当前位置:
X-MOL 学术
›
Angew. Chem. Int. Ed.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
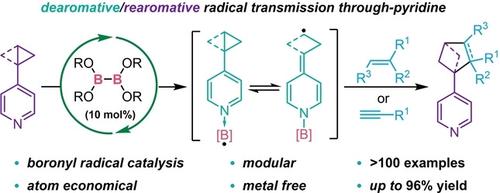
Diboron(4)-Catalyzed Remote [3+2] Cycloaddition of Cyclopropanes via Dearomative/Rearomative Radical Transmission through Pyridine
Angewandte Chemie International Edition ( IF 16.6 ) Pub Date : 2022-11-07 , DOI: 10.1002/anie.202214507 Ming Xu 1 , Zhijun Wang 2 , Zhaohui Sun 1 , Yizhao Ouyang 1 , Zhengwei Ding 3 , Tao Yu 1 , Liang Xu 2 , Pengfei Li 1, 4
Angewandte Chemie International Edition ( IF 16.6 ) Pub Date : 2022-11-07 , DOI: 10.1002/anie.202214507 Ming Xu 1 , Zhijun Wang 2 , Zhaohui Sun 1 , Yizhao Ouyang 1 , Zhengwei Ding 3 , Tao Yu 1 , Liang Xu 2 , Pengfei Li 1, 4
Affiliation
|
Previously challenging pyridinyl cyclopentanes, cyclopentenes and bicyclo[2.1.1]hexanes with multiple substituents now can be efficiently prepared via a remote [3+2] cycloaddition reaction. The reaction features a simple diboron(4) catalyst, modular disconnection mode, broad scope, high atom economy and a unique dearomative/rearomative mechanistic process.
中文翻译:

二硼 (4)-催化远程 [3+2] 环丙烷通过吡啶脱芳/再芳香自由基传递进行环加成
以前具有挑战性的具有多个取代基的吡啶基环戊烷、环戊烯和双环 [2.1.1] 己烷现在可以通过远程 [3+2] 环加成反应有效地制备。该反应具有简单的二硼 (4) 催化剂、模块化断开模式、广泛的范围、高原子经济性和独特的脱芳烃/重芳烃机理过程。
更新日期:2022-11-07
中文翻译:

二硼 (4)-催化远程 [3+2] 环丙烷通过吡啶脱芳/再芳香自由基传递进行环加成
以前具有挑战性的具有多个取代基的吡啶基环戊烷、环戊烯和双环 [2.1.1] 己烷现在可以通过远程 [3+2] 环加成反应有效地制备。该反应具有简单的二硼 (4) 催化剂、模块化断开模式、广泛的范围、高原子经济性和独特的脱芳烃/重芳烃机理过程。



























 京公网安备 11010802027423号
京公网安备 11010802027423号