortiz L, Hernández ME. Continuous intra-amygdalar infusion of GABA in the amygdala kindling model of epilepsy in rat. Epilepsy Res 2004; 58: 19–26.
Gale K. Subcortical structures and pathways involved in convulsive seizure generation. J Clin Neurophysiol 1992;9: 264–277.
Patel S, Millan MH, Meldrum BS. Decrease in excitatory transmission within the lateral habenula and the mediodorsal thalamus protects against limbic seizures in rats. Exp Neurol 1988;101: 63–74.
Cassidy RM, Gale K. Mediodorsal thalamus plays a critical role in the development of limbic motor seizures. J Neurosci 1998;18: 9002–9009.
Applegate CD, Burchfiel JL. Microinjections of GABA agonists into the amygdala complex attenuates kindled seizure expression in the rat. Exp Neurol 1988;102: 185–189.
Kokaia M, Aebischer P, Elmer E, et al. Seizure suppression in kindling epilepsy by intracerebral implants of GABA—but not by noradrenaline-releasing polymer matrices. Exp Brain Res 1994; 100: 385–394.
Maggio R, Gale K. Seizures evoked from area tempestas are subject to control by GABA and glutamate receptors in substantia nigra. Exp Neurol 1989;105: 184–188.
Gernert M, Löscher W. Lack of robust anticonvulsant effects of muscimol microinfusions in the anterior substantia nigra of kindled rats. Eur J Pharmacol 2001;432: 35–41.
Benabid AL, Minotti L, Koudsié A, de Saint Martin A, Hirsch E. Antiepileptic effect of high-frequency stimulation of the subthalamic nucleus (corpus luysi) in a case of medically intractable epilepsy caused by focal dysplasia: a 30-month follow-up: technical case report. Neurosurgery 2002;50: 1385–1391.
Peterson SL. Infusion of NMDA antagonists into the nucleus reticularis pontis oralis inhibits the maximal electroshock seizure response. Brain Res 1995;702: 101–109.
N’Gouemo P, Faingold CL. The periaqueductal grey is a critical site in the neuronal network for audiogenic seizures: modulation by GABA(A), NMDA and opioid receptors. Epilepsy Res 1999;35: 39–46.
Wang PP, Frazier J, Brem H. Local drug delivery to the brain. Adv Drug Deliv Rev 2002;54: 987–1013.
Lindvall O, Kokaia Z, Martinez-Serrano A. Stem cell therapy for human neurodegenerative disorders-how to make it work. Nat Med 2004;10(suppl): 42–50.
Raedt R, Van Dycke A, Vonck K, Boon P. Cell therapy in models for temporal lobe epilepsy. Seizure 2007;16: 565–578.
Roitberg B. Cell transplantation in the central nervous system. Surg Neurol 1999;52: 13–16.
Thompson K, Anantharam V, Behrstock S, et al. Conditionally immortalized cell lines, engineered to produce and release GABA, modulate the development of behavioral seizures. Exp Neurol 2000;161: 481–489.
Thompson KW, Suchomelova LM. Transplants of cells engineered to produce GABA suppress spontaneous seizures. Epilepsia 2004; 45: 4–12.
Gernert M, Thompson KW, Löscher W, Tobin AJ. Genetically engineered GABA-producing cells demonstrate anticonvulsant effects and long-term transgene expression when transplanted into the central piriform cortex of rats. Exp Neurol 2002;176: 183–192.
Thompson KW. Genetically engineered cells with regulatable GABA production can affect afterdischarges and behavioral seizures after transplantation into the dentate gyrus. Neuroscience 2005;133: 1029–1037.
Huber A, Padrun V, Deglon N, et al. Grafts of adenosine-releasing cells suppress seizures in kindling epilepsy. Proc Natl Acad Sci U S A 2001;98: 7611–7616.
Güttinger M, Fedele D, Koch P, et al. Suppression of kindled seizures by paracrine adenosine release from stem cell-derived brain implants. Epilepsia 2005;46: 1162–1169.
Bjorklund A, Lindvall O. Cell replacement therapies for central nervous system disorders. Nat Neurosci 2000;3: 537–544.
Chu K, Kim M, Jung KH, et al. Human neural stem cell transplantation reduces spontaneous recurrent seizures following pilocarpine-induced status epilepticus in adult rats. Brain Res 2004;1023: 213–221.
Miyamoto O, Itano T, Yamamoto Y, et al. Effect of embryonic hippocampal transplantation in amygdaloid kindled rat. Brain Res 1993;603: 143–147.
Matsumoto Y, Sindo A, Kawai N, et al. Transplantation of neural stem cells into epileptic brain. International Congress Series 2003; 1252: 489–492.
Meldrum BS, Chapman AG, Tang E, et al. Cell grafts in epilepsy: therapeutic prospects and problems. Acta Neurologica Scandinavica 2000;175(suppl): 5–52.
Shetty AK, Hattiangady B. Concise review: prospects of stem cell therapy for temporal lobe epilepsy. Stem Cells 2007;25: 2396–2407.
Lavdas AA, Grigoriou M, Pachnis V, Pamavelas JG. The medial ganglionic eminence gives rise to a population of early neurons in the developing cerebral cortex. J Neurosci 1999;19: 7881–7888.
Wichterle H, Garcia-Verdugo JM, Herrera DG, Alvarez-Buylla A. Young neurons from medial ganglionic eminence disperse in adult and embryonic brain. Nat Neurosci 1999;2: 461–466.
Alvarez-Dolado M, Calcagnotto ME, Karkar KM, et al. Cortical inhibition modified by embryonic neural precursors grafted into the postnatal brain. J Neurosci 2006;26: 7380–7389.
Wernig M, Benninger F, Schmandt T, et al. Functional integration of embryonic stem cell-derived neurons in vivo. J Neurosci 2004; 24: 5258–5268.
Rüschenschmidt C, Koch PG, Brustle O, Beck H. Functional properties of ES cell-derived neurons engrafted into the hippocampus of adult normal and chronically epileptic rats. Epilepsia 2005; 46(suppl 5): 174–183.
Fine A, Meldrum BS, Patel S. Modulation of experimentally induced epilepsy by intracerebral grafts of fetal GABAergic neurons. Neuropsychologia 1990;28: 627–634.
Löscher W, Ebert U, Lehmann H, Rosenthal C, Nikkhah G. Seizure suppression in kindling epilepsy by grafts of fetal GABAergic neurons in rat substantia nigra. J Neurosci Res 1998;51: 196–209.
Nolte MW, Löscher W, Herden C, Freed WJ, Gernert M. Benefits and risks of intranigral transplantation of GABA-producing cells subsequent to the establishment of kindling-induced seizures. Neurobiol Dis 2008;31: 342–354.
Schachter SC, Schomer DL, Blume H, et al. Porcine fetal GABA-producing neural cell transplants for human partialonset seizures: safety and feasibility. Epilepsia 1998;39(suppl 6): 67.
Fisher RS, McKhann GM, Stem JM, et al. New therapeutic directions. In: Engel J, Pedley TA, Aicardi J, et al. eds. Epilepsy: a comprehensive textbook. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2008: 1415–1430.
Krauss GL, Miller AD. Scientists clarify basic mechanisms of epilepsy. Highlights of the American Epilepsy Society (AES) 58th Annual Meeting 2005. Available at: http://www.medscape.com/ viewarticle/496939. Accessed November 10, 2008.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Barcia, J.A., Gallego, J.M. Intraventricular and intracerebral delivery of anti-epileptic drugs in the kindling model. Neurotherapeutics 6, 337–343 (2009). https://doi.org/10.1016/j.nurt.2009.01.015
Issue Date:
DOI: https://doi.org/10.1016/j.nurt.2009.01.015