Journal of the American College of Cardiology ( IF 24.0 ) Pub Date : 2022-07-25 , DOI: 10.1016/j.jacc.2022.04.062 Srinivas R Dukkipati 1 , David R Holmes 2 , Shephal K Doshi 3 , Saibal Kar 4 , Sheldon M Singh 5 , Douglas Gibson 6 , Matthew J Price 6 , Andrea Natale 7 , Moussa Mansour 8 , Horst Sievert 9 , Vicki M Houle 10 , Dominic J Allocco 10 , Vivek Y Reddy 1
|
Background
In the U.S. Food and Drug Administration (FDA) clinical trials of left atrial appendage (LAA) closure, a postimplantation peridevice leak (PDL) of ≤5 mm (PDL≤5) was accepted as sufficient LAA “closure.” However, the clinical consequences of these PDLs on subsequent thromboembolism are poorly characterized.
Objectives
We sought to assess the impact of PDL≤5 on clinical outcomes after implantation of the Watchman device.
Methods
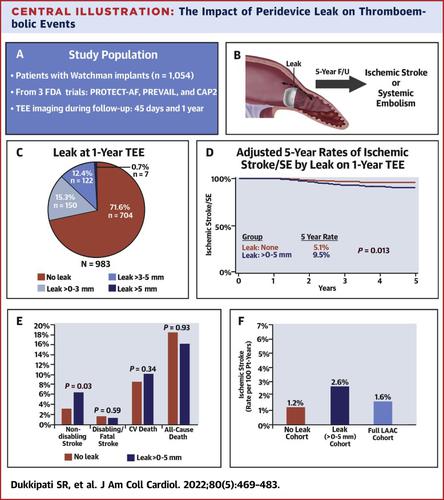
Using combined data from the FDA studies PROTECT-AF (Watchman Left Atrial Appendage System for Embolic Protection in Patients With Atrial Fibrillation), PREVAIL (Evaluation of the Watchman Left Atrial Appendage Closure Device in Patients With Atrial Fibrillation vs Long Term Warfarin Therapy), and CAP2 (Continued Access to PREVAIL), we assessed patients with successful device implantation for PDL by means of protocol-mandated transesophageal echocardiograms (TEEs) at 45 days and 1 year. Five-year outcomes were assessed as a function of the absence or presence of PDL≤5.
Results
The cohort included 1,054 patients: mean age 74 ± 8.3 years, 65% male, and CHA2DS2-VASc 4.1 ± 1.4. TEE imaging at 45 days revealed 634 patients (60.2%) without and 404 (38.3%) with PDL≤5, and 1-year TEE revealed 704 patients (71.6%) without and 272 (27.7%) with PDL≤5. The presence of PDL≤5 at 1 year, but not at 45 days, was associated with an increased 5-year risk of ischemic stroke or systemic embolism (adjusted HR: 1.94; 95% CI: 1.15-3.29; P = 0.014), largely driven by an increase in nondisabling stroke (HR: 1.97; 95% CI: 1.03-3.78; P = 0.04), while disabling or fatal stroke rates were similar (HR: 0.69; 95% CI: 0.19-2.46; P = 0.56). PDL≤5 was not associated with an increased risk of cardiovascular or unexplained death (HR: 1.20; P = 0.45) or all-cause death (HR: 0.87; P = 0.42).
Conclusions
PDL≤5 at 1 year after percutaneous LAA closure with the Watchman device are associated with increased thromboembolism, driven by increased nondisabling stroke, but similar mortality. (Watchman Left Atrial Appendage System for Embolic Protection in Patients With Atrial Fibrillation [PROTECT-AF; NCT00129545]; Evaluation of the Watchman Left Atrial Appendage Closure Device in Patients With Atrial Fibrillation vs Long Term Warfarin Therapy [PREVAIL; NCT01182441]; Continued Access to PREVAIL [CAP2; NCT01760291])
中文翻译:

Peridevice 泄漏对左心耳封堵术后 5 年结果的影响
背景
在美国食品和药物管理局 (FDA) 的左心耳 (LAA) 闭合临床试验中,植入后装置周围泄漏 (PDL) ≤ 5 毫米 (PDL≤5) 被认为是足够的左心耳“闭合”。然而,这些 PDL 对随后的血栓栓塞的临床后果的表征很差。
目标
我们试图评估植入 Watchman 装置后 PDL≤5 对临床结果的影响。
方法
使用来自 FDA 研究的综合数据 PROTECT-AF(用于心房颤动患者栓塞保护的 Watchman 左心耳系统)、PREVAIL(心房颤动患者 Watchman 左心耳闭合装置与长期华法林治疗的对比评估)和CAP2(继续访问 PREVAIL),我们在 45 天和 1 年时通过协议规定的经食管超声心动图 (TEE) 评估了成功植入 PDL 装置的患者。五年结果评估为是否存在 PDL≤5 的函数。
结果
该队列包括 1,054 名患者:平均年龄 74 ± 8.3 岁,65% 为男性,CHA 2 DS 2 -VASc 4.1 ± 1.4。45 天时的 TEE 成像显示 634 名患者 (60.2%) 没有 PDL≤5,404 名患者 (38.3%) 有 PDL≤5,1 年 TEE 显示 704 名患者 (71.6%) 没有 PDL≤5,272 名患者 (27.7%) PDL≤5。1 年时 PDL≤5,但 45 天时不存在,与 5 年缺血性卒中或全身性栓塞风险增加相关(调整后的 HR:1.94;95% CI:1.15-3.29;P = 0.014 ) ,主要受非致残性卒中增加的驱动(HR:1.97;95% CI:1.03-3.78;P = 0.04),而致残性或致死性卒中发生率相似(HR:0.69;95% CI:0.19-2.46;P = 0.56)。PDL≤5 与心血管或不明原因死亡(HR:1.20; P = 0.45)或全因死亡(HR:0.87;P = 0.42)风险增加无关 。
结论
使用 Watchman 装置经皮 LAA 闭合术后 1 年 PDL≤5 与血栓栓塞增加相关,由非致残性卒中增加驱动,但死亡率相似。(用于心房颤动患者栓塞保护的 Watchman 左心耳系统 [PROTECT-AF;NCT00129545];心房颤动患者 Watchman 左心耳闭合装置与长期华法林治疗的对比评估 [PREVAIL;NCT01182441];继续访问优先 [CAP2; NCT01760291])

























 京公网安备 11010802027423号
京公网安备 11010802027423号