Computational and Theoretical Chemistry ( IF 2.8 ) Pub Date : 2019-11-20 , DOI: 10.1016/j.comptc.2019.112661 Zan Lyu , Geniece Hallett-Tapley , Galina Orlova
|
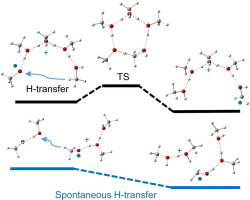
Radical-cationic methanol clusters (CH3OH) +•n, n=2-5, are studied in the gas phase and solvent methanol using hybrid B3LYP, CAM-B3LYP and BHandHLYP exchange-correlation functionals. The CAM-B3LYP predictions are the most consistent. Ionization of parental neutral rings and chains yields distonic radical cations with spin residing on the CH3O• link. Higher energy O-O hemibonded structures are also located. The intra-cluster H-transfer from the methyl group of the CH3OH-terminus of the protonated moiety to the O-atom of the CH3O• terminus in chain radical cations leads to the lowest-energy distonic isomers with the CH2OH• terminus. The CAM-B3LYP activation enthalpies for this reaction in the gas phase are 9.5, 11.1, 13.7, and 17.1 kcal/mol for n=5, 4, 3, 2, respectively. The inter-cluster H-transfer from the CH3O• terminus to a neutral methanol cluster occurs spontaneously, with formation of a protonated cluster weakly bound to the radical cluster. The protonated cluster may serve as a proton carrier.
中文翻译:

自由基阳离子甲醇团簇的DFT研究:结构,键合和H转移
利用杂化B3LYP,CAM-B3LYP和BHandHLYP交换相关功能,研究了气相和溶剂甲醇中的自由基阳离子甲醇簇(CH 3 OH)+• n,n = 2-5。CAM-B3LYP预测最一致。父母中性环和链的电离会产生自旋位于CH 3 O •链上的异位自由基阳离子。还定位了较高能量的OO半键合结构。从质子化部分的CH 3 OH末端的甲基到链自由基阳离子中的CH 3 O •末端的O原子发生簇内H转移,导致CH 2的最低能量二元异构体OH •终点。对于n = 5、4、3、2,在气相中该反应的CAM-B3LYP活化焓分别为9.5、11.1、13.7和17.1kcal / mol。从CH 3 O •末端到中性甲醇簇的簇间H转移是自发发生的,形成了与自由基簇弱结合的质子化簇。质子化簇可以用作质子载体。



























 京公网安备 11010802027423号
京公网安备 11010802027423号