当前位置:
X-MOL 学术
›
Faraday Discuss.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
FBpin and its adducts and their role in catalytic borylations.
Faraday Discussions ( IF 3.4 ) Pub Date : 2019-12-02 , DOI: 10.1039/c9fd00053d Laura Kuehn 1 , Martin Stang , Sabrina Würtemberger-Pietsch , Alexandra Friedrich , Heidi Schneider , Udo Radius , Todd B Marder
Faraday Discussions ( IF 3.4 ) Pub Date : 2019-12-02 , DOI: 10.1039/c9fd00053d Laura Kuehn 1 , Martin Stang , Sabrina Würtemberger-Pietsch , Alexandra Friedrich , Heidi Schneider , Udo Radius , Todd B Marder
Affiliation
|
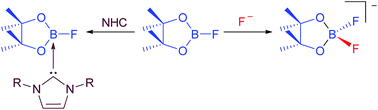
We present herein investigations concerning the reactivity of B2pin2 and FBpin (pin = pinacolato) with respect to fluoride ions and NHCs (NHC = N-Heterocyclic Carbene) in order to understand better the role of added fluoride in our recently reported Ni-catalyzed C-F borylation process, to confirm the nature of the NHC-adducts of FBpin, and to explain the formation of the species detected as side products of the defluoroborylation reaction. We report the calculated gas phase fluoride ion affinities (FIA) of relevant boron species and demonstrate that the presence of one equivalent of added F- in solution will bind and activate B2pin2 (FIA = +247 kJ mol-1), but it will also bind even more effectively to FBpin (FIA = +287 kJ mol-1) formed in the reaction. We prepared the NHC-adducts of FBpin by two different methods and isolated and characterized several examples of the type FBpin·NHC (NHC = iPr2Im, 1; Me2Im, 2; MeiPrIm, 3; nPr2Im, 4; Mes2Im 5). The salts [pinBF2][NMe4] 6, as well as the significantly more soluble analogue [pinBF2][NnBu4] 7, were also isolated and characterized, providing confirmation of the presence of the pinBF2 anion in various stoichiometric and catalytic borylation reactions.
中文翻译:

FBpin及其加合物及其在催化硼化反应中的作用。
我们在此介绍有关B2pin2和FBpin(pin =频哪醇)对氟离子和NHCs(NHC = N-杂环卡宾)的反应性的研究,以便更好地了解添加的氟化物在我们最近报道的Ni催化的CF硼化中的作用。过程中,确认FBpin的NHC加合物的性质,并解释检测到的作为脱氟硼化反应副产物的物种的形成。我们报告了相关硼物种的气相氟离子亲和力(FIA)计算值,并证明了溶液中一当量添加的F-的存在会结合并激活B2pin2(FIA = +247 kJ mol-1),但它也会甚至更有效地与反应中形成的FBpin(FIA = +287 kJ mol-1)结合。我们通过两种不同的方法制备了FBpin的NHC加合物,并分离并表征了FBpin·NHC类型的几个实例(NHC = iPr2Im,1; Me2Im,2; MeiPrIm,3; nPr2Im,4; Mes2Im 5)。还分离并表征了盐[pinBF2] [NMe4] 6以及明显更易溶的类似物[pinBF2] [NnBu4] 7,从而确认了在各种化学计量和催化硼化反应中是否存在pinBF2阴离子。
更新日期:2019-12-04
中文翻译:

FBpin及其加合物及其在催化硼化反应中的作用。
我们在此介绍有关B2pin2和FBpin(pin =频哪醇)对氟离子和NHCs(NHC = N-杂环卡宾)的反应性的研究,以便更好地了解添加的氟化物在我们最近报道的Ni催化的CF硼化中的作用。过程中,确认FBpin的NHC加合物的性质,并解释检测到的作为脱氟硼化反应副产物的物种的形成。我们报告了相关硼物种的气相氟离子亲和力(FIA)计算值,并证明了溶液中一当量添加的F-的存在会结合并激活B2pin2(FIA = +247 kJ mol-1),但它也会甚至更有效地与反应中形成的FBpin(FIA = +287 kJ mol-1)结合。我们通过两种不同的方法制备了FBpin的NHC加合物,并分离并表征了FBpin·NHC类型的几个实例(NHC = iPr2Im,1; Me2Im,2; MeiPrIm,3; nPr2Im,4; Mes2Im 5)。还分离并表征了盐[pinBF2] [NMe4] 6以及明显更易溶的类似物[pinBF2] [NnBu4] 7,从而确认了在各种化学计量和催化硼化反应中是否存在pinBF2阴离子。


























 京公网安备 11010802027423号
京公网安备 11010802027423号