当前位置:
X-MOL 学术
›
Gastroenterology
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Oncogenic Mutations in Armadillo Repeats 5 and 6 of β-Catenin Reduce Binding to APC, Increasing Signaling and Transcription of Target Genes.
Gastroenterology ( IF 29.4 ) Pub Date : 2019-12-17 , DOI: 10.1053/j.gastro.2019.11.302 Pengyu Liu 1 , Binyong Liang 2 , Menggang Liu 3 , Joyce H G Lebbink 4 , Shan Li 1 , Manning Qian 5 , Marla Lavrijsen 1 , Maikel P Peppelenbosch 1 , Xin Chen 6 , Ron Smits 1
Gastroenterology ( IF 29.4 ) Pub Date : 2019-12-17 , DOI: 10.1053/j.gastro.2019.11.302 Pengyu Liu 1 , Binyong Liang 2 , Menggang Liu 3 , Joyce H G Lebbink 4 , Shan Li 1 , Manning Qian 5 , Marla Lavrijsen 1 , Maikel P Peppelenbosch 1 , Xin Chen 6 , Ron Smits 1
Affiliation
|
BACKGROUND & AIMS
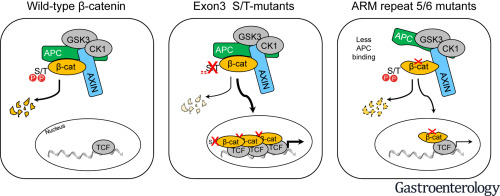
The β-catenin signaling pathway is one of the most commonly deregulated pathways in cancer cells. Amino acid substitutions within armadillo repeats 5 and 6 (K335, W383, and N387) of β-catenin are found in several tumor types, including liver tumors. We investigated the mechanisms by which these substitutions increase signaling and the effects on liver carcinogenesis in mice.
METHODS
Plasmids encoding tagged full-length β-catenin (CTNNB1) or β-catenin with the K335I or N387K substitutions, along with MET, were injected into tails of FVB/N mice. Tumor growth was monitored, and livers were collected and analyzed by histology, immunohistochemistry, and quantitative reverse-transcription polymerase chain reaction. Tagged full-length and mutant forms of β-catenin were expressed in HEK293, HCT116, and SNU449 cells, which were analyzed by immunoblots and immunoprecipitation. A panel of β-catenin variants and cell lines with knock-in mutations were analyzed for differences in N-terminal phosphorylation, half-life, and association with other proteins in the signaling pathway.
RESULTS
Mice injected with plasmids encoding K335I or N387K β-catenin and MET developed larger, more advanced tumors than mice injected with plasmids encoding WT β-catenin and MET. K335I and N387K β-catenin bound APC with lower affinity than WT β-catenin but still interacted with scaffold protein AXIN1 and in the nucleus with TCF7L2. This interaction resulted in increased transcription of genes regulated by β-catenin. Studies of protein structures supported the observed changes in relative binding affinities.
CONCLUSION
Expression of β-catenin with mutations in armadillo repeats 5 and 6, along with MET, promotes formation of liver tumors in mice. In contrast to N-terminal mutations in β-catenin that directly impair its phosphorylation by GSK3 or binding to BTRC, the K335I or N387K substitutions increase signaling via reduced binding to APC. However, these mutant forms of β-catenin still interact with the TCF family of transcription factors in the nucleus. These findings show how these amino acid substitutions increase β-catenin signaling in cancer cells.
中文翻译:

犰狳重复序列5和6中的致癌突变会降低与APC的结合,从而增加信号传导和目标基因的转录。
背景与目的β-catenin信号传导途径是癌细胞中最常见的失控途径之一。β-catenin的犰狳重复序列5和6(K335,W383和N387)中的氨基酸取代存在于多种肿瘤类型中,包括肝肿瘤。我们研究了这些取代增加信号传导的机制以及对小鼠肝癌发生的影响。方法将编码带有K335I或N387K取代标记的全长β-catenin(CTNNB1)或β-catenin的质粒与MET一起注射到FVB / N小鼠的尾巴中。监测肿瘤的生长,并通过组织学,免疫组织化学和定量逆转录聚合酶链反应对肝脏进行收集和分析。在HEK293,HCT116和SNU449细胞中表达了标记的β-catenin全长和突变形式。通过免疫印迹和免疫沉淀法对其进行了分析。分析了一组带有敲入突变的β-catenin变体和细胞系的N末端磷酸化,半衰期以及与信号传导途径中其他蛋白质的缔合差异。结果注射了编码K335I或N387Kβ-catenin和MET的质粒的小鼠比注射了WTβ-catenin和MET的质粒的小鼠出现了更大,更晚期的肿瘤。K335I和N387Kβ-catenin以比WTβ-catenin低的亲和力与APC结合,但仍与支架蛋白AXIN1和TCF7L2相互作用。这种相互作用导致受β-连环蛋白调节的基因转录增加。蛋白质结构的研究支持了观察到的相对结合亲和力的变化。结论犰狳重复序列5和6中具有突变的β-catenin的表达 与MET一起,可促进小鼠肝肿瘤的形成。与β-catenin中的N端突变直接通过GSK3破坏其磷酸化或与BTRC的结合相反,K335I或N387K取代通过减少与APC的结合而增加了信号传导。但是,这些β-catenin的突变形式仍然与细胞核中TCF家族的转录因子相互作用。这些发现表明这些氨基酸取代如何增加癌细胞中的β-catenin信号传导。
更新日期:2019-12-18
中文翻译:

犰狳重复序列5和6中的致癌突变会降低与APC的结合,从而增加信号传导和目标基因的转录。
背景与目的β-catenin信号传导途径是癌细胞中最常见的失控途径之一。β-catenin的犰狳重复序列5和6(K335,W383和N387)中的氨基酸取代存在于多种肿瘤类型中,包括肝肿瘤。我们研究了这些取代增加信号传导的机制以及对小鼠肝癌发生的影响。方法将编码带有K335I或N387K取代标记的全长β-catenin(CTNNB1)或β-catenin的质粒与MET一起注射到FVB / N小鼠的尾巴中。监测肿瘤的生长,并通过组织学,免疫组织化学和定量逆转录聚合酶链反应对肝脏进行收集和分析。在HEK293,HCT116和SNU449细胞中表达了标记的β-catenin全长和突变形式。通过免疫印迹和免疫沉淀法对其进行了分析。分析了一组带有敲入突变的β-catenin变体和细胞系的N末端磷酸化,半衰期以及与信号传导途径中其他蛋白质的缔合差异。结果注射了编码K335I或N387Kβ-catenin和MET的质粒的小鼠比注射了WTβ-catenin和MET的质粒的小鼠出现了更大,更晚期的肿瘤。K335I和N387Kβ-catenin以比WTβ-catenin低的亲和力与APC结合,但仍与支架蛋白AXIN1和TCF7L2相互作用。这种相互作用导致受β-连环蛋白调节的基因转录增加。蛋白质结构的研究支持了观察到的相对结合亲和力的变化。结论犰狳重复序列5和6中具有突变的β-catenin的表达 与MET一起,可促进小鼠肝肿瘤的形成。与β-catenin中的N端突变直接通过GSK3破坏其磷酸化或与BTRC的结合相反,K335I或N387K取代通过减少与APC的结合而增加了信号传导。但是,这些β-catenin的突变形式仍然与细胞核中TCF家族的转录因子相互作用。这些发现表明这些氨基酸取代如何增加癌细胞中的β-catenin信号传导。

























 京公网安备 11010802027423号
京公网安备 11010802027423号