-
PDF
- Split View
-
Views
-
Cite
Cite
Océane Albert, Bernard Jégou, A critical assessment of the endocrine susceptibility of the human testis to phthalates from fetal life to adulthood, Human Reproduction Update, Volume 20, Issue 2, March/April 2014, Pages 231–249, https://doi.org/10.1093/humupd/dmt050
Close - Share Icon Share
Abstract
A very large proportion of the literature on the endocrine disruptors categorized as anti-androgens deals with phthalates, which are produced in large amounts for use as plastic emollients and additives. In this review, we bring together and analyse work on the effects of phthalates in animals and humans at different stages of their development to assess whether or not their possible anti-androgenic properties represent a significant threat to human health.
The database PubMed was systematically searched for all English language articles until July 2013 in each subject area discussed.
We provide an up-to-date exhaustive, comparative and critical assessment of both in vivo and in vitro studies undertaken to explore the effects of phthalates on the human testis from fetal life to adulthood. These results are compared and discussed in the light of the key data reported in the literature for mice and rats.
The current literature highlights the fact that (i) there is a huge difference between the number of studies performed in animals and in humans, with many fewer for humans; (ii) there are differences in the way rats, mice, primates and humans respond to phthalates, for reasons that need to be further explored; (iii) more work is required to clarify the contradictions, in the few existing human epidemiological studies at all stages of development, which may be partly explained by varying methods of exposure assessment; (iv) in accordance with recent findings in rodents, it cannot be excluded that transgenerational effects of phthalates and/or epigenetic changes exist in humans; (v) a number of methodological limitations need to be solved for the in vitro and xenografting models using human fetal testis to fulfil their ‘missing link’ role between epidemiological studies in humans and rodent models; and (vi) epidemiological and in vitro studies generally converge sufficiently to conclude that phthalate anti-androgenicity is plausible in adult men.
Introduction
The endocrine system is comprised of several organs and glands, and is crucial for the development and correct function of all physiological processes. Over the last two decades, the scientific community has lifted the veil on the existence of chemical factors in the environment, referred to as ‘endocrine disruptors’ (EDs), which may deleteriously affect hormonal balances in the body and reproduction. These revelations have alarmed both the general population and public authorities.
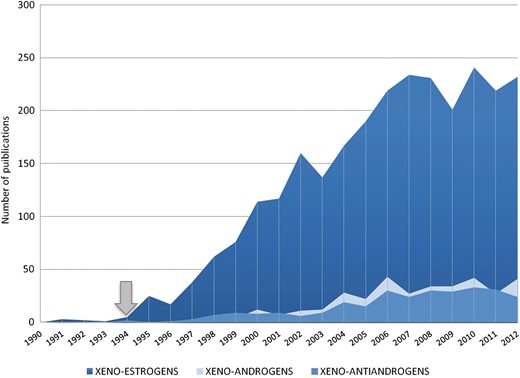
Ever since the emergence of the endocrine disruption issue in the late 1960s, considerable attention has been paid to the chemicals that display estrogenic properties (Fig. 1). It was in 1994, when Kelce et al. (1994) used the term ‘anti-androgenic’ to characterize the activity of the pesticide vinclozolin and its metabolites, that it became apparent that xenoestrogens were not the only EDs displaying sex hormone activities. Since then, numerous molecules and/or their metabolites have been shown to exert agonistic or antagonistic effects on the androgen receptor (AR) and/or inhibit the production of androgens. These effects interfere with major physiological processes, such as masculinization, morphological development of the urogenital system, secondary sex characteristics establishment and possibly with bone metabolism (Oury et al., 2011) (Table I).
Principal known anti-androgenic molecules.
| Use . | Toxicant . | Mode of action . | References . | Species . | Condition . |
|---|---|---|---|---|---|
| Fungicides | Vinclozolin | Inhibits AR transactivation and androgen-dependent gene expression | Kelce et al. (1994, 1997) | Adult rats | In vivo |
| Gray et al. (1999) | Rat fetuses | In utero | |||
| Monosson et al. (1999) | Peripubertal rats | In vivo | |||
| Prochloraz | AR antagonist and inhibits 17,20-lyase and aromatase | Vinggaard et al. (2002) | CHO K1 cells and adult rats | In vitro/in vivo | |
| Procymidone | AR antagonist | Hosokawa et al. (1993) | Adult rats and mice | In vivo | |
| Ostby et al. (1999) | Rat fetuses and newborns | In utero/in vivo | |||
| Pesticides | p,p′-DDT p,p′-DDE | AR antagonist | Kelce et al. (1995) | Developing, prepubertal and adult rats | In utero/in vivo |
| Insecticides | Methoxychlor and HPTE | AR antagonist | Gaido et al. (1997) | Yeast | In vitro |
| Maness et al. (1998) | Human HepG2 cells | In vitro | |||
| Fang et al. (2003) | Recombinant rat AR protein-binding assay | In vitro | |||
| Lindane (γ-HCH isomer) | AR antagonist | Tomczak et al. (1981) | Human occupational exposure | In vivo | |
| Simić et al. (1991) | Adult rats | In vitro | |||
| Aldrin and dieldrin | AR antagonist | Chatterjee et al. (1988) | Adult rats | In vivo | |
| Fang et al. (2003) | Recombinant rat AR protein-binding assay | In vitro | |||
| Herbicides | Linuron | Weak AR antagonist Decreases testosterone pathway enzymatic activity without affecting the enzyme levels | Wolf et al. (1999) | Developing, prepubertal and adult rats | In vivo |
| Lambright et al. (2000) | Adult rats | In vitro | |||
| Plasticizers | Phthalates | Inhibit testosterone synthesis | Li and Kim (2003) | Fetal rat testes culture | In vitro |
| Swan et al. (2005) | Human prenatal exposure | In vivo | |||
| Bisphenol A | Inhibits testosterone production Decreases 3β-HSD and 17β-HSD activities AR antagonist | Gaido et al. (1997) | Yeast | In vitro | |
| Akingbemi et al. (2004) | Prepubertal rat | In vivo | |||
| D'Cruz et al. (2012) | Adult rat | In vivo | |||
| Flame retardants | Polybrominated diphenyl ethers | AR antagonists | Stoker et al. (2005) | Post-natal and adult rats | In vivo/in vitro |
| Tetrabromomethyl-cyclohexanes | AR agonists | Khalaf et al. (2009) | Human HepG2 and LnCap cells | In vitro | |
| Pharmaceutical drugs | Mild analgesics | Inhibit testosterone synthesis and androgen response to hCG | Barkay et al. (1984) | Infertile men | In vivo |
| Berkowitz and Lapinski (1996) | Humans | In utero | |||
| Conte et al. (1999) | Adult men | In vivo | |||
| Kristensen et al. (2012) | Rat fetuses | In vitro | |||
| Albert et al. (2013) | Adult men | In vitro | |||
| Surfactant | Nonylphenol | Weak AR antagonist | Gaido et al. (1997) | Yeast | In vitro |
| Lee et al. (2003) | Yeast | In vitro | |||
| Octylphenol | Reduces expression of mRNA and protein for P450c17 in Leydig cells | Majdic et al. (1996) | Rat fetuses | In utero | |
| Imidazole | Ketoconazole | Inhibits cytochrome P450 testosterone biosynthesis | Pont et al. (1982) | Humans | In vivo |
| Use . | Toxicant . | Mode of action . | References . | Species . | Condition . |
|---|---|---|---|---|---|
| Fungicides | Vinclozolin | Inhibits AR transactivation and androgen-dependent gene expression | Kelce et al. (1994, 1997) | Adult rats | In vivo |
| Gray et al. (1999) | Rat fetuses | In utero | |||
| Monosson et al. (1999) | Peripubertal rats | In vivo | |||
| Prochloraz | AR antagonist and inhibits 17,20-lyase and aromatase | Vinggaard et al. (2002) | CHO K1 cells and adult rats | In vitro/in vivo | |
| Procymidone | AR antagonist | Hosokawa et al. (1993) | Adult rats and mice | In vivo | |
| Ostby et al. (1999) | Rat fetuses and newborns | In utero/in vivo | |||
| Pesticides | p,p′-DDT p,p′-DDE | AR antagonist | Kelce et al. (1995) | Developing, prepubertal and adult rats | In utero/in vivo |
| Insecticides | Methoxychlor and HPTE | AR antagonist | Gaido et al. (1997) | Yeast | In vitro |
| Maness et al. (1998) | Human HepG2 cells | In vitro | |||
| Fang et al. (2003) | Recombinant rat AR protein-binding assay | In vitro | |||
| Lindane (γ-HCH isomer) | AR antagonist | Tomczak et al. (1981) | Human occupational exposure | In vivo | |
| Simić et al. (1991) | Adult rats | In vitro | |||
| Aldrin and dieldrin | AR antagonist | Chatterjee et al. (1988) | Adult rats | In vivo | |
| Fang et al. (2003) | Recombinant rat AR protein-binding assay | In vitro | |||
| Herbicides | Linuron | Weak AR antagonist Decreases testosterone pathway enzymatic activity without affecting the enzyme levels | Wolf et al. (1999) | Developing, prepubertal and adult rats | In vivo |
| Lambright et al. (2000) | Adult rats | In vitro | |||
| Plasticizers | Phthalates | Inhibit testosterone synthesis | Li and Kim (2003) | Fetal rat testes culture | In vitro |
| Swan et al. (2005) | Human prenatal exposure | In vivo | |||
| Bisphenol A | Inhibits testosterone production Decreases 3β-HSD and 17β-HSD activities AR antagonist | Gaido et al. (1997) | Yeast | In vitro | |
| Akingbemi et al. (2004) | Prepubertal rat | In vivo | |||
| D'Cruz et al. (2012) | Adult rat | In vivo | |||
| Flame retardants | Polybrominated diphenyl ethers | AR antagonists | Stoker et al. (2005) | Post-natal and adult rats | In vivo/in vitro |
| Tetrabromomethyl-cyclohexanes | AR agonists | Khalaf et al. (2009) | Human HepG2 and LnCap cells | In vitro | |
| Pharmaceutical drugs | Mild analgesics | Inhibit testosterone synthesis and androgen response to hCG | Barkay et al. (1984) | Infertile men | In vivo |
| Berkowitz and Lapinski (1996) | Humans | In utero | |||
| Conte et al. (1999) | Adult men | In vivo | |||
| Kristensen et al. (2012) | Rat fetuses | In vitro | |||
| Albert et al. (2013) | Adult men | In vitro | |||
| Surfactant | Nonylphenol | Weak AR antagonist | Gaido et al. (1997) | Yeast | In vitro |
| Lee et al. (2003) | Yeast | In vitro | |||
| Octylphenol | Reduces expression of mRNA and protein for P450c17 in Leydig cells | Majdic et al. (1996) | Rat fetuses | In utero | |
| Imidazole | Ketoconazole | Inhibits cytochrome P450 testosterone biosynthesis | Pont et al. (1982) | Humans | In vivo |
The table lists compounds which have been proved to display anti-androgenic properties, either on the AR or on steroidogenesis. The associated references are to the initial studies thought to be the first to have demonstrated the androgenic activities of the compounds of interest.
AR, androgen receptor; 3β-HSD, 3β-hydroxysteroid dehydrogenase; 17β-HSD3, 17β-hydroxysteroid dehydrogenase 3; CHO, chinese hamster ovary; hCG, human chorionic gonadotrophin.
Principal known anti-androgenic molecules.
| Use . | Toxicant . | Mode of action . | References . | Species . | Condition . |
|---|---|---|---|---|---|
| Fungicides | Vinclozolin | Inhibits AR transactivation and androgen-dependent gene expression | Kelce et al. (1994, 1997) | Adult rats | In vivo |
| Gray et al. (1999) | Rat fetuses | In utero | |||
| Monosson et al. (1999) | Peripubertal rats | In vivo | |||
| Prochloraz | AR antagonist and inhibits 17,20-lyase and aromatase | Vinggaard et al. (2002) | CHO K1 cells and adult rats | In vitro/in vivo | |
| Procymidone | AR antagonist | Hosokawa et al. (1993) | Adult rats and mice | In vivo | |
| Ostby et al. (1999) | Rat fetuses and newborns | In utero/in vivo | |||
| Pesticides | p,p′-DDT p,p′-DDE | AR antagonist | Kelce et al. (1995) | Developing, prepubertal and adult rats | In utero/in vivo |
| Insecticides | Methoxychlor and HPTE | AR antagonist | Gaido et al. (1997) | Yeast | In vitro |
| Maness et al. (1998) | Human HepG2 cells | In vitro | |||
| Fang et al. (2003) | Recombinant rat AR protein-binding assay | In vitro | |||
| Lindane (γ-HCH isomer) | AR antagonist | Tomczak et al. (1981) | Human occupational exposure | In vivo | |
| Simić et al. (1991) | Adult rats | In vitro | |||
| Aldrin and dieldrin | AR antagonist | Chatterjee et al. (1988) | Adult rats | In vivo | |
| Fang et al. (2003) | Recombinant rat AR protein-binding assay | In vitro | |||
| Herbicides | Linuron | Weak AR antagonist Decreases testosterone pathway enzymatic activity without affecting the enzyme levels | Wolf et al. (1999) | Developing, prepubertal and adult rats | In vivo |
| Lambright et al. (2000) | Adult rats | In vitro | |||
| Plasticizers | Phthalates | Inhibit testosterone synthesis | Li and Kim (2003) | Fetal rat testes culture | In vitro |
| Swan et al. (2005) | Human prenatal exposure | In vivo | |||
| Bisphenol A | Inhibits testosterone production Decreases 3β-HSD and 17β-HSD activities AR antagonist | Gaido et al. (1997) | Yeast | In vitro | |
| Akingbemi et al. (2004) | Prepubertal rat | In vivo | |||
| D'Cruz et al. (2012) | Adult rat | In vivo | |||
| Flame retardants | Polybrominated diphenyl ethers | AR antagonists | Stoker et al. (2005) | Post-natal and adult rats | In vivo/in vitro |
| Tetrabromomethyl-cyclohexanes | AR agonists | Khalaf et al. (2009) | Human HepG2 and LnCap cells | In vitro | |
| Pharmaceutical drugs | Mild analgesics | Inhibit testosterone synthesis and androgen response to hCG | Barkay et al. (1984) | Infertile men | In vivo |
| Berkowitz and Lapinski (1996) | Humans | In utero | |||
| Conte et al. (1999) | Adult men | In vivo | |||
| Kristensen et al. (2012) | Rat fetuses | In vitro | |||
| Albert et al. (2013) | Adult men | In vitro | |||
| Surfactant | Nonylphenol | Weak AR antagonist | Gaido et al. (1997) | Yeast | In vitro |
| Lee et al. (2003) | Yeast | In vitro | |||
| Octylphenol | Reduces expression of mRNA and protein for P450c17 in Leydig cells | Majdic et al. (1996) | Rat fetuses | In utero | |
| Imidazole | Ketoconazole | Inhibits cytochrome P450 testosterone biosynthesis | Pont et al. (1982) | Humans | In vivo |
| Use . | Toxicant . | Mode of action . | References . | Species . | Condition . |
|---|---|---|---|---|---|
| Fungicides | Vinclozolin | Inhibits AR transactivation and androgen-dependent gene expression | Kelce et al. (1994, 1997) | Adult rats | In vivo |
| Gray et al. (1999) | Rat fetuses | In utero | |||
| Monosson et al. (1999) | Peripubertal rats | In vivo | |||
| Prochloraz | AR antagonist and inhibits 17,20-lyase and aromatase | Vinggaard et al. (2002) | CHO K1 cells and adult rats | In vitro/in vivo | |
| Procymidone | AR antagonist | Hosokawa et al. (1993) | Adult rats and mice | In vivo | |
| Ostby et al. (1999) | Rat fetuses and newborns | In utero/in vivo | |||
| Pesticides | p,p′-DDT p,p′-DDE | AR antagonist | Kelce et al. (1995) | Developing, prepubertal and adult rats | In utero/in vivo |
| Insecticides | Methoxychlor and HPTE | AR antagonist | Gaido et al. (1997) | Yeast | In vitro |
| Maness et al. (1998) | Human HepG2 cells | In vitro | |||
| Fang et al. (2003) | Recombinant rat AR protein-binding assay | In vitro | |||
| Lindane (γ-HCH isomer) | AR antagonist | Tomczak et al. (1981) | Human occupational exposure | In vivo | |
| Simić et al. (1991) | Adult rats | In vitro | |||
| Aldrin and dieldrin | AR antagonist | Chatterjee et al. (1988) | Adult rats | In vivo | |
| Fang et al. (2003) | Recombinant rat AR protein-binding assay | In vitro | |||
| Herbicides | Linuron | Weak AR antagonist Decreases testosterone pathway enzymatic activity without affecting the enzyme levels | Wolf et al. (1999) | Developing, prepubertal and adult rats | In vivo |
| Lambright et al. (2000) | Adult rats | In vitro | |||
| Plasticizers | Phthalates | Inhibit testosterone synthesis | Li and Kim (2003) | Fetal rat testes culture | In vitro |
| Swan et al. (2005) | Human prenatal exposure | In vivo | |||
| Bisphenol A | Inhibits testosterone production Decreases 3β-HSD and 17β-HSD activities AR antagonist | Gaido et al. (1997) | Yeast | In vitro | |
| Akingbemi et al. (2004) | Prepubertal rat | In vivo | |||
| D'Cruz et al. (2012) | Adult rat | In vivo | |||
| Flame retardants | Polybrominated diphenyl ethers | AR antagonists | Stoker et al. (2005) | Post-natal and adult rats | In vivo/in vitro |
| Tetrabromomethyl-cyclohexanes | AR agonists | Khalaf et al. (2009) | Human HepG2 and LnCap cells | In vitro | |
| Pharmaceutical drugs | Mild analgesics | Inhibit testosterone synthesis and androgen response to hCG | Barkay et al. (1984) | Infertile men | In vivo |
| Berkowitz and Lapinski (1996) | Humans | In utero | |||
| Conte et al. (1999) | Adult men | In vivo | |||
| Kristensen et al. (2012) | Rat fetuses | In vitro | |||
| Albert et al. (2013) | Adult men | In vitro | |||
| Surfactant | Nonylphenol | Weak AR antagonist | Gaido et al. (1997) | Yeast | In vitro |
| Lee et al. (2003) | Yeast | In vitro | |||
| Octylphenol | Reduces expression of mRNA and protein for P450c17 in Leydig cells | Majdic et al. (1996) | Rat fetuses | In utero | |
| Imidazole | Ketoconazole | Inhibits cytochrome P450 testosterone biosynthesis | Pont et al. (1982) | Humans | In vivo |
The table lists compounds which have been proved to display anti-androgenic properties, either on the AR or on steroidogenesis. The associated references are to the initial studies thought to be the first to have demonstrated the androgenic activities of the compounds of interest.
AR, androgen receptor; 3β-HSD, 3β-hydroxysteroid dehydrogenase; 17β-HSD3, 17β-hydroxysteroid dehydrogenase 3; CHO, chinese hamster ovary; hCG, human chorionic gonadotrophin.
PubMed survey reporting the number of published studies addressing the key words xenoestrogens, xenoandrogens and xeno-anti-androgens in the PubMed database. The database was searched with the terms ‘xeno(-)’, ‘disruptor(s)’, ‘contaminant(s)’ and ‘pollutant(s)’ associated with ‘(o)estrogenic’, ‘androgenic’ or ‘anti(-)androgenic’, respectively. The number of studies was counted for each year between 1990 and 2012. The arrow indicates the first appearance of the term ‘anti-androgenic’ (Kelce et al., 1994).
A very large proportion of the literature on xeno-anti-androgens deals with phthalates. These compounds are produced in very large amounts for use as plastic emollients and additives in industrial applications and, therefore, in a number of consumer products (Sears and Darby, 1982). Di-(2-ethylhexyl) phthalate (DEHP) is the most commonly used chemical additive to provide flexibility to polyvinylchloride, and is, therefore, ubiquitous, for example in toys, packaging films and sheets, medical tubing and blood storage bags, insecticides, lubricants or care products (Kavlock et al., 2006; Schettler, 2006). Di-n-butyl phthalate (DBP) is more widely used in latex products, cellulose plastics and solvents for some dyes, and di-ethyl phthalate is a constituent of >70% of cosmetics and personal care products tested (Crinnion, 2010). Phthalates are not covalently bound to the plastics to which they are added, and can, therefore, leach out over time (Thomas and Thomas, 1984). Numerous studies have now reported extensive and widespread environmental and human exposure as a consequence (Fay et al., 1999; Loff et al., 2000; Koch et al., 2003; McKee et al., 2004; Silva et al., 2004; Hauser and Calafat, 2005; Wittassek et al., 2011; Kessler et al., 2012). The major route of exposure is ingestion/diet (Wormuth et al., 2006; Frederiksen et al., 2007; Fromme et al., 2007; Koch et al., 2013), in particular for high molecular weight phthalates (Clark et al., 2003), with exposure also occurring through the placenta or skin (Janjua et al., 2008) or through inhalation (Heudorf et al., 2007). However, all possible routes of contamination are involved (Duty et al., 2005; Wormuth et al., 2006; Joensen et al., 2012). Of particular concern is the high exposure of children and neonates in medical intensive care (Calafat et al., 2004; Green et al., 2005; Weuve et al., 2006). In the rat gut, phthalate diesters are biotransformed to monoester forms, then oxidized, glucuronidated and eliminated in the urine (Williams and Blanchfield, 1975), and in humans, it is likely that the stomach acid lipases (Carrière et al., 1993) hydrolyse DEHP into mono-(2-ethylhexyl) phthalate (MEHP) (Kessler et al., 2012).
In this review, we bring together and analyse work on the effects of phthalates in animals and humans at different stages of their development. Our aim is to assess whether or not phthalates represent a significant threat to human reproductive health notably via their possible anti-androgenic properties. The phthalates which will be referred to throughout this study are represented in Table II.
Molecular structures of the phthalates falling within the scope of this review.
Adapted from Koch and Calafat (2009). All the phthalates and the metabolites cited in the review are in bold. Only the secondary metabolites related to this review are cited.
Molecular structures of the phthalates falling within the scope of this review.
Adapted from Koch and Calafat (2009). All the phthalates and the metabolites cited in the review are in bold. Only the secondary metabolites related to this review are cited.
Methods
Literature search strategy and inclusion criteria
We performed an extensive search of the PubMed database for English language articles published up to July 2013, including clinical, epidemiological, experimental, in vivo and in vitro studies and xenografting models, using the following keywords: ‘phthalate’ AND ‘testis’ AND (‘rat’ OR ‘mouse’ OR ‘mice’ OR ‘marmoset’ OR ‘human’). After detailed screening of titles, abstracts and full texts, we selected the studies referring to each category discussed, i.e. rodents and human fetal, neonatal, prepubertal, pubertal and adult testis and each approach discussed, i.e. in vivo, in vitro, xenografting or epidemiology. In total, 200 articles were screened and reviewed.
For Fig. 1, the PubMed database was searched with the terms ‘xeno(-)’ OR ‘disruptor(s)’ OR ‘contaminant(s)’ OR ‘pollutant(s)’ associated with ‘(o)estrogenic’ OR ‘androgenic’ OR ‘anti(-) androgenic’, respectively, between 1990 and 2013. For Table III, the PubMed database was searched using the terms ‘phthalate’ AND ‘testis’ AND (‘mouse’ OR ‘rat’ OR ‘marmoset’ OR ‘human’) and papers were individually checked for relevance.
PubMed survey reporting the number of studies in the PubMed database addressing the effects of phthalates on the testis in the mouse, rat, marmoset and humans at different ages of exposure
The numbers of articles found in PubMed using the terms ‘phthalates’ and ‘testis’ associated with either ‘mouse’, ‘rat’, ‘marmoset’ or ‘human’ and individually checked for relevance are reported.
Note that a few studies have addressed the issue of the toxicity of the phthalates on the testis of other species such as the hamster and guinea pig (Gray et al., 1982), the ferret (Lake et al., 1976), the rabbit (Higuchi et al., 2003), the frog Xenopus laevis (Lee and Veeramachaneni, 2005) and the fish (Mankidy et al., 2013).
PubMed survey reporting the number of studies in the PubMed database addressing the effects of phthalates on the testis in the mouse, rat, marmoset and humans at different ages of exposure
The numbers of articles found in PubMed using the terms ‘phthalates’ and ‘testis’ associated with either ‘mouse’, ‘rat’, ‘marmoset’ or ‘human’ and individually checked for relevance are reported.
Note that a few studies have addressed the issue of the toxicity of the phthalates on the testis of other species such as the hamster and guinea pig (Gray et al., 1982), the ferret (Lake et al., 1976), the rabbit (Higuchi et al., 2003), the frog Xenopus laevis (Lee and Veeramachaneni, 2005) and the fish (Mankidy et al., 2013).
Background in rodents
Before discussing the issue of the effects of phthalates on human testicular function, it is important to briefly summarize current knowledge about their effects in mouse and rat models, as the latter constitutes the source of the overwhelming majority of the relevant data (Table III). More information on the reprotoxicity of phthalates can be found elsewhere (Foster et al., 2001; Howdeshell et al., 2008; Wu et al., 2010; Knez, 2013; Mankidy et al., 2013; Martinez-Arguelles et al., 2013).
In addition to the findings outlined below, it is important to note that recent data suggest that potential transgenerational disorders are also induced by phthalate exposure, such as epigenetic inheritance of reproductive disease or sperm epimutations (Manikkam et al., 2013), reduction of anogenital distance (AGD), reproductive organ weights or sperm count in F1 males (Gray et al., 2009) in the rat, or disruption of testicular germ cell organization and spermatogonial stem cell function in the mouse (Doyle et al., 2013).
The rat model
In vivo exposure of the rat
Most studies on the effects of phthalates on the testis have involved in vivo exposure in rodent models (73%), and more than one-third have involved in utero exposure in rats. Most of the in utero studies in the rat have concluded that exposure to doses of phthalates ranging from 250 to 750 mg/kg/day would decrease testicular testosterone synthesis (Parks et al., 2000) and induce the development of abnormalities of the male reproductive tract, such as hypospadias (Mylchreest et al., 1998; Wolf et al., 1999; Parks et al., 2000), testicular maldescent (Shono et al., 2000; Imajima et al., 2001; McKinnell et al., 2005), lesions of the gubernaculum (Wilson et al., 2004, 2007), malformations of the epididymis, vas deferens, seminal vesicles or prostate (Mylchreest et al., 1998), retained nipples (Foster, 2006) or decreased AGD (Mylchreest et al., 1998; Parks et al., 2000; van den Driesche et al., 2012a); these effects were found to depend on the compound considered, its dose, the rat strain used and the male genital tract development window at which the fetuses are exposed (Ema et al., 2000; Gray et al., 2000; Wilson et al., 2004, 2007; Carruthers and Foster, 2005; Welsh et al., 2008). They were unlikely to result from antagonistic effects on the androgen receptor (Mylchreest et al., 1998; Parks et al., 2000). Instead, both DEHP and DBP were found to act by inhibiting fetal rat testosterone production (Parks et al., 2000; Mylchreest et al., 2002). In DBP-exposed male rats, numerous multi-nucleated gonocytes (MNGs) were evidenced (Parks et al., 2000; Mylchreest et al., 2002; Fisher et al., 2003; Ferrara et al., 2006), which did not survive beyond post-natal day (PND) 3 (Fisher et al., 2003), and complete recovery of normal germ cell function/numbers was observed by adulthood, associated with a possible slight hypofertility (Ferrara et al., 2006). Numerous studies exploring the mechanisms of action of phthalates on the fetal rat testis also report alterations of the expression of a number of genes involved in testicular function, including Insl3 (insulin-like factor 3) encoding for a fetal hormone which is known in particular for its involvement in the first phase of testicular descent (Wilson et al., 2004; McKinnell et al., 2005; Lague and Tremblay, 2008; Bay et al., 2011; Ivell and Anand-Ivell, 2011). Repression of the transcription of genes that are involved in cholesterol transport (Scarb1, Star) (Shultz et al., 2001; Boberg et al., 2008) or steroid biosynthesis (Cyp11a1, Hsd3b1, Cyp17a1) (Liu et al., 2005; van den Driesche et al., 2012b) of the PPARα gene (Boberg et al., 2008) or of Sertoli cells–gonocytes interactions (c-kit; Shultz et al., 2001) by phthalates was demonstrated in the fetal rat testis. Of note is that if not all phthalate congeners are toxic to the testis (Hannas et al., 2011, 2012), the testis-toxic ones are all likely to act via similar mechanisms involving blockage of cholesterol transport and synthesis as well as fetal testosterone production (Gray et al., 2000; Liu et al., 2005).
Most studies have focused on the consequences of exposure to phthalates in the fetal, perinatal and juvenile rat. A perinatal exposure of rats gavaged from gestational day (GD) 7 to PND 16 with a relatively low-dose of DEHP (10 mg/kg/day) caused adverse anti-androgenic effects on the male rat characterized by a decreased AGD, an increased incidence of nipple retention, a reduction of prostate weight and the occurrence of mild external genitalia dysgenesis. The highest doses (300–900 mg/kg/day) led to alterations in testis morphology (Christiansen et al., 2010). Exposure of prepubertal rats to up to 0.4–2.2 g/kg/day phthalates was found to alter the seminiferous epithelium (Creasy et al., 1983; Dalgaard et al., 2001) and delay spermatogenesis (Alam et al., 2010). However, evidence of the sensitivity of the adult rat testis to these compounds is weaker. Indeed, although most relevant studies concluded that exposure to 2.2–20 g/kg/day phthalates induced morphological alterations of the testis, mostly decreased testis weight (Agarwal et al., 1986), Sertoli cell damage (Creasy et al., 1987) and germ cell apoptosis (Moffit et al., 2007), they report only non-significant trends for decreased testosterone production (Agarwal et al., 1986). One study showed no effect of exposure to phthalates on the mature rat testis, although the prepubertal testis was affected, which led the authors to conclude that the effects of phthalates were age-specific in this species (Kondo et al., 2006). Nevertheless, in utero exposure to 234–1250 mg/kg/day phthalates has been shown to induce persistent alterations in the prepubertal and mature rat, including decreased testosterone production (Culty et al., 2008), penile length, testis, ventral prostate or seminal vesicle weight, or AGD (Macleod et al., 2010).
Rat fetal testis organotypic cultures
While there is no doubt that the models based on in utero exposure are essential for investigating the consequences of exposure to phthalates on the fetal testis, these approaches have intrinsic limitations. In particular, they do not allow detailed analysis of the origin of the observed hormonal disturbances, which could be due either to an alteration of the endocrine system or the health status of the mother and/or of the fetus, or to direct effects on the fetal testis itself. Consequently, as a complement to exposure during pregnancy, several groups have developed culture system models using microdissected fetal testes at different ages. These ex vivo models have the potential to allow (i) the direct investigation of the effects of molecules of interest; (ii) the precise study of their kinetics of action; (iii) the rigorous control of the doses and durations of exposure; (iv) the determination of the nature of the active compounds and their concentrations; (v) attempts to identify the main and primary cellular targets as time-course studies are feasible; and (vi) a decrease in the cost and the labour involved.
Fetal (GD 13 and 18) and neonatal (PND 3) rat testis cultures were first used to investigate the direct effects of MEHP on testicular cells (Li and Kim, 2003). Exposure to 5 × 10−5 to 2 × 10−4 M MEHP was not found to affect the early steps (GD13) of fetal testis formation: no alterations were found for testicular chord formation, anti-Müllerian hormone (AMH, the hormone responsible for the regression of the Müllerian ducts) immunostaining within fetal Sertoli cells or the number of gonocytes. Chauvigné et al. (2009) first established that fetal rat testes in culture were unable to metabolize DEHP between GD14.5 and GD17.5; the same authors developed an organotypic culture system previously applied to study the effects of estradiol and diethylstilbestrol (Lassurguere et al., 2003) and found that 10−5 M DEHP increased testosterone production by GD14.5 fetal testes after 3 days of culture (we will refer to this as a pro-androgenic effect), whereas its primary metabolite MEHP as well as the 5-hydroxy-metabolite (5-OH-MEHP) of MEHP itself inhibited this production in a time- and dose-dependent manner (anti-androgenic effect). Using this fetal gonad assay (FEGA), the same authors also demonstrated that MEHP caused a decrease in the number of gonocytes, consecutive to both a decrease in their proliferation and an increase in their apoptotic rate, and disrupted AMH production by fetal Sertoli cells (Chauvigné et al., 2009). Time-course experiments identified Leydig cells as the first cell type to be chronologically affected by MEHP and revealed that there was a subsequent decrease in the germ cell pool, concomitant to a decrease in Sertoli cell function. By tracking the distribution of 14C-DEHP and 14C-MEHP in their culture systems, the authors also found that when added at concentrations of 10–5 M, these two phthalates were present in the testicular fetal explants at only 0.3–0.4% of the initial concentration, corresponding to ∼2.5 μg/l. Thus, the active concentrations of MEHP were within an order of magnitude of the concentrations found in plasma from pregnant women (1.150 μg DEHP/l and 680 μg MEHP/l) (Latini et al., 2003), and in neonate cord blood (2.050 μg DEHP/l and 520 μg MEHP/l) (Latini et al., 2003). Further experiments demonstrated that MEHP directly alters expression of the Cyp17 gene as well as the activity of the CYP17 lyase itself (which is responsible for the conversion of 17α hydroxy-progesterone into androstenedione) and also affects the expression of other Leydig cell genes, including those encoding INSL3 and inhibin A (Chauvigné et al., 2011).
Li and Kim (2003), studying later developmental stages, namely GD18 and PND3, showed that MEHP decreased the levels of the Sertoli cell products, AMH and GATA-4 (immunohistochemistry), and impaired Sertoli cell proliferation. MEHP exposure also induced dose-dependent apoptosis of gonocytes in the neonatal testis at GD18 and PND3. Conversely, Hallmark et al. (2007) showed that 48 h of exposure to 10−3 M mono-n-butyl phthalate (MBP) did not affect basal or 22-OH cholesterol-stimulated testosterone production by GD19.5 fetal rat testis explants in vitro, although it significantly reduced human chorionic gonadotrophin (hCG)-stimulated steroidogenesis.
These various age-related differences in the effects of phthalates on the fetal rat testes in vitro highlight the fundamental importance of the developmental stage at which the testes are exposed; note that in vivo studies demonstrate that the period from GD15.5 to GD19.5 is critical in the rat (Ema et al., 2000; Carruthers and Foster, 2005; Welsh et al., 2008), which corresponds well with the in vitro observations described above.
Thus, applying some ex vivo rat fetal testis cultures clearly met with the objectives listed at the beginning of this paragraph, as a number of key features observed after in utero treatments in the rat were confirmed and extended. However, if it is true that these in vitro tests ‘do not exactly recapitulate the testis phenotype observed after in-vivo exposure’ (Johnson et al., 2012), this may well be at least partly explained by the fact that the phthalate-induced effects seen in vitro result from a direct exposure, excluding any indirect effects which may secondarily result from the effects of the compounds on the pregnant dam and/or on the fetus itself as a whole.
The mouse model
In vivo exposure of the mouse
In the mouse (21% of all studies; Table III), in utero exposure to 500 mg/kg/day MEHP, 1.5 g/kg/day DBP or 1 g/kg/day MBP from GD14 to GD16 resulted in fetal testicular testosterone levels being higher than in controls (pro-androgenic effect), and 1 g/kg/day MEHP/MBP from GD15 to GD17 had no significant effect in C57Bl/6J and C3H/HeJ strains (Gaido et al., 2007). Exposure to 500 mg/kg/day DBP from GD11.5 to GD17.5 of C57Bl/6J mice did not alter fetal testicular testosterone levels (hereafter referred to as a norm-androgenic effect) (van den Driesche et al., 2012b). Accordingly, neonatal C57Bl/6Crl and CD-1 mice AGD was unaffected by in utero exposure to 500 mg/kg DBP from GD14 to GD18 (Heger et al., 2012). However, fetal exposure of CD-1 mice to 100 or 500 mg/kg DBP from GD12 to GD20 resulted in the formation of MNGs, and caused multiple gene expression alterations in the mouse testis, including that of genes involved in lipid metabolism and cholesterol synthesis (Johnson et al., 2011). Moreover, after administration of DBP to prepubertal mice (C57Bl/6J), delayed spermatogenesis and abnormal Sertoli cell maturation were observed at doses ranging from 10 to 500 mg/kg/day, and decreased serum testosterone levels were observed at 500 mg/kg/day (Moody et al., 2013). After 10 days of prepubertal exposure, the AGD relative to trunk length was decreased at all doses tested. Long-term effects were also evidenced, as a reduced AGD and indications of disrupted spermatogenesis were seen in the adult mice exposed prepubertally to treatments from 1 mg/kg/day. Thus, differences could be observed in the androgenic status of the mouse testis whether the treatments occurred before (norm- and pro-androgenic effects) or after birth (anti-androgenic effects).
Mouse fetal testis organotypic cultures
Work with organ cultures of fetal C57Bl/6 mouse testis shows that exposure to MEHP reduces fetal germ cell number independent of their steroidogenic status (Lehraiki et al., 2009). Indeed, at both GD13.5 (full testis) and GD18.5 (testis explants), a high concentration of MEHP (2 × 10−4 M) significantly increased basal testosterone production and, at GD18.5 only, decreased the androgenic response of fetal explants to luteinizing hormone (LH) and to dibutyryl cyclic adenosine monophosphate. This pro/norm-androgenic action of MEHP in the mouse corroborates the in utero findings described above. This reveals the importance of species-specific differences in the testicular response to phthalate exposure (Johnson et al., 2012): several of these compounds exert anti-androgenic activities in the rat, whereas they are norm-androgenic, or even pro-androgenic, in the mouse at the same fetal age.
Phthalates and the androgenic status of the human fetal testis
To date, studies in humans represent <10% of the total research effort devoted to phthalates and the testis.
Phthalate exposure during pregnancy
Pregnant women (in blood plasma) as well as the human fetus (in neonate cord blood) have been found to be exposed to both DEHP and MEHP (Latini et al., 2003). This has to be considered in the light of the observations showing that after gavage of the pregnant rat with phthalate diesters, phthalate monoesters, which are suspected to be the most reprotoxic metabolites (Ema and Miyawaki, 2001; Chauvigné et al., 2009), rapidly cross the placenta, thus raising concern about exposure of the human fetus during pregnancy. Swan et al. (2005) were the first to look at the relationship between prenatal exposure to phthalates and the AGD in baby boys, which is considered to be a strong sensitive marker of an anti-androgenic exposure. Their work revealed that the age-adjusted anogenital index (AGI, which corresponds to AGD individually adjusted on weight at examination) was shorter than normal in 0–40-month-old boys whose mothers had elevated prenatal exposure to phthalates, as assessed from four urinary phthalate metabolites, namely monoethyl phthalate (MEP), MBP, monobenzyl phthalate (MBzP) and mono-iso-butyl phthalate. The results suggested that phthalic acid esters may ‘undervirilize’ humans as they do in rats. The same group also found significant associations between reduced AGD and exposure both to each and the sum of three DEHP metabolites (MEHP, mono-(2-ethyl-5-oxohexyl) phthalate (MEOHP) and mono-(2-ethyl-5-hydroxyhexyl) phthalate (MEHHP)). A direct inverse relationship between high concentrations of DEHP urinary metabolites (mostly MEHP) and penile width was described, and MEHP metabolite concentrations were significantly and inversely related to testicular descent (Swan, 2008). In contrast, Huang et al. (2009) found no association between either AGD or AGI in male newborns and prenatal phthalate exposure in utero, as assessed from the concentrations of phthalate monoesters (MBP, MEHP, MEP, MBzP and monomethyl phthalate (MMP)) in the mothers' amniotic fluid and urine. According to these authors, measurements of AGD at birth may reflect the effects of phthalate exposure in utero, whereas those at 3 months of age or older (Swan et al., 2005) may be affected by both post-natal exposure to phthalates and increasing body weight and length, explaining the divergence between their findings and those reported by Swan et al. (2005). However, in a more recent study, maternal urinary MEHP levels were found to be significantly inversely correlated with the AGI (AGD individually adjusted on birthweight or length) in newborn boys, which was not the case for MEP and MnBP (Suzuki et al., 2012). The authors explain those minor discrepancies with Swan's studies (2008) by statistical MEP-AGI and MnBP-AGI correlation artefacts due to significant correlations between the concentrations of MEP or MnBP and MEHP in urine samples, possibly indicating multiple sources or routes of exposure. However, the authors give no explanation of the inconsistencies between their conclusions and that of Huang et al. (2009) regarding MEHP-AGI correlation. Lastly, maternal exposure to phthalates in the workplace was found to be associated with the risk of hypospadias, thus reinforcing the presumption of anti-androgenic effects of these compounds during pregnancy (Ormond et al., 2009).
In any case, there is a need for further studies to understand the somewhat contradictory observations seen in epidemiology and the underlying mechanisms of the effects of phthalates during pregnancy.
Direct exposure of the human fetal testis to phthalates
Human fetal testis organotypic cultures
Because they are somewhat contradictory, epidemiological studies of the possible effects of prenatal exposure to phthalates are yet insufficient for definite conclusions about the anti-androgenicity of these compounds in humans, in particular during fetal life. This has highlighted the need for experimental models that would allow complementary investigations in humans and bridge the gap between the information generated in animal models and that from epidemiological studies in humans.
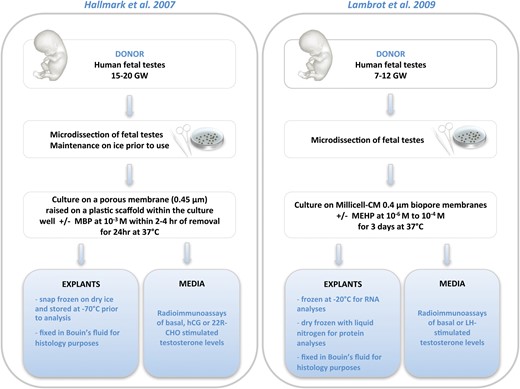
The first model system, based on the organotypic culture of human fetal testes recovered from legal abortion products, was set up and used to study the effect of an ED, the 4-octylphenol, in 2001 (Bendsen et al., 2001). Six years later, Fowler et al. (2007) demonstrated that dieldrin (a pesticide with anti-androgenic properties; Table I) was able to reduce LH-induced testosterone secretion by the human fetal testis in culture. The same year, Hallmark et al. (2007) used human fetal testes in vitro (Fig. 2) to investigate the direct effects of phthalates and showed that basal and hCG- or 22-OH cholesterol-stimulated testosterone production by 15–20 gestational weeks (GW) fetal testis in culture remained unaffected by up to 48 h of exposure to 10−3 M MBP. This prompted the authors to question the limitations and viability of the culture-model system: ‘One of the main conclusions of the present studies is that use of fetal testis explants to study the mechanisms of action of DBP/MBP appears limited and is perhaps nonviable’ (Hallmark et al., 2007).
Comparative representation of human fetal testis explant culture models. The protocols are summarized from the original publications by Hallmark et al. (2007) and Lambrot et al. (2009). MBP, mono-n-butyl phthalate; MEHP, mono-(2-ethylhexyl) phthalate; LH, luteinizing hormone; 22R-CHO, 22R-hydroxycholesterol; GW, gestational week.
Accordingly, 10−4, 10−5 and 10−6 M of MEHP had no significant effect on basal testosterone production by 7–12 GW human fetal testes cultured up to 3 days (Fig. 2); 10−4 M MEHP had no effect on LH-stimulated testosterone production either. However, both 10−5 and 10−4 M MEHP reduced the number of germ cells by increasing apoptosis, without modification of their proliferation rate (Lambrot et al., 2009). Whether this was a consequence of a phthalate-induced alteration of the Sertoli cell function cannot be deduced from the inconclusive PCR assessment of AMH presented in this study. Interpretation of this apoptosis-only phenotype in the apparent absence of a disruption of testosterone levels (or of any other hormone level) should integrate the fact that a direct chemical-induced toxicity on gonocytes cannot be excluded under experimental conditions that inevitably disturb their physiological protective microenvironment. Recent experiments have also shown that exposure to a high concentration of MEHP (10−4 M) for 72 h of 7–12 GW human testes up-regulated the somatic expression of two members of the nuclear receptor superfamily, namely LXRA and SREBP (1c and 2), thereby affecting downstream genes involved in the gonadal lipid and cholesterol synthesis (Muczynski et al., 2012b).
The use of short-term cultures (1–3 days maximum, Fig. 2) in Hallmark et al. (2007) and Lambrot et al. (2009) raises the potential criticism that longer periods of exposure would better mimic the exposure of the human fetus in utero.
The apparent resistance of the human fetal testis in culture to the putative anti-androgenicity of phthalates contrasts with what was previously observed in the rat both in utero and in vitro, where phthalates generally displayed anti-androgenic capabilities. It is also discordant with various findings for the fetal mouse testis, where in vitro phthalates can display pro/norm-androgenic effects. These discordances between species have further fed the debate on the importance of species-specificity in the response to phthalates (Koop et al., 1999; Gray et al., 2000; Johnson et al., 2012), and illustrate the complexity of human risk assessment.
Human fetal testis xenografting
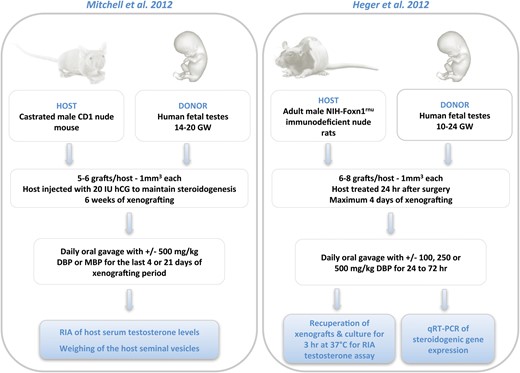
Only two teams have used and promoted innovative xenografting techniques to investigate the effects of phthalates on the fetal human testis. They used different protocols, both of which are described in Fig. 3.
Comparative representation of human fetal testis xenograft models. The protocols are summarized from the original publications by Mitchell et al. (2012) and Heger et al. (2012). DBP, di-n-butyl phthalate; MBP, monobutyl phthalate; GW, gestational week.
In the elegant procedure developed by Mitchell et al. (2010; 2012), castrated male nude mice were xenografted with human 14–20 GW fetal testes fragments, and 6 weeks later treated with 500 mg/kg/day DBP for 4 or 21 days. Under these conditions which allow the testosterone produced by the human fetal grafts to be assayed in the circulating blood of the mouse recipient, the authors did not find any significant ‘difference in serum testosterone levels or seminal vesicle weight between vehicle and DBP-treated hosts, irrespective of whether treatment was for 4 or 21 days’. Nevertheless, there was a >30% reduction of testosterone production which, though non-significant (P = 0.241, n = 3–4), was induced by the exposure of the mice xenografted with human fetal testis tissue to 500 mg/kg/day MBP for 21 days. Experiments performed in parallel with the a priori phthalate-sensitive 17.5 GD fetal rat testis grafted in the same strain of mice showed that the testosterone production by fetal rat testis xenografts exposed to DBP for 4 days was only decreased by 20%, which was not significant (P = 0.062, n = 6). However, in contrast to the experiment with human tissue, there was a significant reduction in seminal vesicle weight and a significant decrease in the mRNAs for Cyp11A1 and Star in the rat testis, as assessed by qRT–PCR. These results lead Mitchell et al. (2012) to conclude that it is ‘unlikely’ that xenografted human fetal testis is sensitive to this phthalate, asserting that ‘DBP/MBP exposure of the normally developing/functioning human fetal testis is probably without any effect on steroidogenesis’. This is a sensibly cautious conclusion in view of the relatively low sensitivity of the rat xenografting model system used as a ‘positive’ control for anti-androgenicity, and the relatively small number of repeats performed with human testes for MBP. It would also be interesting to know to what extent the adrenals may adjust to the suppression of testicular testosterone production after castration of the recipient mice, as (i) any adrenal supplementary testosterone contribution would interfere with the levels of the testosterone measured in the xenografted system, and (ii) phthalates have also been shown to disrupt the adrenal gland steroidogenesis and physiology (Jiang et al., 2007; Supornsilchai et al., 2007; Martinez-Arguelles et al., 2011).
Concomitantly to Mitchell et al. (2012), Heger et al. (2012) investigated the effects of phthalates using another xenograft model based on the use of 10–24 GW (average: 18.6 GW) human fetal testis explants xenografted into non-castrated adult male immunodeficient nude rats. These experiments involved exposure to concentrations of DBP from 100 to 500 mg/kg/day for only 2 days. After exposure, the testis fragments were harvested from the hosts and incubated; testosterone production was then assayed ex vivo (Fig. 3). Unsurprisingly, considering the technical challenge of properly recovering several 1 mm3 human testis fragments 2 days after their xenografting under the skin of the recipient rat, the authors found that ‘the results from the testosterone assay performed with the human fetal testes were highly variable’. As a substitute to their inability to assay testosterone itself or to weigh the accessory glands (the rat hosts were not castrated), the authors used RT-qPCR analysis to assess the expression of CYP11A1, CYP17A1, SCARB1, STAR and INSL3 genes in the recovered testes fragments. They showed no significant differences between untreated controls and those treated with any DBP dose and concluded that ‘human fetal testis xenografts are resistant to phthalate-induced endocrine disruption’. However, although the differences were not significant, the average value for CYP11A1 expression presented in this study appeared to be decreased by ∼30% after exposure to 250 mg/kg/day DBP for 2 days (n = 10), and INSL3 expression by the Leydig cells also appeared to be >80% lower after exposure to 100 mg/kg/day DBP (n = 6). Of note is that the authors observed a DBP-induced induction of MNG which may be due to a DBP-induced Sertoli cell dysfunction. However, Sertoli cell function was not investigated is this study. In any case, interpretation on the cause of induction of the MNGs should also necessarily integrate the consideration of a direct toxicity of DBP on the germ line as described above.
Phthalates and the androgenic status of the human neonatal testis
Neonatal boys do not escape from exposure to phthalates in their environment. Thus, neonates receiving intensive medical treatments through polyvinyl chloride (PVC)-based devices displayed phthalate esters exposure levels up to 10–20 mg/kg/day (Loff et al., 2000). Furthermore, the urine concentrations of MBzP are significantly higher in 2–6-month-old infants with PVC flooring in their bedrooms (Carlstedt et al., 2012). Significant dose-dependent associations were found between neonatal exposure to phthalate monoesters in breast milk and levels or ratios of reproductive hormones in boys at 3 months of age: MEP and MBP correlated positively with SHBG (sex hormone-binding globulin); MMP, MEP and MBP correlated with the LH-to-free testosterone ratio; and mono-iso-nonyl phthalate (MiNP) correlated with LH. MBP levels were also negatively correlated with free testosterone (Main et al., 2006). In the same study, no association was found between phthalate levels and cryptorchidism.
As there is a scarcity of data for neonatal boys, the results of two studies, which used in vivo exposure of young male marmosets to tackle the neonatal exposure issue, are of importance. Four-day-old co-twin marmosets were treated with vehicle or 500 mg/kg/day MBP for 14 days, and there was no significant difference in plasma testosterone levels (Hallmark et al., 2007) or in the germ cell morphology or number (McKinnell et al., 2009). However, blood testosterone levels were lower in 2- to 7-day-old marmosets exposed to a single oral dose of 500 mg/kg MBP for 5 h than in controls (Hallmark et al., 2007), which establishes that the neonatal primate testes are likely to be sensitive to short-term exposure to phthalates.
Phthalates and the androgenic status of the human prepubertal and pubertal testes
Male marmosets (Callithrix jacchus) treated daily with 0, 100, 500 or 2500 mg/kg of DEHP by oral gavage for 13 or 65 weeks from weaning (3 months of age) to sexual maturity (18 months of age) did not present any change in their testis weight, testicular zinc levels, blood levels of testosterone and estradiol or microscopic appearance of their testes (Kurata et al., 1998; Tomonari et al., 2006).
The latter experiments on preadolescent non-human primates are important as potential sensitivity to phthalate exposure is not limited to the fetal and neonatal periods: puberty, despite being crucial for the establishment of sexual maturity, and adulthood have largely, and wrongly, been neglected.
A Danish study estimates that the maximal exposure levels of DEHP alone in small children to be 100 µg/kg/day (Müller et al., 2003). Chinese school children were shown to present urine concentrations of ∼120 ng/ml of DEHP metabolites (Wang et al., 2013). Others found a median estimate of 4–8 µg/kg/day and a 95th percentile estimate of 15–25 µg/kg/day in German children (Wittassek et al., 2007). The fact that, in the latter study, exposure levels of several children exceeded the EU tolerable daily intake (TDI; 50 µg/kg/day) shows that children are exposed to considerable doses of phthalates, higher than adults in the general population (Wittassek et al., 2011).
A recent health risk assessment for phthalate exposure associated with childcare facilities and indoor playgrounds showed that ingestion and dermal exposure, in addition to eating contaminated food, are probably major routes of phthalate exposure (Kim et al., 2011). A cumulative risk assessment of phthalate exposure in Danish children and adolescents recently showed that a number of them have exceeded the regulatory acceptable levels of exposure for anti-androgenic effects as defined by the European Food and Safety Administration (EFSA) TDI, even though only five phthalic compounds, namely di-iso-butyl phthalate, DBP, DEHP, benzylbutyl phthalate and di-iso-nonyl phthalate (DiNP), were investigated (Soeborg et al., 2012).
Plasma concentrations of DEHP and MEHP were found to be significantly higher than normal in Turkish adolescents with pubertal gynaecomastia, which corresponds to an abnormal enlargement of male breast attributable to the proliferation of ductile elements (Durmaz et al., 2010). In contrast, a cross-sectional Danish study did not find any association between phthalate exposure, testicular growth, measurable testosterone or pubertal gynecomastia (Mieritz et al., 2012). As discussed by the latter authors, these divergent findings may be explained by the fact that ‘the phthalate level is up to 30 times higher in the serum of the Turkish boys than in the urine from the Danish boys in [this] study’. This contrasts with what was observed in girls, for whom delayed pubarche (but not thelarche) was associated with high phthalate concentrations in urine samples (Frederiksen et al., 2012). A more recent study on Danish boys in which urinary phthalates have been measured twice a year during a 5-year period showed that excretion of MBzP metabolites above the geometric group mean was associated with lower levels of adrenal androgens but positively associated with testosterone levels at 13 years of age (Mouritsen et al., 2013). Furthermore, in the same study, a lower age at pubarche was observed in boys with excretion of MBP (exposure to DBP) above the geometric group mean (11.0 years old versus 12.3 years old, P = 0.005).
Phthalates and male adult reproductive function
Phthalate exposure during adulthood
Considerable efforts have been devoted to the determination of phthalate levels in environmental and biological animal and human samples (Duty et al., 2003; Kato et al., 2003; Takatori et al., 2004; Buchta et al., 2005; Jönsson et al., 2005; Koch et al., 2005a, b, c, d; Hauser et al., 2006, 2007; Pan et al., 2006; Wirth et al., 2008; Han et al., 2009; Herr et al., 2009; Meeker et al., 2009; Frederiksen et al., 2010; Mendiola et al., 2011; Pant et al., 2011; Ikonomou et al., 2012; Joensen et al., 2012; Kessler et al., 2012; Olsen et al., 2012).
A study examining the relationship between urinary metabolites of phthalates and semen characteristics in healthy adult Swedish men failed to demonstrate any association between MBP, MBzP and MEHP exposure and reproductive biomarkers such as semen volume, sperm concentration and motility, sperm chromatin integrity or the serum concentrations of various hormones, including testosterone (Jönsson et al., 2005). Accordingly, Hauser et al. (2006) found no convincing relationship between three DEHP metabolites (including MEHP) and semen quality. The same conclusion was held in a study on 349 men from subfertile couples who showed no significant association between sperm concentration and motility and concentrations of DEHP metabolites (5-oxo-MEHP, 5-OH-MEHP, 5-carboxy-mono(2-ethylhexyl) phthalate and MEHP) determined in spot urine samples (Herr et al., 2009). In contrast, Duty et al. (2003) evidenced a dose–response relation between exposure to MBP and sperm motility and concentration, as well as between exposure to MBzP and sperm concentration, in an American cohort study. Jönsson et al. (2005) explain these discrepancies between their study and that of Duty et al. by the fact that their investigation was based on Swedish men aged from 18 to 21 years old in the general population, whereas Duty et al. used a cohort of men from subfertile couples with an average age of 36.4 years. A significant dose–response relationship was also found between the levels of MBP in the urine of subfertile men and low sperm concentration and motility, as well as ‘suggestive evidence’ of an association between high MBzP exposure and low sperm concentration. However, no relationship was found between MEP, MMEP or DEHP metabolites and these semen characteristics (Jönsson et al., 2005). Hauser et al. (2007) found an association between urinary levels of MEHP and sperm DNA damage, while a decrease in sperm motility and sperm cytotoxicity was evidenced after incubations with DEHP and MEHP (Pant et al., 2011). A cohort of the male partners from subfertile couples in the Great Lakes area in the USA reported a significant association between low sperm concentrations and above-median concentrations of MEP, and between abnormal sperm morphology and above-median mono-(3-carboxypropyl) phthalate concentrations (Wirth et al., 2008). Pan et al. (2006) reported that male workers occupationally exposed to MBP (129.6 μg/g creatinine) and MEHP (5.7 μg/g creatinine) had significantly lower free testosterone levels than unexposed men. Another study also showed that urinary MEHP concentrations were inversely associated with free testosterone and estradiol levels in infertile adult men (Meeker et al., 2009). Accordingly, urinary MEHP, MEOHP and MEHHP levels in fertile men were found to be associated with minor alterations of markers of free testosterone (total, calculated free testosterone and free androgen index, which is the ratio of total testosterone to SHBG levels) (Mendiola et al., 2011). More recently, a cohort study on 881 healthy Danish men evidenced negative associations between urinary proportions of MEHP and MiNP and serum testosterone/LH or follicle-stimulating hormone (FSH) ratios. MEHP was also negatively associated with total testosterone, free testosterone and ratios of testosterone/estradiol, whereas MiNP was positively correlated with SHBG (Meeker et al., 2009). However, consistent with Jönsson et al. (2005) and Herr et al. (2009), no convincing association between urinary proportions of MEHP and MiNP and semen quality variables, such as semen volume, sperm concentration, total sperm count, sperm motility or morphology, could be observed (Joensen et al., 2012). Finally, the percentage of the MEHP and MiNP metabolites calculated from the concentrations of their respective parent compounds DEHP and DiNP excreted in the urine of Danish men was negatively associated with the testosterone/LH and testosterone/FSH ratios. The percentage of MEHP was also negatively associated with total testosterone, free testosterone and ratio of testosterone/estradiol (Joensen et al., 2012).
Direct exposure of the human adult testis to phthalates
Only three studies have addressed the issue of the direct effects of phthalates in the human adult testis. Desdoits-Lethimonier et al. (2012) used an organotypic culture model of adult human testis explants and showed that both DEHP and MEHP can alter testosterone production by the human testis, but none of the phthalates tested had any effect on the Sertoli cell and germ cell activities and functions investigated, or on the Leydig cell number. The effects of both phthalates appeared to be specific for steroidogenesis, as INSL3 production by Leydig cells was not affected. This study also revealed that the human adult testis is able to metabolize both of these phthalates. Using the 14C-DEHP distribution assay previously designed by Chauvigné et al. (2009), the authors were able to estimate that, when 10−5 M of the phthalates were included in the culture medium for 24 h, only ∼3–6% of the DEHP and MEHP became associated with the testis explants. Combined with the findings for testicular DEHP biotransformation, this allowed the authors to estimate that phthalate concentrations of ∼1 mg/ml could display significant anti-androgenic effects; this dose is about the same order of magnitude as the highest concentrations found in men in epidemiological studies associating exposure to phthalates and impairment of the androgenic status. The anti-androgenic properties of DEHP and MEHP were confirmed by work with the human steroidogenic cell line NCI-H295R (Desdoits-Lethimonier et al., 2012).
Complementary in some aspects to the study on human testis explants, data from experiments with human testis microsomes confirmed that DEHP can be rapidly metabolized into MEHP by the human testis (Choi et al., 2012). Also, phthalates appear to inhibit human testicular 3β-hydroxysteroid dehydrogenase (3β-HSD) and 17β-hydroxysteroid dehydrogenase 3 (17β-HSD3) activities, and these enzymes play critical roles in the biochemical pathways by which cholesterol is converted into androgens (Yuan et al., 2012).
Conclusion
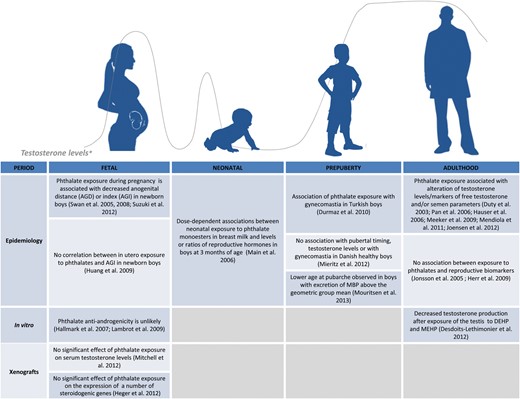
Despite >20 years of investigations, an overview of the studies of the effects of phthalates on the human male genital tract and reproductive functions (Fig. 4) indicates that no firm or general conclusion can yet be drawn as to whether or not phthalates exert anti-androgenic effects in humans, in particular before adulthood. The current literature highlights several notable points.
Summary of major studies on the effects of phthalates on the human testis at different ages. *Lifelong testosterone levels were adapted from Wilson (1999). To complement the human data, it is also interesting to note that no germ cell number or differentiation alterations after exposure to phthalates were seen in the young marmoset (McKinnell et al., 2009), while testosterone was found to decrease after exposure of neonatal marmosets to MBP (Hallmark et al., 2007). No changes in testis weight or morphology or in blood levels of testosterone were found after exposure to phthalates in the preadolescent marmoset (Kurata et al., 1998; Tomonari et al., 2006). DEHP, di-(2-ethylhexyl) phthalate; MBP, monobutyl phthalate; MEHP, mono-(2-ethylhexyl) phthalate.
Number of experiments in animals versus humans
There is a huge difference between the number of studies in animals and in humans, the latter being far too low and somewhat contradictory.
Inter-species differences
There are differences in the ways rats, mice, primates and humans respond to exposure to phthalates in vivo. The overwhelming majority of studies used rats, which appear to be particularly susceptible to the anti-androgenicity of phthalates. While exposure of the fetal mouse testis to phthalates does not seem to affect the constitutive production of testosterone by the Leydig cell, these compounds display anti-androgenic capabilities post-natally. Issues which should also be central in considering the interpretation of the divergent effects of phthalates between species are such as: (i) the large differences existing between mammalian species in the organization, structure and biology of the interstitial tissue (Fawcett et al., 1973; Huhtaniemi et al., 1982); (ii) the possible existence of age-dependent windows of susceptibility to phthalates during fetal life and after birth which vary among species; (iii) the intrinsic difference of timing scale in the development of the testis between species; or (iv) the differences between experimental protocols between the various laboratories (e.g. differences in the animal strains used, culture conditions).
Human exposure to phthalates
When considering data on human exposure to phthalates, a number of important aspects should be taken into consideration (Calafat and McKee, 2006; Wittassek et al., 2011), such as: (i) the relatively short half-life of the phthalates leading to possible significant intra-individual variations; (ii) the fact that biomonitoring of phthalates like DEHP and MEHP in body fluids can be impaired by analytical disturbances resulting from their unavoidable analytical background (Takatori et al., 2004; Kessler et al., 2012); (iii) the use of different biomatrices to assess body burden and the comparison of urinary levels versus serum levels (or semen levels); (iv) the inclusion of different metabolites, in relation to possible inter-individual differences in the metabolism of these compounds; (v) the intrinsic difficulty there is in estimating exposure itself (e.g. for urine: urinary excretion expressed as amount per bodyweight or as concentration and as total amount; Mouritsen et al., 2013); and (vi) the determination of accurate molar conversion factors to relate external phthalate exposure to body fluids metabolite concentrations. The way these points may be apprehended often differs between studies, further complicating the appreciation of human exposure, and may at least partly explain the divergent results between human studies at all the ages at which human data on exposure have been established.
Epidemiology
Regarding epidemiological studies, (i) there is insufficient data on fetal, post-natal, prepubertal and pubertal boys, despite a number of indications that the health risk posed by phthalates on reproductive health should be taken into consideration, (ii) more work is required to clarify the contradictions in the too few existing epidemiological studies dealing with phthalate exposure during pregnancy, and (iii) epidemiological and in vitro studies relevant to adult men generally converge sufficiently to support the contention that phthalate anti-androgenicity is plausible. In addition, the possible inherited consequences of an exposure to phthalates should also be taken into consideration in future (and difficult) epidemiological studies.
In vitro and xenograft studies
Few studies have yet investigated the direct effects of phthalates on the fetal human testis. They conclude that fetal human testis appears ‘unlikely’ to be sensitive to phthalate exposure, or even ‘resistant’. However, for the useful in vitro and xenografting models to fulfil their ‘missing link’ roles between rodent models and epidemiological studies in humans, a number of limitations of the current approaches using the human fetal testis need to be addressed: (i) a larger number of independent experiments are required when these system models are used, to better circumvent both the intrinsic and substantial individual variability resulting from the use of human biological materials and the problems associated with pooling testes at different fetal ages, which in particular may result in missing any potential critical windows of testicular sensitivity that have been identified both in rats (Carruthers and Foster, 2005; Welsh et al., 2008) and in humans (Mazaud-Guittot et al., 2013,); (ii) whenever possible, the durations of exposure should be optimized (i.e. extended) prior to concluding the absence of effects from short-term exposure findings; (iii) great caution should be taken in the interpretation of the changes occurring in the germ line following phthalate exposure, in particular when these changes do not seem to be associated with the disruption of any of the testicular endocrine parameters (i.e. actual hormone level assessment), as the disturbance of the germ cell microenvironment inherent in these culture/xenografting model systems is likely to make germ cells more vulnerable to chemical toxicity; (iv) the important issue of the potential testicular phthalate-induced epigenetic changes during fetal life should also be addressed in vitro and/or in the xenograft model system. Caution before concluding that phthalates are innocuous in the human fetal testis should be kept until these issues have been addressed.
Regarding the in vitro models specifically, (i) more attention should also be paid to the assessment of critical experiment parameters in the in vitro experiments that affect their sensitivity such as solvent type, biotransformation of the compound tested or incubation times, as recently done for the yeast estrogen screening assay (Dhooge et al., 2006); (ii) optimization of each in vitro assay for a given species should be a major objective, rather than adopting consensual culture conditions for the different species (mouse, rat, human) as the structure and organization of the interstitial compartment of the testis where the androgen-producing Leydig cells reside are different between mammals; (iii) when addressing endocrine disruption, actual assays of hormones (e.g. INSL3, inhibin B, etc.) themselves are by far more pertinent than assessing the levels of their mRNA by PCR as RNA levels represent at a given time only the steady-state levels between their transcription, translation and degradation. In this respect, it should be kept in mind that ‘it is the peptide [the hormone], not the message, that binds […] receptors’, as stated by Wilson et al. (2007), or that the mRNAs of the receptors do not have the ability to bind the hormone(s).
Regarding the xenograft model system, and in addition to different points listed above: (i) other possible sources of reproductive hormones than the testicular source may be considered, as they may interfere with the determination of testicular hormone levels; and (ii) the potential differences in the metabolizing capabilities of the parent compounds between the animal hosts (nude rat or nude mouse) and humans should be investigated and, if any, should also be taken into account in the study design and interpretation of the data.
Overall, the difficulties encountered in the development of adequate experimental approaches, their interpretation and their integration in assessing the risk associated with exposure to phthalates are very instructive about the challenge posed by EDs in general.
Authors' roles
O.A. and B.J. both contributed to the critical analysis of the literature and drafting of the manuscript.
Funding
The authors are grateful to Inserm, University of Rennes 1, EHESP-School of Public Health, Agence nationale de sécurité sanitaire (Anses), Institut National de l'EnviRonnement industriel et des rISques (INERIS) and Region Bretagne for their support.
Conflict of interest
The authors declare they have no competing interests be it financial, personal or professional.
Acknowledgements
The authors thank the colleagues from different disciplines and laboratories for information, discussion and advice they have provided for this review, to be as documented and integrated as possible. They especially thank Laurianne Lesné for the helpful discussions and support during the writing process.