Computational and Theoretical Chemistry ( IF 2.8 ) Pub Date : 2019-11-17 , DOI: 10.1016/j.comptc.2019.112660 Xiufang Hou , Feng Fu , Chuan Bai , Guoming Lei
|
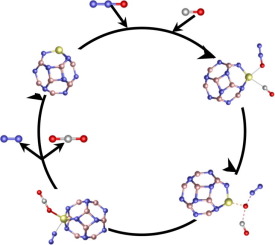
In this study, CO oxidation by N2O, catalyzed by pristine B12N12+ and B11N12M+ (M = Al, Ga, and In) nanocages doped with Al, Ga, and In were investigated using density functional theory. The study investigated the potential of utilizing the four B11N12M+ (M = B, Al, Ga, and In) nanocages as an efficient catalyst for the CO–N2O reaction. The reaction proceeds in one step reaction mechanism rather than the traditional two. In other words, the coadsorption of N2O and CO at B11N12M+ (M = B, Al, Ga, and In) nanocages followed the transition state and formation of CO2 and N2 species. The activation energy for the reaction was 78.6, −32.6, −2.5, and −7.4 kJ/mol when catalyzed by B11N12M+ for M = B, Al, Ga, and In, respectively, with respect to the reactants. Obviously, B11N12Al+ is the best catalyst for the reaction. The B11N12M+ configurations, density-of-states (DOS), and reaction mechanism were obtained to explain why.
中文翻译:

氮化硼B 11 N 12 M +(M = B,Al,Ga和In)纳米笼作为N 2 O氧化CO的催化剂:密度泛函研究
在这项研究中,使用密度泛函研究了由原始B 12 N 12 +和B 11 N 12 M +(M = Al,Ga和In)掺杂Al,Ga和In的纳米笼催化的N 2 O氧化CO。理论。该研究调查了利用四种B 11 N 12 M +(M = B,Al,Ga和In)纳米笼作为CO–N 2 O反应的有效催化剂的潜力。该反应以一步反应机理进行,而不是传统的两步反应机理。换句话说,N 2 O和CO在B 11 N 12 M +上的共吸附(M = B,Al,Ga和In)纳米笼子遵循过渡态以及CO 2和N 2物种的形成。当由B 11 N 12 M +分别针对反应物的M = B,Al,Ga和In催化时,该反应的活化能为78.6,-32.6,-2.5和-7.4 kJ / mol 。显然,B 11 N 12 Al +是反应的最佳催化剂。获得了B 11 N 12 M +构型,态密度(DOS)和反应机理来解释其原因。

























 京公网安备 11010802027423号
京公网安备 11010802027423号