当前位置:
X-MOL 学术
›
Org. Chem. Front.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
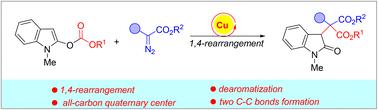
Copper-catalyzed dearomative 1,4-carboxylate rearrangement of 2-carbonateindoles
Organic Chemistry Frontiers ( IF 5.4 ) Pub Date : 2022-11-14 , DOI: 10.1039/d2qo01647h Yan Zhu 1 , Ying Shao 1 , Shengbiao Tang 1 , Jiangtao Sun 1
Organic Chemistry Frontiers ( IF 5.4 ) Pub Date : 2022-11-14 , DOI: 10.1039/d2qo01647h Yan Zhu 1 , Ying Shao 1 , Shengbiao Tang 1 , Jiangtao Sun 1
Affiliation
|
A novel dearomative 1,4-rearrangement reaction of 2-carbonateindoles with aryl diazoacetates has been developed in the presence of a copper catalyst, which might proceed through a tandem formation of a zwitterionic intermediate, intramolecular cyclization and ring-opening reaction to give the final rearrangement products containing an all-carbon quaternary center with two newly formed C–C bonds. In this sequence, the disruption of the aromaticity of indole has been realized, associated with 1,4-carboxylate rearrangements.
中文翻译:

铜催化的 2-碳酸酯吲哚的脱芳烃 1,4-羧酸重排
在铜催化剂存在下,开发了一种新型的 2-碳酸酯吲哚与芳基重氮乙酸酯的脱芳烃 1,4-重排反应,该反应可能通过串联形成两性离子中间体、分子内环化和开环反应得到最终产物含有全碳季中心和两个新形成的 C-C 键的重排产物。在这个序列中,已经实现了吲哚芳香性的破坏,与 1,4-羧酸重排相关。
更新日期:2022-11-14
中文翻译:

铜催化的 2-碳酸酯吲哚的脱芳烃 1,4-羧酸重排
在铜催化剂存在下,开发了一种新型的 2-碳酸酯吲哚与芳基重氮乙酸酯的脱芳烃 1,4-重排反应,该反应可能通过串联形成两性离子中间体、分子内环化和开环反应得到最终产物含有全碳季中心和两个新形成的 C-C 键的重排产物。在这个序列中,已经实现了吲哚芳香性的破坏,与 1,4-羧酸重排相关。



























 京公网安备 11010802027423号
京公网安备 11010802027423号