Redox Biology ( IF 11.4 ) Pub Date : 2019-12-23 , DOI: 10.1016/j.redox.2019.101412 Lu Li 1 , Jingqi Fu 1 , Dan Liu 1 , Jing Sun 1 , Yongyong Hou 1 , Chengjie Chen 1 , Junbo Shao 1 , Linlin Wang 2 , Xin Wang 1 , Rui Zhao 2 , Huihui Wang 3 , Melvin E Andersen 4 , Qiang Zhang 5 , Yuanyuan Xu 3 , Jingbo Pi 1
|
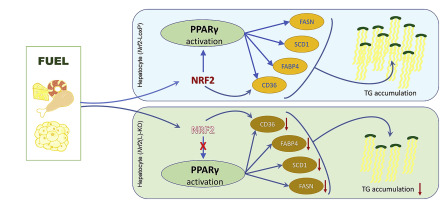
Non-alcoholic fatty liver disease (NAFLD) is an emerging global disease with increasing prevalence. However, the mechanism of NAFLD development is not fully understood. To elucidate the cell-specific role of nuclear factor erythroid-derived 2-like 2 (NRF2) in the pathogenesis of NAFLD, we utilized hepatocyte- and macrophage-specific Nrf2-knockout [Nrf2(L)-KO and Nrf2(Mϕ)-KO] mice to examine the progress of NAFLD induced by high-fat diet (HFD). Compared to Nrf2-LoxP littermates, Nrf2(L)-KO mice showed less liver enlargement, milder inflammation and less hepatic steatosis after HFD feeding. In contrast, Nrf2(Mϕ)-KO mice displayed no significant difference in HFD-induced hepatic steatosis from Nrf2-LoxP control mice. Mechanistic investigations revealed that Nrf2 deficiency in hepatocytes dampens the expression of peroxisome proliferator-activated receptor γ (PPARγ) and its downstream lipogenic genes in the liver and/or primary hepatocytes induced by HFD and palmitate exposure, respectively. While PPARγ agonists augmented PPARγ expression and its transcriptional activity in primary hepatocytes in a NRF2-dependent manner, forced overexpression of PPARγ1 or γ2 distinctively reversed the decreased expression of their downstream genes fatty acid binding protein 4, lipoprotein lipase and/or fatty acid synthase caused by Nrf2 deficiency. We conclude that NRF2-dependent expression of PPARγ in hepatocytes is a critical initiating process in the development of NAFLD, suggesting that inhibition of NRF2 specifically in hepatocytes may be a valuable approach to prevent the disease.
中文翻译:

肝细胞特异性 Nrf2 缺乏可减轻高脂饮食诱导的肝脂肪变性:涉及 PPARγ 表达减少。
非酒精性脂肪肝(NAFLD)是一种新兴的全球疾病,患病率不断上升。然而,NAFLD 发展的机制尚不完全清楚。为了阐明核因子红细胞衍生 2 样 2 (NRF2) 在 NAFLD 发病机制中的细胞特异性作用,我们利用了肝细胞和巨噬细胞特异性Nrf2 -敲除 [ Nrf2 (L)-KO 和Nrf2 (Mφ)- KO]小鼠检查高脂饮食(HFD)诱导的 NAFLD 的进展。与Nrf2 -LoxP 同窝小鼠相比,Nrf2 (L)-KO 小鼠在 HFD 喂养后表现出较少的肝脏肿大、较轻的炎症和较少的肝脏脂肪变性。相比之下,Nrf2 (Mphi)-KO 小鼠在 HFD 诱导的肝脂肪变性方面与Nrf2 -LoxP 对照小鼠没有显着差异。机制研究表明,肝细胞中的Nrf2缺陷会抑制 HFD 和棕榈酸酯暴露分别诱导的肝脏和/或原代肝细胞中过氧化物酶体增殖物激活受体 γ (PPARγ) 及其下游脂肪生成基因的表达。虽然 PPARγ 激动剂以 NRF2 依赖性方式增强原代肝细胞中 PPARγ 的表达及其转录活性,但 PPARγ1 或 γ2 的强制过表达明显逆转了其下游基因脂肪酸结合蛋白 4、脂蛋白脂肪酶和/或脂肪酸合酶引起的表达下降。由于Nrf2缺乏。我们得出的结论是,肝细胞中 NRF2 依赖性 PPARγ 表达是 NAFLD 发展的关键起始过程,这表明在肝细胞中特异性抑制 NRF2 可能是预防该疾病的一种有价值的方法。

























 京公网安备 11010802027423号
京公网安备 11010802027423号