Phytomedicine ( IF 7.9 ) Pub Date : 2023-05-13 , DOI: 10.1016/j.phymed.2023.154878 Jiamin Li 1 , Junzhen Zhou 1 , Na Zhao 2 , Zhendong Li 3 , Xinwen Xu 1 , Jingjing Tang 1 , Ziyu Li 2 , Xiaoying Zhang 4 , Yuerui Wu 5 , Qiang Li 3 , Qing Zhang 1 , Jianwei Jiang 2
|
Background
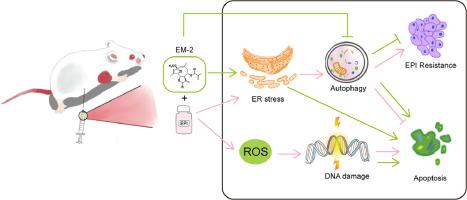
EM-2, a natural sesquiterpene lactone isolated from Elephantopus mollis H.B.K., showed a good anti-breast cancer effect when combined with epirubicin (EPI). However, its synergistic sensitization mechanism remains unclear.
Purpose
This study aimed to determine the therapeutic effect and possible synergistic mechanism of EM-2 with EPI in vivo and in vitro and to provide an experimental basis for the treatment of human breast cancer.
Methods
Cell proliferation was measured with MTT and colony formation assays. Apoptosis and reactive oxygen species (ROS) levels were examined through flow cytometry, and the expression levels of proteins related to apoptosis, autophagy, endoplasmic reticulum stress, and DNA damage were detected through Western blot analysis. Moreover, the caspase inhibitor Z-VAD-FMK, autophagy inhibitors bafilomycin A1 and chloroquine, ER stress inhibitor 4-phenylbutyric acid, and ROS scavenger N-acetyl cysteine were applied to verify signaling pathways. Breast cancer cell lines were used to evaluate the antitumor functions of EM-2 and EPI in vitro and in vivo.
Results
We demonstrated that in MDA-MB-231 and SKBR3 cells, the IC50 of EPI combined with EM-2 (IC20) was 37.909 and 33.889 times lower than that of EPI alone, respectively. Further study verified that in EPI-resistant lines (MDA-MB-231/EPI), the IC50 of EPI combined with EM-2 (IC20) was 26.305 times lower than that of EPI alone. Mechanistically, EM-2 could reverse the protective effect of EPI against autophagy in SKBR3 and MDA-MB-231 cells. EM-2 and EPI could trigger ER stress. When EM-2 and EPI were used in combination, ER stress was continuously activated, and ER stress-mediated apoptosis was induced. Meanwhile, EM-2 combined with EPI promoted DNA damage then induced apoptosis. In vivo, the volume of breast cancer xenografts in the combination group was smaller than that in the control, EM-2, and EPI groups. Immunohistochemical experiments demonstrated that the combination of EM-2 and EPI could block autophagy and promote ER stress in vivo.
Conclusion
EM-2 enhances the sensitivity of MDA-MB-231, SKBR3, and EPI-resistant cells to EPI.
中文翻译:

EM-2 是一种来自 Elephantopus mollis HBK 的天然倍半萜内酯,通过阻断保护性自噬增强乳腺癌细胞对表柔比星的敏感性
背景
EM-2 是一种从Elephantopus mollis HBK中分离出来的天然倍半萜内酯,与表柔比星 (EPI) 联合使用时显示出良好的抗乳腺癌作用。然而,其协同致敏机制仍不清楚。
目的
本研究旨在探讨EM-2与EPI在体内外的治疗作用及可能的协同作用机制,为人类乳腺癌的治疗提供实验依据。
方法
用MTT和集落形成测定法测量细胞增殖。通过流式细胞术检测细胞凋亡和活性氧(ROS)水平,通过Western blot分析检测细胞凋亡、自噬、内质网应激和DNA损伤相关蛋白的表达水平。此外,应用半胱天冬酶抑制剂 Z-VAD-FMK、自噬抑制剂巴弗洛霉素 A1 和氯喹、内质网应激抑制剂 4-苯基丁酸和 ROS 清除剂N-乙酰半胱氨酸来验证信号通路。乳腺癌细胞系用于评估 EM-2 和 EPI 的体外和体内抗肿瘤功能。
结果
我们证明,在 MDA-MB-231 和 SKBR3 细胞中, EPI 联合 EM-2 的IC 50 (IC 20 ) 分别比单独的 EPI 低 37.909 和 33.889 倍。进一步研究证实,在EPI抗性株系(MDA-MB-231/EPI)中, EPI联合EM-2的IC 50 (IC 20 )比单独EPI低26.305倍。从机制上讲,EM-2 可以逆转 EPI 对 SKBR3 和 MDA-MB-231 细胞自噬的保护作用。EM-2 和 EPI 可以触发 ER 压力。当EM-2和EPI联合使用时,内质网应激持续激活,诱导内质网应激介导的细胞凋亡。同时,EM-2联合EPI促进DNA损伤,诱导细胞凋亡。体内,联合组乳腺癌异种移植物体积小于对照组、EM-2、EPI组。免疫组织化学实验表明,EM-2 和 EPI 的结合可以阻断自噬并促进体内ER 应激。
结论
EM-2 增强 MDA-MB-231、SKBR3 和 EPI 抗性细胞对 EPI 的敏感性。

























 京公网安备 11010802027423号
京公网安备 11010802027423号