Chemical Engineering Journal ( IF 15.1 ) Pub Date : 2022-07-22 , DOI: 10.1016/j.cej.2022.138239 Jiabai Cai , Huan Li , Kai Feng , Yingchao Cheng , Shuting He , Masaki Takaoka
|
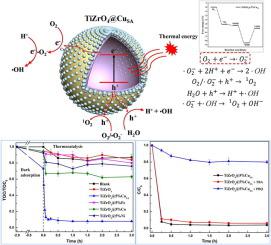
Humic acid (HA), a refractory organic pollutant, has always been a key obstackle in wastewater treatment (such as biochemical landfill leachate). And catalytic air oxidation is one of the most promising methods to degrade HA efficiently and economically. In this study, a novel high-loading Cu single-atom catalyst (TiZrO4@CuSA) was facilely synthesized using isovolume vacuum impregnation and co-calcination method, and then used to accelerate the oxidation of HA at low temperatures. The TiZrO4@5%CuSA catalytic system showed a superior performance, and 91 % of HA was degraded and 90 % of organic carbon was removed from HA solution within 10 min at 90 °C. Aberration-corrected electron microscopy, electron paramagnetic resonance spectroscopy, and density functional theory calculations were applied to reveal the catalytic mechanism of TiZrO 4@5%CuSA. The active center of Cu single-atom co-catalyst with Cu − O coordination sites can quickly transfer electrons to O2 in the water to form O2.−, exhibiting high activation efficiency. Gas chromatography (GC), liquid chromatograph-mass spectrometer (LC-MS), liquid chromatography-organic carbon detection (LC-OCD), and Fourier-transform infrared spectroscopy (FT-IR) spectroscopy provided the results of intermediate products, including 3-phenoxy-benzaldehy, phenylglyoxal, ethanol, acetic acid, butyric acid, and valeric acid, and thus a catalytic degradation pathway of HA was speculated. Based on the new catalyst, a new process can be conceived for efficient and economic removal of HA from HA-containing wastewater.
中文翻译:

钛锆氧化物@铜单原子活化氧低温降解腐植酸:机理与途径
腐植酸(HA)是一种难降解的有机污染物,一直是废水处理(如生化垃圾渗滤液)的关键障碍。催化空气氧化是高效、经济地降解 HA 的最有前景的方法之一。本研究采用等容真空浸渍共煅法简便合成了一种新型高负载Cu单原子催化剂(TiZrO 4 @Cu SA),并用于加速HA的低温氧化。TiZrO 4 @5%Cu SA催化体系表现出优异的性能,在 90°C 下 10 分钟内,91% 的 HA 被降解,90% 的有机碳从 HA 溶液中去除。应用像差校正电子显微镜、电子顺磁共振光谱和密度泛函理论计算来揭示TiZrO 4 @5%Cu SA的催化机理。具有Cu-O配位点的Cu单原子助催化剂的活性中心可以快速将电子转移到水中的O 2 上形成O 2 。,表现出很高的活化效率。气相色谱(GC)、液相色谱-质谱(LC-MS)、液相色谱-有机碳检测(LC-OCD)和傅里叶变换红外光谱(FT-IR)光谱提供了中间产物的结果,包括3 -苯氧基-苯甲醛、苯乙二醛、乙醇、乙酸、丁酸和戊酸,从而推测了HA的催化降解途径。基于新催化剂,可以构思一种新工艺,用于从含 HA 的废水中高效、经济地去除 HA。



























 京公网安备 11010802027423号
京公网安备 11010802027423号