Abstract
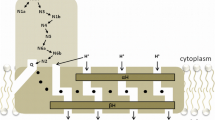
Proton pumping NADH:ubiquinone oxidoreductase (complex I) is the most complicated and least understood enzyme of the respiratory chain. All redox prosthetic groups reside in the peripheral arm of the L-shaped structure. The NADH oxidation domain harbouring the FMN cofactor is connected via a chain of iron–sulfur clusters to the ubiquinone reduction site that is located in a large pocket formed by the PSST- and 49-kDa subunits of complex I. An access path for ubiquinone and different partially overlapping inhibitor binding regions were defined within this pocket by site directed mutagenesis. A combination of biochemical and single particle analysis studies suggests that the ubiquinone reduction site is located well above the membrane domain. Therefore, direct coupling mechanisms seem unlikely and the redox energy must be converted into a conformational change that drives proton pumping across the membrane arm. It is not known which of the subunits and how many are involved in proton translocation. Complex I is a major source of reactive oxygen species (ROS) that are predominantly formed by electron transfer from FMNH2. Mitochondrial complex I can cycle between active and deactive forms that can be distinguished by the reactivity towards divalent cations and thiol-reactive agents. The physiological role of this phenomenon is yet unclear but it could contribute to the regulation of complex I activity in-vivo.
Similar content being viewed by others
References
Abdrakhmanova A, Zickermann K, Kerscher S, Zickermann V, Brandt U (2006) Biochim Biophys Acta 1757:1676–1682
Baranova EA, Holt PJ, Sazanov LA (2007a) J Mol Biol 366:140–154
Baranova EA, Morgan DJ, Sazanov LA (2007b) J Struct Biol 159:238–242
Belogrudov G, Hatefi Y (1994) Biochemistry 33:4571–4576
Brandt U (1997) Biochim Biophys Acta 1318:79–91
Brandt U (1999) BioFactors 9:95–101
Brandt U (2006) Annu Rev Biochem 75:69–92
Brandt U, Kerscher S, Dröse S, Zwicker K, Zickermann V (2003) FEBS Lett 545:9–17
Cardol P, Vanrobaeys F, Devreese B, Van Beeumen J, Matagne RF, Remacle C (2004) Biochim Biophys Acta 1658:212–224
Carroll J, Fearnley IM, Skehel JM, Shannon RJ, Hirst J, Walker JE (2006) J Biol Chem 281:32724–32727
Cernescu A, Maly T, Prisner TF (2008) J Magn Reson 192:78–84
Clason T, Zickermann V, Ruiz T, Brandt U, Radermacher M (2007) J Struct Biol 159:433–442
Darrouzet E, Issartel JP, Lunardi J, Dupuis A (1998) FEBS Lett 431:34–38
Degli Esposti M (1998) Biochim Biophys Acta 1364:222–235
Di Bernardo S, Yano T, Yagi T (2000) Biochemistry 39:9411–9418
Dutton PL, Moser CC, Sled VD, Daldal F, Ohnishi T (1998) Biochim Biophys Acta 1364:245–257
Esterhazy D, King MS, Yakovlev G, Hirst J (2008) Biochemistry 47:3964–3971
Euro L, Belevich G, Verkhovsky MI, Wikström M, Verkhovskaya M (2008) Biochim Biophys Acta 1777:1166–1172
Fendel U, Tocilescu MA, Kerscher S, Brandt U (2008) Biochim Biophys Acta (BBA)—Bioenerg 1777:660–665
Fridovich I (1978) Science 201:875–880
Friedrich T (2001) J Bioenerg Biomembr 33:169–177
Friedrich T, Weiss H (1997) J Theor Biol 187:529–540
Galkin A, Brandt U (2005) J Biol Chem 280:30129–30135
Galkin A, Moncada S (2007) J Biol Chem 282:37748–37753
Galkin A, Dröse S, Brandt U (2006) Biochim Biophys Acta 1757:1575–1581
Galkin A, Meyer B, Wittig I, Karas M, Schägger H, Vinogradov AD, Brandt U (2008) J Biol Chem 283:20907–20913
Gavrikova EV, Vinogradov AD (1999) FEBS Lett 455:36–40
Genova ML, Ventura B, Giuliano G, Bovina C, Formiggini G, Parenti Castelli G, Lenaz G (2001) FEBS Lett 505:364–368
Grgic L, Zwicker K, Kashani-Poor N, Kerscher S, Brandt U (2004) J Biol Chem 279:21193–21199
Guenebaut V, Schlitt A, Weiss H, Leonard K, Friedrich T (1998) J Mol Biol 276:105–112
Hinkle PC, Butow RA, Racker E, Chance B (1967) J Biol Chem 242:5169–5173
Holt PJ, Morgan DJ, Sazanov LA (2003) J Biol Chem 278:43114–43120
Kao MC, Di Bernardo S, Perego M, Nakamaru-Ogiso E, Matsuno-Yagi A, Yagi T (2004a) J Biol Chem 279:32360–32366
Kao MC, Matsuno-Yagi A, Yagi T (2004b) Biochemistry 43:3750–3755
Kao MC, Di Bernardo S, Nakamaru-Ogiso E, Miyoshi H, Matsuno-Yagi A, Yagi T (2005a) Biochemistry 44:3562–3571
Kao MC, Nakamaru-Ogiso E, Matsuno-Yagi A, Yagi T (2005b) Biochemistry 44:9545–9554
Kashani-Poor N, Zwicker K, Kerscher S, Brandt U (2001) J Biol Chem 276:24082–24087
Kerscher S, Kashani-Poor N, Zwicker K, Zickermann V, Brandt U (2001) J Bioenerg Biomembr 33:187–196
Kervinen M, Patsi J, Finel M, Hassinen IE (2004) Biochemistry 43:773–781
Krishnamoorthy G, Hinkle PC (1988) J Biol Chem 263:17566–17575
Kurki S, Zickermann V, Kervinen M, Hassinen IE, Finel M (2000) Biochemistry 39:13496–13502
Kushnareva Y, Murphy AN, Andreyev A (2002) Biochem J 368:545–553
Kussmaul L, Hirst J (2006) Proc Natl Acad Sci U S A 103:7607–7612
Lambert AJ, Brand MD (2004a) J Biol Chem 279:39414–39420
Lambert AJ, Brand MD (2004b) Biochem J 382:511–517
Lambert AJ, Buckingham JA, Brand MD (2008) FEBS Lett 582:1711–1714
Liu Y, Fiskum G, Schubert D (2002) J Neurochem 80:780–787
Maklashina E, Kotlyar AB, Cecchini G (2003) Biochim Biophys Acta 1606:95–103
Maly T, MacMillan F, Zwicker K, Kashani-Poor N, Brandt U, Prisner TF (2004) Biochemistry 43:3969–3978
Mamedova AA, Holt PJ, Carroll J, Sazanov LA (2004) J Biol Chem 279:23830–23836
Mathiesen C, Hägerhall C (2002) Biochim Biophys Acta 1556:121–132
McFarland R, Kirby DM, Fowler KJ, Ohtake A, Ryan MT, Amor DJ, Fletcher JM, Dixon JW, Collins FA, Turnbull DM, Taylor RW, Thorburn DR (2004) Ann Neurol 55:58–64
Mitchell P (1979) Science 206:1148–1159
Morgner N, Zickermann V, Kerscher S, Wittig I, Abdrakhmanova A, Barth HD, Brutschy B, Brandt U (2008) Biochim Biophys Acta (in press)
Moser CC, Farid TA, Chobot SE, Dutton PL (2006) Biochim Biophys Acta—Bioenerg 1757:1096–1109
Nakamaru-Ogiso E, Sakamoto K, Matsuno-Yagi A, Miyoshi H, Yagi T (2003) Biochemistry 42:746–754
Ohnishi T (1998) Biochim Biophys Acta 1364:186–206
Ohnishi T, Nakamaru-Ogiso E (2008) Biochim Biophys Acta 1777:703–710
Ohnishi T, Salerno JC (2005) FEBS Lett 579:4555–4561
Ohnishi T, Johnson JE Jr, Yano T, LoBrutto R, Widger WR (2005) FEBS Lett 579:500–506
Okun JG, Lümmen P, Brandt U (1999) J Biol Chem 274:2625–2630
Prieur I, Lunardi J, Dupuis A (2001) Biochim Biophys Acta 1504:173–178
Radermacher M, Ruiz T, Clason T, Benjamin S, Brandt U, Zickermann V (2006) J Struct Biol 154:269–279
Runswick MJ, Fearnley IM, Skehel JM, Walker JE (1991) FEBS Lett 286:121–124
Sarzi E, Brown MD, Lebon S, Chretien D, Munnich A, Rotig A, Procaccio V (2007) Am J Med Genet A 143:33–41
Sazanov LA, Hinchliffe P (2006) Science 311:1430–1436
Schuler F, Yano T, Di Bernardo S, Yagi T, Yankovskaya V, Singer TP, Casida JE (1999) Proc Natl Acad Sci USA 96:4149–4153
Schulte U, Haupt V, Abelmann A, Fecke W, Brors B, Rasmussen T, Friedrich T, Weiss H (1999) J Mol Biol 292:569–580
Tocilescu MA, Fendel U, Zwicker K, Kerscher S, Brandt U (2007) J Biol Chem 282:29514–29520
Torres-Bacete J, Nakamaru-Ogiso E, Matsuno-Yagi A, Yagi T (2007) J Biol Chem 282:36914–36922
Turrens JF (2003) J Physiol 552:335–344
Verkhovskaya ML, Belevich N, Euro L, Wikström M, Verkhovsky MI (2008) Proc Natl Acad of Sci U S A 105:3763–3767
Vinogradov AD (1998) Biochim Biophys Acta 1364:169–185
Vinogradov AD, Grivennikova VG (2005) Biochemistry (Mosc.) 70:120–127
Vinogradov AD, Sled VD, Burbaev DS, Grivennikova VGX, Moroz IA, Ohnishi T (1995) FEBS Lett 370:83–87
Votyakova TV, Reynolds IJ (2001) J Neurochem 79:266–277
Waletko A, Zwicker K, Abdrakhmanova A, Zickermann V, Brandt U, Kerscher S (2005) J Biol Chem 280:5622–5625
Wikström MKF (1984) FEBS Lett 169:300–304
Yakovlev G, Reda T, Hirst J (2007) Proc Natl Acad Sci U S A 104:12720–12725
Yamaguchi M, Belogrudov G, Hatefi Y (1998) J Biol Chem 273:8094–8098
Yano T, Sklar J, Nakamaru-Ogiso E, Yagi T, Ohnishi T (2003) J Biol Chem 278:15514–15522
Zickermann V, Barquera B, Wikström MKF, Finel M (1998) Biochemistry 37:11792–11796
Zickermann V, Bostina M, Hunte C, Ruiz T, Radermacher M, Brandt U (2003) J Biol Chem 278:29072–29078
Zickermann V, Zwicker K, Tocilescu MA, Kerscher S, Brandt U (2007) Biochim Biophys Acta 1767:393–400
Zwicker K, Galkin A, Dröse S, Grgic L, Kerscher S, Brandt U (2006) J Biol Chem 281:23013–23017
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zickermann, V., Dröse, S., Tocilescu, M.A. et al. Challenges in elucidating structure and mechanism of proton pumping NADH:ubiquinone oxidoreductase (complex I). J Bioenerg Biomembr 40, 475–483 (2008). https://doi.org/10.1007/s10863-008-9171-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-008-9171-9