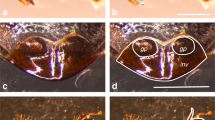
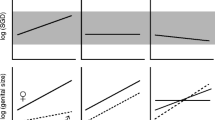
Abstract
Insects show relatively little genital variation within species compared to extraordinary and often rapid diversification among species. It has been suggested that selection for reproductive isolation through differences in genital shape might explain this phenomenon. This hypothesis predicts that populations diverge faster in genital shape than in genital size. We tested this prediction in males from 10 dung beetle species with known phylogenetic relationships from the genus Onthophagus (Coleoptera: Scarabaeidae), including four species for which we were able to sample multiple populations. Specifically, we compared intra- and interspecific differentiation in shape and relative sizes of genitalia and calculated their respective evolutionary rates. We compared these rates to two similarly sized non-genital traits, the head and the fore-tibia. We found significant intraspecific variation in genital shape in all four species for which multiple populations were sampled, but for three of them we also identified significant relative size variation. We also found that genital shape evolved at higher rates than relative genital size. Genital shape evolved faster than head shape, but not fore-tibia shape. However, shapes of all measured structures evolved faster than their relative size. We discuss the functional constraints that may bias the developmental evolution of relative size and shape of genitalia and other morphological traits.




Similar content being viewed by others
References
Adams DC (2014) Quantifying and comparing phylogenetic evolutionary rates for shape and other high-dimensional phenotypic data. Syst Biol 63(2):166–177. https://doi.org/10.1093/sysbio/syt105
Arnegard ME, McIntyre PB, Harmon LB, Zelditch ML, Crampton WGR, Davis JK, Sullivan JP, Lavoue S, Hopkins CD (2010) Sexual signal evolution outpaces ecological divergence during electric fish species radiation. Am Nat 176(3):335–356. https://doi.org/10.1086/655221
Arnqvist G (1998) Comparative evidence for the evolution of genitalia by sexual selection. Nature 325:784–786
Aspöck H, Aspöck U, Rausch H (1991) Die Raphidiopteren der Erde. Goecke & Evers, Krefeld
Bath E, Tatarnic N, Bonduriansky R (2012) Asymmetric reproductive isolation and interference in neriid flies: the roles of genital morphology and behavior. Anim Behav 84:1331–1339
Bertin A, Fairbairn DJ (2007) The form of sexual selection on male genitalia cannot be inferred from within-population variance and allometry—a case study in Aquarius remigis. Evolution 61(4):825–837. https://doi.org/10.1111/j.1558-5646.2007.00074.x
Bookstein FL (1991) Morphometric tools for landmark data: geometry and biology. Cambridge Univ Press, New York
Brower AV (1994) Rapid morphological radiation and convergence among races of the butterfly Heliconius erato inferred from patterns of mitochondrial DNA evolution. PNAS 91(14):6491–6495. https://doi.org/10.1073/pnas.91.14.6491
Carlson BA, Hasan SM, Hollmann M, Milller DB, Harmon LJ, Arnegard ME (2011) Brain evolution triggers increased diversification of electric fishes. Science 332(6029):583–586. https://doi.org/10.1126/science.1201524
Dryden IL, Mardia KV (1998) Statistical shape analysis. Wiley, Chichester
Eberhard WG (1977) Fighting behavior of male Golofa porteri beetles (Scarabaeidae: Dynastinae). Psyche 83:292–298
Eberhard WG (1985) Sexual selection and animal genitalia. Harvard University Press, Massachusetts. https://doi.org/10.4159/harvard.9780674330702
Eberhard WG (2009) Static allometry and animal genitalia. Evolution 63(1):48–66. https://doi.org/10.1111/j.1558-5646.2008.00528.x
Eberhard WG, Huber BA, Rodriguez RL, Briceno RD, Salas I, Rodriquez V (1998) One size fits all? Relationships between the size and degree of variation in genitalia and other body parts in twenty species of insects and spiders. Evolution 52(2):415–431. https://doi.org/10.1111/j.1558-5646.1998.tb01642.x
Eberhard W, Rodriguez RL, Polihronakis M (2009) Pitfalls in understanding the functional significance of genital allometry. J Evol Biol 22(3):435–445. https://doi.org/10.1111/j.1420-9101.2008.01654.x
Emlen DJ (1994) Environmental control of horn length dimorphism in the beetle Onthophagus acuminatus Coleoptera, Scarabaeidae. Proc R Soc London Ser B 256(1346):131–136. https://doi.org/10.1098/rspb.1994.0060
Emlen DJ (1997) Alternative reproductive tactics and male-dimorphism in the horned beetle Onthophagus acuminatus (Coleoptera: Scarabaeidae). Behav Ecol and Sociobiol 41(5):335–341. https://doi.org/10.1007/s002650050393
Emlen DJ, Nijhout HF (2000) The development and evolution of exaggerated morphologies in insects. Annu Rev Entomol 45(1):661–708. https://doi.org/10.1146/annurev.ento.45.1.661
Emlen DJ, Marangelo J, Ball B, Cunningham CW (2005) Diversity in the weapons of sexual selection: horn evolution in the beetle genus Onthophagus (Coleoptera: Scarabaeidae). Evolution 59(5):1060–1084. https://doi.org/10.1111/j.0014-3820.2005.tb01044.x
Estrada AP, Prieto R (2011) Genetic variation in isolated populations of Drosophila mercatorum (Diptera: Drosophilidae) in semi-arid ecosystems. Rev Col Entomol 37:318–326
Felsenstein J (1985) Phylogenies and the comparative method. Am Nat 125(1):1–15. https://doi.org/10.1086/284325
Frankino WA, Zwaan BJ, Stern DL, Brakefield PM (2005) Natural selection and developmental constraints in the evolution of allometries. Science 307(5710):718–720. https://doi.org/10.1126/science.1105409
Garland T (1992) Rate tests for phenotypic evolution using phylogenetically independent contrasts. Am Nat 140(3):509–519. https://doi.org/10.1086/285424
Grimaldi D, Nguyen T (1999) Monograph on the spittlebug flies, genus Cladochaeta (Diptera: Drosophilidae: Cladochaetini). B Am Museum Nat Hist 241:1–326
Grimaldi D, Engel MS (2005) Evolution of insects. Cambridge University Press, New York
Hanski I, Cambefort Y (eds) (1991) Dung beetle ecology. Princeton University Press, Princeton. https://doi.org/10.1515/9781400862092
Hosken DJ, Stockley P (2004) Sexual selection and genital evolution. Trends Ecol Evol 19:87–93
Hotzy C, Arnqvist G (2009) Sperm competition favors harmful males in seed beetles. Curr Biol 19(5):404–407. https://doi.org/10.1016/j.cub.2009.01.045
House CM, Simmons LW (2003) Genital morphology and fertilization success in the dung beetle Onthophagus taurus: an example of sexually selected male genitalia. Proc R Soc London Ser B 270:447–455
House CM, Simmons LW (2005) The evolution of male genitalia: patterns of genetic variation and covariation in the genital sclerites of the dung beetle Onthophagus taurus. J Evol Biol 18(5):1281–1292. https://doi.org/10.1111/j.1420-9101.2005.00926.x
House CM, Simmons LW (2006) Offensive and defensive sperm competition roles in the dung beetle Onthophagus taurus (Coleoptera: Scarabaeidae). Behav Ecol Sociobiol 60(2):131–136. https://doi.org/10.1007/s00265-005-0149-x
House CM, Simmons LW (2007) No evidence for condition-dependent expression of male genitalia in the dung beetle Onthophagus taurus. J Evol Biol 20(4):1322–1332. https://doi.org/10.1111/j.1420-9101.2007.01346.x
House CM, Simmons LW (2012) The genetics of primary and secondary sexual character trade-offs in a horned beetle. J Evol Biol 25:1711–1717
Hunt J, Simmons LW (2002) Confidence of paternity and paternal care: covariation revealed through the experimental manipulation of the mating system in the beetle Onthophagus taurus. J Evol Biol 5:784–795
Huxley J (1932) Problems of relative growth. MacVeagh, London
International Union for Conservation of Nature and Natural Resources (2008) http://www.iucn.org/about/work/programmes/species/red_list/2008_red_list_summary_statistics/
Ito J, Ohkubo Y, Hasegawa E (2017) Morph-specific weapon-correlated traits in a male dimorphic stag beetle Prosopocoilus inclinatus (Coleoptera: Lucanidae). Ann Entomol Soc Am 110:281–285
Kamimura Y, Mitsumoto H (2012) Lock-and-key structural isolation between sibling Drosophila species. Entomol Sci 15:197–201
Klingenberg CP (2011) MorphoJ: an integrated software package for geometric morphometrics. Mol Ecol Res 11:353–357
Krikken J, Huijbregts J (2008) Sulawesi large-eyed Onthophagus and their relatives: seven new species, with a key (Coleoptera: Scarabaeidae: Scarabaeinae). Tijdschr Entomol 151(2):155–171. https://doi.org/10.1163/22119434-900000261
Langley CH, Fitch WM (1974) An examination of the constancy of the rates of mole evolution. J Mol Evol 3(3):161–177. https://doi.org/10.1007/BF01797451
Lynch M (1990) The rate of morphological evolution in mammals from the standpoint of the neutral expectation. Am Nat 136(6):727–741. https://doi.org/10.1086/285128
Macagno ALM, Pizzo A, Roggero A, Rolando A, Palestrini C (2009) Horn polyphenism and related head shape variation in a single-horned dung beetle: Onthophagus (Palaeonthophagus) fracticornis (Preyssler, 1790) (Coleoptera: Scarabaeidae). J Zool Syst Evol Res 47:96–102
Macagno ALM, Pizzo A, Parzer HF, Palestrini C, Rolando A, Moczek AP (2011a) Shape—but not size—codivergence between male and female copulatory structures in Onthophagus beetles. PLoS One 6(12):e28893. https://doi.org/10.1371/journal.pone.0028893
Macagno ALM, Pizzo A, Rolando A, Palestrini C (2011b) Size and shape interspecific divergence patterns partly reflect phylogeny in an Onthophagus species-complex (Coleoptera: Scarabaeidae). Zool J Linnean Soc 162(3):482–498. https://doi.org/10.1111/j.1096-3642.2010.00684.x
Macagno ALM, Moczek AP, Pizzo A (2016) Concerted differentiation of nesting depth and digging appendages among tunneling dung beetle populations and species. Am Nat 187:E000–E000
Mahalanobis PC (1936) On the generalized distance in statistics. Proc Natl Inst Sci Ind 2:49–55
Manly BFJ (2007) Bootstrap and Monte Carlo methods in biology, 3rd edn. Chapman and Hall, Boca Raton, Florida
Martins EP, Garland T (1991) Phylogenetic analyses of the correlated evolution of continuous characters: a simulation study. Evolution 45(3):534–557. https://doi.org/10.1111/j.1558-5646.1991.tb04328.x
Martins EP, Hansen TF (1997) Phylogenies and the comparative method: a general approach to incorporating phylogenetic information into the analysis of interspecific data. Am Nat 149(4):646–667. https://doi.org/10.1086/286013
McPeek MA, Shen L, Torrey JZ (2008) The tempo and mode of three-dimensional morphological evolution in male reproductive structures. Am Nat 171:E158–E178
McPeek MA, Symes LB, Zong DM, McPeek CL (2011) Species recognition and patterns of population variation in the reproductive structures of a damselfly genus. Evolution 65(2):419–428. https://doi.org/10.1111/j.1558-5646.2010.01138.x
Minitab 17 Statistical Software (2010) Computer software. Minitab, Inc., State College, PA. www.minitab.com
Moczek AP, Nijhout HF (2004) Trade-offs during the development of primary and secondary sexual traits in a horned beetle. Am Nat 163(2):184–191. https://doi.org/10.1086/381741
Moczek AP, Cochrane J (2006) Intraspecific female brood parasitism in the dung beetle Onthophagus taurus. Ecol Entomol 31(4):316–321. https://doi.org/10.1111/j.1365-2311.2006.00773.x
Okada Y, Suzaki Y, Miyatake T, Okada K (2012) Effect of weapon-supportive traits on fighting success in armed insects. Anim Behav 83(4):1001–1006. https://doi.org/10.1016/j.anbehav.2012.01.021
O’Meara BC, Ané C, Sanderson MJ, Wainwright PC (2006) Testing for different rates of continuous trait evolution using likelihood. Evolution 60(5):922–933. https://doi.org/10.1111/j.0014-3820.2006.tb01171.x
Palestrini C, Rolando A, Laiolo P (2000) Allometric relationships and character evolution in Onthophagus taurus (Coleoptera: Scarabaeidae). Can J Zool 78:1199–1206
Parzer HF, Moczek AP (2008) Rapid antagonistic coevolution between primary and secondary sexual characters in horned beetles. Evolution 62(9):2423–2428. https://doi.org/10.1111/j.1558-5646.2008.00448.x
Piera M, Zunino M (1986) Analisi sistematica, filogenetica e biogeografica di un gruppo di specie del sottogenere Palaeonthophagus Zunino, 1979 (Coleoptera, Scarabaeidae: genere Onthophagus): il gruppo ovatus. Boll Mus Reg Sci Nat Torino 4:413–467
Pizzo A, Mercurio D, Palestrini C, Roggero A, Rolando A (2006a) Male differentiation patterns in two polyphenic sister species of the genus Onthophagus Latreille, 1802 (Coleoptera: Scarabaeidae): a geometric morphometric approach. J Zool Syst Evol Res 44:54–62
Pizzo A, Roggero A, Palestrini C, Cervella P, Del Pero M, Rolando A (2006b) Genetic and morphological differentiation patterns between sister species: the case of Onthophagus taurus and Onthophagus illyricus (Coleoptera, Scarabaeidae). Biol J Linn Soc 89(2):197–211. https://doi.org/10.1111/j.1095-8312.2006.00674.x
Pizzo A, Roggero A, Palestrini C, Moczek AP, Rolando A (2008) Rapid shape divergences between natural and introduced populations of a horned beetle partly mirror divergences between species. Evol Dev 10(2):166–175. https://doi.org/10.1111/j.1525-142X.2008.00224.x
Pizzo A, Mazzone F, Rolando A, Palestrini C (2011) Combination of geometric morphometric and genetic approaches applied to a debated taxonomical issue: the status of Onthophagus massai (Coleoptera, Scarabaeidae) as an endemic species vicarious to Onthophagus fracticornis in Sicily. Zoology 114(4):199–212. https://doi.org/10.1016/j.zool.2011.03.003
Pizzo A, Macagno ALM, Dusini S, Palestrini C (2012) Trade-off between horns and other functional traits in two Onthophagus species (Scarabaeidae, Coleoptera). Zoomorphology 131(1):57–68. https://doi.org/10.1007/s00435-012-0148-1
Polak M, Rashed A (2010) Microscale laser surgery reveals adaptive function of male intromittent genitalia. Proc R Soc London Ser B 277(1686):1371–1376. https://doi.org/10.1098/rspb.2009.1720
Polly PD (2016a) Geometric morphometrics for Mathematica. Version 12.0. Department of Geological Sciences, Indiana University: Bloomington, Indian Forester http://mypage.iu.edu/~pdpolly/Software.html
Polly PD (2016b) Phylogenetics for Mathematica. Version 4.0. Department of Geological Sciences, Indiana University: Bloomington, Indian Forester http://mypage.iu.edu/~pdpolly/Software.html
Price DL (2004) Species diversity and seasonal abundance of scarabaeoid dung beetles (Coleoptera: Scarabaeidae, Geotrupidae and Trogidae) attracted to cow dung in central New Jersey. J New York Entomol Soc 112(4):334–347.
Revell LJ, Harmon LJ (2008) Testing quantitative genetic hypotheses about the evolutionary rate matrix for continuous characters. Evol Ecol Res 10:311–331
Richmond MP, Johnson S, Markow TA (2012) Evolution of reproductive morphology among recently diverged taxa in the Drosophila mojavensis species cluster. Ecol Evol 2:397–408
Roessner E, Schoenfeld J, Ahrens D (2010) Onthophagus (Palaeonthophagus) medius (Kugelann, 1792)—a good western palaearctic species in the Onthophagus vacca complex (Coleoptera: Scarabaeidae: Scarabaeinae: Onthophagini). Zootaxa 2629:1–28
Rohlf FJ (2010a) tpsUtil. http://life.bio.sunysb.edu/morph/
Rohlf FJ (2010b) tpsDIG2. http://life.bio.sunysb.edu/morph/
Rohlf FJ, Slice D (1990) Extensions of the Procrustes method for the optimal superimposition of landmarks. Syst Zool 39(1):40–59. https://doi.org/10.2307/2992207
Rowe L, Arnqvist G (2012) Sexual selection and the evolution of genital shape and complexity in water striders. Evolution 66(1):40–54. https://doi.org/10.1111/j.1558-5646.2011.01411.x
Sanderson MJ (2002) Estimating absolute rates of molecular evolution and divergence times: a penalized likelihood approach. Mol Biol Evol 19:101–109
Sanderson MJ (2006) r8s version 1.71. Analysis of rates (“r8s”) of evolution. http://loco.biosci.arizona.edu/r8s/
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675. https://doi.org/10.1038/nmeth.2089
Scoble MJ (1995) The Lepidoptera: form, function and diversity. Oxford University Press, Oxford
Scudder GGE (1971) Comparative morphology of insect genitalia. Annu Rev Entomol 16(1):379–406. https://doi.org/10.1146/annurev.en.16.010171.002115
Simmons LW, Emlen DJ (2006) Evolutionary trade-off between weapons and testes. Proc Natl Acad Sci U S A 103(44):16346–16351. https://doi.org/10.1073/pnas.0603474103
Simmons LW, Kotiaho JS (2007) The effects of reproduction on courtship, fertility and longevity within and between alternative male mating tactics of the horned beetle, Onthophagus binodis. J Evol Biol 20(2):488–495. https://doi.org/10.1111/j.1420-9101.2006.01274.x
Simmons LW, House CM, Hunt J, Garcia-Gonzalez F (2009) Evolutionary response to sexual selection in male genital morphology. Curr Biol 19(17):1442–1446. https://doi.org/10.1016/j.cub.2009.06.056
Simmons LW, Garcia-Gonzalez F (2011) Experimental coevolution of male and female genital morphology. Nature Comm 2:374. https://doi.org/10.1038/ncomms1379
Snell-Rood EC, Moczek AP (2012) Insulin signaling as a mechanism underlying developmental plasticity: the role of FOXO in a nutritional polyphenism. PLoS One 7(4):e34857. https://doi.org/10.1371/journal.pone.0034857
Tarasov SI, Kabakov ON (2010) Two new species of Onthophagus (Coleoptera: Scarabaeidae) from Indochina, with a discussion of some problems with the classification of Serrophorus and similar subgenera. Zootaxa 2344:17–28
Tomkins JL, Kotiaho JS, LeBas NR (2005) Phenotypic plasticity in the developmental integration of morphological trade-offs and secondary sexual trait compensation. Proc R Soc London Ser B 272(1562):543–551. https://doi.org/10.1098/rspb.2004.2950
Turelli M, Gillespie JH, Lande R (1988) Rate tests for selection on quantitative characters during macroevolution and microevolution. Evolution 42(5):1085–1089. https://doi.org/10.1111/j.1558-5646.1988.tb02526.x
Tuxen SL (1970) Taxonomist’s glossary of genitalia in insects. Munksgaard, Copenhagen
Werner M, Simmons LW (2008) The evolution of male genitalia: functional integration of genital sclerites in the dung beetle Onthophagus taurus. Biol J Linn Soc 93(2):257–266. https://doi.org/10.1111/j.1095-8312.2007.00924.x
Workman MS, Leamy LJ, Routman EJ, Cheverud JM (2002) Analysis of quantitative trait locus effects on the size and shape of mandibular molars in mice. Genetics 160(4):1573–1586
Zelditch ML, Swiderski DL, Sheets HD, Fink WL (2004) Geometric morphometrics for biologists: a primer. Elsevier Academic Press, London
Acknowledgements
We would like to thank Tami Cruickshank who graciously calculated divergence times, Anna Macagno for statistical questions, Teiya Kijimoto for providing images for each trait, Elise Morton for helping us with the creation of the size figures, and two anonymous reviewers for constructive comments that improved the manuscript.
Funding
This study was supported in part by National Science Foundation grants IOS 0445661 and IOS 0718522 to APM.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Special Issue “Size and Shape: Integration of morphometrics, mathematical modelling, developmental and evolutionary biology”, Guest Editors: Nico Posnien—Nikola-Michael Prpic.
Communicated by Nikola-Michael Prpic
Electronic supplementary material
ESM 1
(DOCX 1726 kb)
Rights and permissions
About this article
Cite this article
Parzer, H.F., David Polly, P. & Moczek, A.P. The evolution of relative trait size and shape: insights from the genitalia of dung beetles. Dev Genes Evol 228, 83–93 (2018). https://doi.org/10.1007/s00427-018-0602-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00427-018-0602-2