Electrochemistry Communications ( IF 5.4 ) Pub Date : 2019-11-26 , DOI: 10.1016/j.elecom.2019.106617 Baskar Senthilkumar , Ramakrishnan Kalai Selvan , Prabeer Barpanda
|
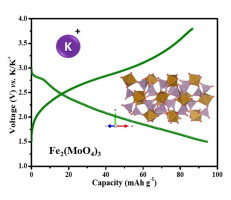
Reversible potassium-ion intercalation in anti-NASICON-type iron molybdate Fe2(MoO4)3 is demonstrated for the first time. Submicron-sized Fe2(MoO4)3 particles are synthesized by solution combustion synthesis route using glycine as fuel. The monoclinic anti-NASICON type Fe2(MoO4)3 positive electrode exhibits an initial discharge capacity of ∼94 mAh g-1 involving an Fe3+/Fe2+ redox activity centered around 2.6 V (vs. K/K+). Employing ex situ X-ray diffraction, the K+ (de)intercalation is found to involve an underlying solid-solution redox mechanism. This anti-NASICON Fe2(MoO4)3 molybdate is found to be an efficient polyanionic cathode candidate for potassium-ion batteries.
中文翻译:

抗NASICON型钼酸铁Fe 2(MoO 4)3中的钾离子嵌入
首次证明了抗NASICON型钼酸铁Fe 2(MoO 4)3中可逆的钾离子嵌入。以甘氨酸为燃料,通过固溶燃烧合成路线合成了亚微米级的Fe 2(MoO 4)3颗粒。单斜晶抗NASICON型Fe 2(MoO 4)3正极具有约94 mAh g -1的初始放电容量,涉及以2.6 V(vs. K / K +)为中心的Fe 3+ / Fe 2+氧化还原活性。。利用异位X射线衍射,K +发现(脱)插层涉及潜在的固溶氧化还原机理。发现该抗NASICON Fe 2(MoO 4)3钼酸盐是钾离子电池的有效聚阴离子阴极候选物。


























 京公网安备 11010802027423号
京公网安备 11010802027423号