Redox Biology ( IF 11.4 ) Pub Date : 2019-12-26 , DOI: 10.1016/j.redox.2019.101413 Tzu-Chi Chen , Jian-Ying Chuang , Chiung-Yuan Ko , Tzu-Jen Kao , Pei-Yu Yang , Chun-Hui Yu , Ming-Sheng Liu , Siou-Lian Hu , Yu-Ting Tsai , Hardy Chan , Wen-Chang Chang , Tsung-I. Hsu
|
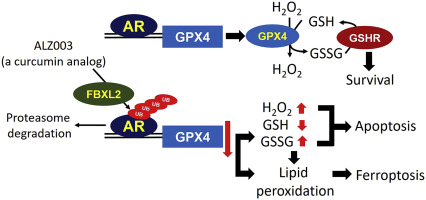
Drug resistance is the main obstacle in the improvement of chemotherapeutic efficacy in glioblastoma. Previously, we showed that dehydroepiandrosterone (DHEA), one kind of androgen/neurosteroid, potentiates glioblastoma to acquire resistance through attenuating DNA damage. Androgen receptor (AR) activated by DHEA or other types of androgen was reported to promote drug resistance in prostate cancer. However, in DHEA-enriched microenvironment, the role of AR in acquiring resistance of glioblastoma remains unknown. In this study, we found that AR expression is significantly correlated with poor prognosis, and AR obviously induced the resistance to temozolomide (TMZ) treatment. Herein, we observed that ALZ003, a curcumin analog, induces FBXL2-mediated AR ubiquitination, leading to degradation. Importantly, ALZ003 significantly inhibited the survival of TMZ-sensitive and –resistant glioblastoma in vitro and in vivo. The accumulation of reactive oxygen species (ROS), lipid peroxidation and suppression of glutathione peroxidase (GPX) 4, which are characteristics of ferroptosis, were observed in glioblastoma cell after treatment of ALZ003. Furthermore, overexpression of AR prevented ferroptosis in the presence of GPX4. To evaluate the therapeutic effect in vivo, we transplanted TMZ-sensitive or -resistant U87MG cells into mouse brain followed by intravenous administration with ALZ003. In addition to inhibiting the growth of glioblastoma, ALZ003 significantly extended the survival period of transplanted mice, and significantly decreased AR expression in the tumor area. Taken together, AR potentiates TMZ resistance for glioblastoma, and ALZ003-mediated AR ubiquitination might open a new insight into therapeutic strategy for TMZ resistant glioblastoma.
中文翻译:

姜黄素类似物诱导的AR泛素化通过破坏GPX4介导的氧化还原稳态而抑制了耐替莫唑胺的成胶质细胞瘤的生长。
耐药性是胶质母细胞瘤化疗疗效改善的主要障碍。以前,我们表明脱氢表雄酮(DHEA)是一种雄激素/神经甾体,可通过减轻DNA损伤来增强胶质母细胞瘤以获得耐药性。据报道,由脱氢表雄酮(DHEA)或其他类型的雄激素激活的雄激素受体(AR)可以提高前列腺癌的耐药性。但是,在富含DHEA的微环境中,AR在获得胶质母细胞瘤耐药性中的作用仍然未知。在这项研究中,我们发现AR表达与不良预后显着相关,并且AR明显诱导出对替莫唑胺(TMZ)治疗的耐药性。在这里,我们观察到姜黄素类似物ALZ003诱导FBXL2介导的AR泛素化,从而导致降解。重要的,体外和体内。在治疗ALZ003后,在胶质母细胞瘤细胞中观察到了活性氧(ROS)的积累,脂质过氧化和谷胱甘肽过氧化物酶(GPX)4的抑制,这是肥大症的特征。此外,在GPX4的存在下,AR的过表达防止了铁锈病。评估体内治疗效果,我们将TMZ敏感或耐药的U87MG细胞移植到小鼠大脑中,然后静脉注射ALZ003。除了抑制胶质母细胞瘤的生长外,ALZ003还显着延长了移植小鼠的生存期,并显着降低了肿瘤区域中AR的表达。综上所述,AR增强了胶质母细胞瘤对TMZ的耐药性,而ALZ003介导的AR泛素化可能为TMZ耐药性胶质母细胞瘤的治疗策略打开新的视野。

























 京公网安备 11010802027423号
京公网安备 11010802027423号