Summary
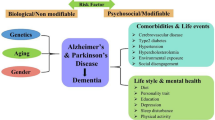
During the last century, the world population has shown a staggering increase in its proportion of elderly members and thus neurodegenerative diseases like Alzheimer’s disease (AD) and Parkinson’s disease (PD), respectively, are becoming an increasing burden on society. Among the diverse, significant challenges facing clinicians, is the improvement of diagnostic measures to detect early and subtle symptoms, a phase in which prevention efforts might be expected to have their greatest impact and provide a measure of disease progression that can be evaluated during the course of drug treatment. At present, clinical diagnosis of AD and PD is based on a constellation of symptoms and manifestations, although the disease originated several years earlier. Given the multiple etiological nature of AD and PD, it is reasonable to assume that the initial causative pathobiological processes may differ between the affected individuals. Therefore, the availability of biological markers or biomarkers will help not only early disease diagnosis, but also delineate the pathological mechanisms more definitively and reliably than the traditional cognitive and neurological phenotypes. In the current article, we review the literature on biochemical, genetic, and neuroimaging biomarkers and discuss their predictive value as indicative for disease vulnerability to detect individuals at risk for PD and AD, and to determine the clinical efficacy of novel, disease-modifying (neuroprotective) strategies.
Article PDF
Similar content being viewed by others
References
Fratiglioni L, Launer LJ, Andersen K, et al. Incidence of dementia and major subtypes in Europe: a collaborative study of population-based cohorts. Neurologic Diseases in the Elderly Research Group. Neurology 2000;54:S10-S15.
Lobo A, Launer LJ, Fratiglioni L, et al. Prevalence of dementia and major subtypes in Europe: A collaborative study of population-based cohorts. Neurologic Diseases in the Elderly Research Group. Neurology 2000;54:S4-S9.
Graham JE, Rockwood K, Beattie BL, et al. Prevalence and severity of cognitive impairment with and without dementia in an elderly population. Lancet 1997;349:1793–1796.
Poewe W, Wenning G. The differential diagnosis of Parkinson’s disease. Eur J Neurol 2002;9 Suppl 3:23–30.
Galasko D, Hansen LA, Katzman R, et al. Clinical-neuropathological correlations in Alzheimer’s disease and related dementias. Arch Neurol 1994;51:888–895.
Hulette C, Nochlin D, McKeel D, et al. Clinical-neuropathologic findings in multi-infarct dementia: a report of six autopsied cases. Neurology 1997;48:668–672.
Lyketsos CG, Szekely CA, Mielke MM, Rosenberg PB, Zandi PP. Developing new treatments for Alzheimer’s disease: the who, what, when, and how of biomarker-guided therapies. Int Psychogeriatr 2008;20: 871–889.
Kawas C, Gray S, Brookmeyer R, Fozard J, Zonderman A. Age-specific incidence rates of Alzheimer’s disease: the Baltimore Longitudinal Study of Aging. Neurology 2000;54:2072–2077.
Youdim MB. The path from anti Parkinson drug selegiline and rasagiline to multifunctional neuroprotective anti Alzheimer drugs ladostigil and m30. Curr Alzheimer Res 2006;3:541–550.
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM. Clinical diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 1984;34:939–944.
Sunderland T, Hampel H, Takeda M, Putnam KT, Cohen RM. Biomarkers in the diagnosis of Alzheimer’s disease: are we ready? J Geriatr Psychiatry Neurol 2006;19:172–179.
Clark CM, Davatzikos C, Borthakur A, et al. Biomarkers for early detection of Alzheimer pathology. Neurosignals 2008;16:11–18.
Halperin I, Korczyn AD. [Mild cognitive impairment (MCI): characteristics, risk factors and prevention]. Harefuah 2006;145:229–234, 243.
Hänninen T, Hallikainen M, Tuomainen S, Vanhanen M, Soininen H. Prevalence of mild cognitive impairment: a population-based study in elderly subjects. Acta Neurol Scand 2002;106:148–154.
Bertram L, Tanzi RE. The current status of Alzheimer’s disease genetics: what do we tell the patients? Pharmacol Res 2004;50:385–396.
Tanzi RE, Bertram L. New frontiers in Alzheimer’s disease genetics. Neuron 2001;32:181–184.
Mulder C, Scheltens P, Visser JJ, van Kamp GJ, Schutgens RB. Genetic and biochemical markers for Alzheimer’s disease: recent developments. Ann Clin Biochem 2000;37 (Pt 5):593–607.
Otto M, Lewczuk P, Wiltfang J. Neurochemical approaches of cerebrospinal fluid diagnostics in neurodegenerative diseases. Methods 2008;44:289–298.
Corder EH, Saunders AM, Strittmatter WJ, et al. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science 1993;261:921–923.
Resnick SM, Goldszal AF, Davatzikos C, et al. One-year age changes in MRI brain volumes in older adults. Cereb Cortex 2000;10:464–472.
Sullivan EV, Pfefferbaum A, Adalsteinsson E, Swan GE, Carmelli D. Differential rates of regional brain change in callosal and ventricular size: a 4-year longitudinal MRI study of elderly men. Cereb Cortex 2002;12: 438–445.
Chetelat G, Baron JC. Early diagnosis of Alzheimer’s disease: contribution of structural neuroimaging. Neuroimage 2003;18:525–541.
Borthakur A, Wheaton AJ, Gougoutas AJ, et al. In vivo measurement of T1rho dispersion in the human brain at 1.5 tesla. J Magn Reson Imaging 2004;19:403–409.
Klunk WE, Panchalingam K, Moossy J, McClure RJ, Pettegrew JW. N-acetyl-L-aspartate and other amino acid metabolites in Alzheimer’s disease brain: a preliminary proton nuclear magnetic resonance study. Neurology 1992;42:1578–1585.
Kwo-On-Yuen PF, Newmark RD, Budinger TF, Kaye JA, Ball MJ, Jagust WJ. Brain N-acetyl-L-aspartic acid in Alzheimer’s disease: a proton magnetic resonance spectroscopy study. Brain Res 1994;667: 167–174.
Jessen F, Block W, Traber F, et al. Proton MR spectroscopy detects a relative decrease of N-acetylaspartate in the medial temporal lobe of patients with AD. Neurology 2000;55:684–688.
Schuff N, Capizzano AA, Du AT, et al. Selective reduction of N-acetylaspartate in medial temporal and parietal lobes in AD. Neurology 2002;58:928–935.
Kantarci K, Jack CR, Jr., Xu YC, et al. Regional metabolic patterns in mild cognitive impairment and Alzheimer’s disease: A 1H MRS study. Neurology 2000;55:210–217.
MacKay S, Ezekiel F, Di Sclafani V, et al. Alzheimer disease and subcortical ischemic vascular dementia: evaluation by combining MR imaging segmentation and H-1 MR spectroscopic imaging. Radiology 1996;198:537–545.
Kantarci K, Jack CR Jr, Xu YC, et al. Regional metabolic patterns in mild cognitive impairment and Alzheimer’s disease: a 1H MRS study. Neurology 2000;55:210–217.
Foster NL, Heidebrink JL, Clark CM, et al. FDG-PET improves accuracy in distinguishing frontotemporal dementia and Alzheimer’s disease. Brain 2007;130:2616–2635.
Silverman DH, Small GW, Chang CY, et al. Positron emission tomography in evaluation of dementia: Regional brain metabolism and long-term outcome. JAMA 2001;286:2120–2127.
Jagust W. Molecular neuroimaging in Alzheimer’s disease. NeuroRx 2004;1:206–212.
Johnson KA, Holman BL, Rosen TJ, Nagel JS, English RJ, Growdon JH. Iofetamine I 123 single photon emission computed tomography is accurate in the diagnosis of Alzheimer’s disease. Arch Intern Med 1990;150:752–756.
Eberling JL, Jagust WJ, Reed BR, Baker MG. Reduced temporal lobe blood flow in Alzheimer’s disease. Neurobiol Aging 1992;13:483–491.
Johnson KA, Jones K, Holman BL, et al. Preclinical prediction of Alzheimer’s disease using SPECT. Neurology 1998;50:1563–1571.
Archer HA, Edison P, Brooks DJ, et al. Amyloid load and cerebral atrophy in Alzheimer’s disease: an 11C-PIB positron emission tomography study. Ann Neurol 2006;60:145–147.
Engler H, Forsberg A, Almkvist O, et al. Two-year follow-up of amyloid deposition in patients with Alzheimer’s disease. Brain 2006;129:2856–2866.
Jagust W, Thisted R, Devous MD, Sr., et al. SPECT perfusion imaging in the diagnosis of Alzheimer’s disease: a clinical-pathologic study. Neurology 2001;56:950–956.
Shoji M, Golde TE, Ghiso J, et al. Production of the Alzheimer amyloid beta protein by normal proteolytic processing. Science 1992;258:126–129.
Blennow K. Cerebrospinal fluid protein biomarkers for Alzheimer’s disease. NeuroRx 2004;1:213–225.
Blennow K, Vanmechelen E, Hampel H. CSF total tau, Abeta42 and phosphorylated tau protein as biomarkers for Alzheimer’s disease. Mol Neurobiol 2001;24:87–97.
Clark CM, Xie S, Chittams J, et al. Cerebrospinal fluid tau and beta-amyloid: how well do these biomarkers reflect autopsy-confirmed dementia diagnoses? Arch Neurol 2003;60:1696–1702.
Arai H, Nakagawa T, Kosaka Y, et al. Elevated cerebrospinal fluid tau protein level as a predictor of dementia in memory-impaired patients. Alzheimer’s Research 1997;3:211–213.
Schonknecht P, Pantel J, Hartmann T, et al. Cerebrospinal fluid tau levels in Alzheimer’s disease are elevated when compared with vascular dementia but do not correlate with measures of cerebral atrophy. Psychiatry Res 2003;120:231–238.
Hampel H, Buerger K, Kohnken R, et al. Tracking of Alzheimer’s disease progression with cerebrospinal fluid tau protein phosphorylated at threonine 231. Ann Neurol 2001;49:545–546.
Galasko D, Clark C, Chang L, Miller B, Green RC, Motter R, Seubert P. Assessment of CSF levels of tau protein in mildly demented patients with Alzheimer’s disease. Neurology 1997;48: 632–635.
Samuels SC, Silverman JM, Marin DB, et al. CSF beta-amyloid, cognition, and APOE genotype in Alzheimer’s disease. Neurology 1999;52:547–551.
Hulstaert F, Blennow K, Ivanoiu A, et al. Improved discrimination of AD patients using beta-amyloid(1–42) and tau levels in CSF. Neurology 1999;52:1555–1562.
Andreasen N, Minthon L, Davidsson P, et al. Evaluation of CSF-tau and CSF-Abeta42 as diagnostic markers for Alzheimer disease in clinical practice. Arch Neurol 2001;58:373–379.
Ringman JM, Younkin SG, Pratico D, et al. Biochemical markers in persons with preclinical familial Alzheimer disease. Neurology 2008;71:85–92.
Obeso JA, Rodriguez-Oroz MC, Rodriguez M, et al. Pathophysiology of the basal ganglia in Parkinson’s disease. Trends Neurosci 2000;23:S8–19.
Mandel S, Amit T, Kalfon L, Youdim MB. Applying transcriptomic and proteomic knowledge to Parkinson’s disease drug discovery. Expert opinion on drug discovery 2007;2:1225–1240.
Vendette M, Gagnon JF, Decary A, et al. REM sleep behavior disorder predicts cognitive impairment in Parkinson’s disease without dementia. Neurology 2007;69:1843–1849.
Postuma RB, Gagnon JF, Vendette M, Charland K, Montplaisir J. REM sleep behaviour disorder in Parkinson’s disease is associated with specific motor features. J Neurol Neurosurg Psychiatry 2008;79:1117–1121.
Burn DJ. Beyond the iron mask: towards better recognition and treatment of depression associated with Parkinson’s disease. Mov Disord 2002;17:445–454.
Remy P, Doder M, Lees A, Turjanski N, Brooks D. Depression in Parkinson’s disease: loss of dopamine and noradrenaline innervation in the limbic system. Brain 2005;128:1314–1322.
McKinnon JH, Demaerschalk BM, Caviness JN, Wellik KE, Adler CH, Wingerchuk DM. Sniffing out Parkinson disease: can olfactory testing differentiate parkinsonian disorders? Neurologist 2007;13:382–385.
Hudry J, Thobois S, Broussolle E, Adeleine P, Royet JP. Evidence for deficiencies in perceptual and semantic olfactory processes in Parkinson’s disease. Chem Senses 2003;28:537–543.
Montgomery EB, Jr., Koller WC, LaMantia TJ, et al. Early detection of probable idiopathic Parkinson’s disease: I. Development of a diagnostic test battery. Mov Disord 2000;15:467–473.
Montgomery EB, Jr., Lyons K, Koller WC. Early detection of probable idiopathic Parkinson’s disease: II. A prospective application of a diagnostic test battery. Mov Disord 2000;15:474–478.
Troster AI. Neuropsychological characteristics of dementia with Lewy bodies and Parkinson’s disease with dementia: differentiation, early detection, and implications for “mild cognitive impairment” and biomarkers. Neuropsychol Rev 2008;18:103–119.
Sommer C, Lauria G. Skin biopsy in the management of peripheral neuropathy. Lancet Neurol 2007;6: 632–642.
Rossi A, Giovenali P, Benvenuti M, Di Iorio W, Calabresi P. Skin biopsy: a new diagnostic tool for autonomic dysfunctions in Parkinson’s disease? Lancet Neurol 2007;6:848–849; author reply 849.
Harrington MG, Merril CR. Two-dimensional electrophoresis and “ultrasensitive” silver staining of cerebrospinal fluid proteins in neurological diseases. Clin Chem 1984;30:1933–1937.
Nagai Y, Ueno S, Saeki Y, Soga F, Hirano M, Yanagihara T. Decrease of the D3 dopamine receptor mRNA expression in lymphocytes from patients with Parkinson’s disease. Neurology 1996;46:791–795.
Barbanti P, Fabbrini G, Ricci A, et al. Increased expression of dopamine receptors on lymphocytes in Parkinson’s disease. Mov Disord 1999;14:764–771.
Caronti B, Antonini G, Calderaro C, et al. Dopamine transporter immunoreactivity in peripheral blood lymphocytes in Parkinson’s disease. J Neural Transm 2001;108:803–807.
Parker WD, Jr., Boyson SJ, Parks JK. Abnormalities of the electron transport chain in idiopathic Parkinson’s disease. Ann Neurol 1989;26:719–723.
Benecke R, Strumper P, Weiss H. Electron transfer complexes I and IV of platelets are abnormal in Parkinson’s disease but normal in Parkinson-plus syndromes. Brain 1993;116 (Pt 6):1451–1463.
Haas RH, Nasirian F, Nakano K, et al. Low platelet mitochondrial complex I and complex II/III activity in early untreated Parkinson’s disease. Ann Neurol 1995;37:714–722.
Migliore L, Petrozzi L, Lucetti C, et al. Oxidative damage and cytogenetic analysis in leukocytes of Parkinson’s disease patients. Neurology 2002;58:1809–1815.
Harrington MG, Fonteh AN, Biringer RG, AF RH, Cowan RP. Prostaglandin D synthase isoforms from cerebrospinal fluid vary with brain pathology. Dis Markers 2006;22:73–81.
Davis JW, Grandinetti A, Waslien CI, Ross GW, White LR, Morens DM. Observations on serum uric acid levels and the risk of idiopathic Parkinson’s disease. Am J Epidemiol 1996;144:480–484.
Weisskopf MG, O’Reilly E, Chen H, Schwarzschild MA, Ascherio A. Plasma urate and risk of Parkinson’s disease. Am J Epidemiol 2007;166:561–567.
Schwarzschild MA, Schwid SR, Marek K, et al. Serum urate as a predictor of clinical and radiographic progression in Parkinson disease. Arch Neurol 2008;65:716–723.
Goldknopf IL. Blood-based proteomics for personalized medicine: examples from neurodegenerative disease. Expert Rev Proteomics 2008;5:1–8.
Sheta EA, Appel SH, Goldknopf IL. 2D gel blood serum biomarkers reveal differential clinical proteomics of the neurodegenerative diseases. Expert Rev Proteomics 2006;3:45–62.
Goldknopf IL, Sheta EA, Bryson J, et al. Complement C3c and related protein biomarkers in amyotrophic lateral sclerosis and Parkinson’s disease. Biochem Biophys Res Commun 2006;342:1034–1039.
Zhang J, Sokal I, Peskind ER, et al. CSF multianalyte profile distinguishes Alzheimer and Parkinson diseases. Am J Clin Pathol 2008;129:526–529.
Gowers WR. A manual of diseases of the nervous system. Philadelphia: P. Blakiston; 1888. pp. 1027–1049.
Leroux P-D. Contribution à l’étude des causes de la paralysie agitante. (in French). Thèse de Paris, imprimeur de la faculté de médecine 1880.
Rosner S, Giladi N, Orr-Urtreger A. Advances in the genetics of Parkinson’s disease. Acta Pharmacol Sin 2008;29:21–34.
Mollenhauer B, Cullen V, Kahn I, et al. Direct quantification of CSF alpha-synuclein by ELISA and first cross-sectional study in patients with neurodegeneration. Exp Neurol 2008;213:315–325.
El-Agnaf OM, Salem SA, Paleologou KE, et al. Detection of oligomeric forms of alpha-synuclein protein in human plasma as a potential biomarker for Parkinson’s disease. Faseb J 2006;20:419–425.
Maita C, Tsuji S, Yabe I, et al. Secretion of DJ-1 into the serum of patients with Parkinson’s disease. Neurosci Lett 2008;431:86–89.
Waragai M, Wei J, Fujita M, et al. Increased level of DJ-1 in the cerebrospinal fluids of sporadic Parkinson’s disease. Biochem Biophys Res Commun 2006;345:967–972.
Waragai M, Nakai M, Wei J, et al. Plasma levels of DJ-1 as a possible marker for progression of sporadic Parkinson’s disease. Neurosci Lett 2007;425:18–22.
Zetterberg H, Ruetschi U, Portelius E, et al. Clinical proteomics in neurodegenerative disorders. Acta Neurol Scand 2008;118:1–11.
Brooks DJ. Technology insight: imaging neurodegeneration in Parkinson’s disease. Nat Clin Pract Neurol 2008;4: 267–277.
Eckert T, Tang C, Eidelberg D. Assessment of the progression of Parkinson’s disease: a metabolic network approach. Lancet Neurol 2007;6:926–932.
Berg D. Biomarkers for the early detection of Parkinson’s and Alzheimer’s disease. Neurodegener Dis 2008;5:133–136.
Berg D, Siefker C, Becker G. Echogenicity of the substantia nigra in Parkinson’s disease and its relation to clinical findings. J Neurol 2001;248:684–689.
Berg D, Roggendorf W, Schroder U, et al. Echogenicity of the substantia nigra: association with increased iron content and marker for susceptibility to nigrostriatal injury. Arch Neurol 2002;59:999–1005.
Zecca L, Youdim MB, Riederer P, Connor JR, Crichton RR. Iron, brain ageing and neurodegenerative disorders. Nat Rev Neurosci 2004;5:863–873.
Koeppen AH. The history of iron in the brain. J Neurol Sci 1995;134(Suppl):1–9.
Braak H, Muller CM, Rub U, et al. Pathology associated with sporadic Parkinson’s disease-where does it end? J Neural Transm Suppl 2006:89–97.
Sullivan PF, Fan C, Perou CM. Evaluating the comparability of gene expression in blood and brain. Am J Med Genet B Neuropsychiatr Genet 2006;141B:261–268.
Burczynski ME, Domer AJ. Transcriptional profiling of peripheral blood cells in clinical pharmacogenomic studies. Pharmacogenomics 2006;7:187–202.
Glatt SJ, Everall IP, Kiemen WS, et al. Comparative gene expression analysis of blood and brain provides concurrent validation of SELENBP1 up-regulation in schizophrenia. Proc Natl Acad Sci U S A 2005;102:15533–15538.
Scherzer CR, Eklund AC, Morse LJ, et al. Molecular markers of early Parkinson’s disease based on gene expression in blood. Proc Natl Acad Sci U S A 2007;104:955–960.
Scherzer CR, Grass JA, Liao Z, et al. GATA transcription factors directly regulate the Parkinson’s disease-linked gene alphasynuclein. Proc Natl Acad Sci U S A 2008;105:10907–10912.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Halperin, I., Morelli, M., Korczyn, A.D. et al. Biomarkers for evaluation of clinical efficacy of multipotential neuroprotective drugs for Alzheimer’s and Parkinson’s diseases. Neurotherapeutics 6, 128–140 (2009). https://doi.org/10.1016/j.nurt.2008.10.033
Issue Date:
DOI: https://doi.org/10.1016/j.nurt.2008.10.033