当前位置:
X-MOL 学术
›
Green Synth. Catal.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
TMSCF2Br as both a C1 synthon and a F1 reagent
Green Synthesis and Catalysis Pub Date : 2022-02-16 , DOI: 10.1016/j.gresc.2022.02.001 Danhua Ge , Zhenhua Jia , Xue-Qiang Chu
中文翻译:

TMSCF2Br 作为 C1 合成子和 F1 试剂
更新日期:2022-02-16
Green Synthesis and Catalysis Pub Date : 2022-02-16 , DOI: 10.1016/j.gresc.2022.02.001 Danhua Ge , Zhenhua Jia , Xue-Qiang Chu
|
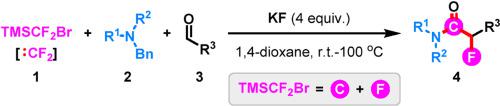
This highlight summarizes a recent development of KF-promoted three-component fluorination-aminocarbonylation reaction of aldehydes, tertiary amines, and TMSCF2Br for the synthesis of structurally important α-fluoroamide derivatives. The key to the success of this method could be attributed to the multiple functions of difluorocarbene precursor (TMSCF2Br) as both a C1 synthon and a F1 reagent through the process of C–F bond cleavage and C–F bond re-formation.
中文翻译:

TMSCF2Br 作为 C1 合成子和 F1 试剂
这一亮点总结了 KF 促进的醛、叔胺和 TMSCF 2 Br的三组分氟化-氨基羰基化反应的最新发展,用于合成结构上重要的α-氟酰胺衍生物。该方法成功的关键可归因于二氟卡宾前体 (TMSCF 2 Br) 通过 C-F 键断裂和 C-F 键重新形成过程作为 C1 合成子和 F1 试剂的多重功能。



























 京公网安备 11010802027423号
京公网安备 11010802027423号