当前位置:
X-MOL 学术
›
J. Mol. Biol.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Structural Insights Into PfARO and Characterization of its Interaction With PfAIP.
Journal of Molecular Biology ( IF 5.6 ) Pub Date : 2019-12-23 , DOI: 10.1016/j.jmb.2019.12.024 Michael Geiger 1 , Chris Brown 2 , Jan Stephan Wichers 1 , Jan Strauss 1 , Andrés Lill 3 , Roland Thuenauer 3 , Benjamin Liffner 4 , Louisa Wilcke 5 , Sarah Lemcke 1 , Dorothee Heincke 1 , Samuel Pazicky 6 , Anna Bachmann 1 , Christian Löw 6 , Danny William Wilson 7 , Michael Filarsky 3 , Paul-Christian Burda 1 , Kun Zhang 2 , Murray Junop 2 , Tim Wolf Gilberger 1
Journal of Molecular Biology ( IF 5.6 ) Pub Date : 2019-12-23 , DOI: 10.1016/j.jmb.2019.12.024 Michael Geiger 1 , Chris Brown 2 , Jan Stephan Wichers 1 , Jan Strauss 1 , Andrés Lill 3 , Roland Thuenauer 3 , Benjamin Liffner 4 , Louisa Wilcke 5 , Sarah Lemcke 1 , Dorothee Heincke 1 , Samuel Pazicky 6 , Anna Bachmann 1 , Christian Löw 6 , Danny William Wilson 7 , Michael Filarsky 3 , Paul-Christian Burda 1 , Kun Zhang 2 , Murray Junop 2 , Tim Wolf Gilberger 1
Affiliation
|
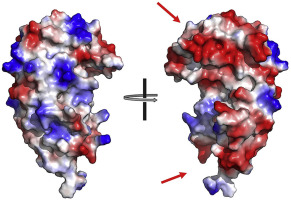
Apicomplexan parasites contain rhoptries, which are specialized secretory organelles that coordinate host cell invasion. During the process of invasion, rhoptries secrete their contents to facilitate interaction with, and entry into, the host cell. Here, we report the crystal structure of the rhoptry protein Armadillo Repeats-Only (ARO) from the human malaria parasite, Plasmodium falciparum (PfARO). The structure of PfARO comprises five tandem Armadillo-like (ARM) repeats, with adjacent ARM repeats stacked in a head-to-tail orientation resulting in PfARO adopting an elongated curved shape. Interestingly, the concave face of PfARO contains two distinct patches of highly conserved residues that appear to play an important role in protein-protein interaction. We functionally characterized the P. falciparum homolog of ARO interacting protein (PfAIP) and demonstrate that it localizes to the rhoptries. We show that conditional mislocalization of PfAIP leads to deficient red blood cell invasion. Guided by the structure, we identified mutations of PfARO that lead to mislocalization of PfAIP. Using proximity-based biotinylation we probe into PfAIP interacting proteins.
中文翻译:

深入了解PfARO及其与PfAIP相互作用的特性。
蚜虫寄生虫含有rhoptries,它们是协调宿主细胞入侵的专门的分泌细胞器。在入侵过程中,眼花opt分泌其内容物,以促进与宿主细胞的相互作用并进入宿主细胞。在这里,我们报告从人类疟疾寄生虫,恶性疟原虫(PfARO)的Rhoptry蛋白仅犰狳重复(ARO)的晶体结构。PfARO的结构包括五个串联的犰狳类(ARM)重复序列,相邻的ARM重复序列以头对尾方向堆叠,导致PfARO采用细长的弯曲形状。有趣的是,PfARO的凹面包含两个不同的高度保守残基补丁,它们似乎在蛋白质-蛋白质相互作用中起重要作用。我们在功能上表征了P。与ARO相互作用蛋白(PfAIP)的falciparum同源,并证明它定位于rhoptries。我们表明,PfAIP的条件错误定位会导致红细胞入侵不足。在结构的指导下,我们鉴定出导致PfAIP定位错误的PfARO突变。使用基于接近度的生物素化,我们探究了PfAIP相互作用蛋白。
更新日期:2019-12-23
中文翻译:

深入了解PfARO及其与PfAIP相互作用的特性。
蚜虫寄生虫含有rhoptries,它们是协调宿主细胞入侵的专门的分泌细胞器。在入侵过程中,眼花opt分泌其内容物,以促进与宿主细胞的相互作用并进入宿主细胞。在这里,我们报告从人类疟疾寄生虫,恶性疟原虫(PfARO)的Rhoptry蛋白仅犰狳重复(ARO)的晶体结构。PfARO的结构包括五个串联的犰狳类(ARM)重复序列,相邻的ARM重复序列以头对尾方向堆叠,导致PfARO采用细长的弯曲形状。有趣的是,PfARO的凹面包含两个不同的高度保守残基补丁,它们似乎在蛋白质-蛋白质相互作用中起重要作用。我们在功能上表征了P。与ARO相互作用蛋白(PfAIP)的falciparum同源,并证明它定位于rhoptries。我们表明,PfAIP的条件错误定位会导致红细胞入侵不足。在结构的指导下,我们鉴定出导致PfAIP定位错误的PfARO突变。使用基于接近度的生物素化,我们探究了PfAIP相互作用蛋白。

























 京公网安备 11010802027423号
京公网安备 11010802027423号