Applied Catalysis B: Environment and Energy ( IF 22.1 ) Pub Date : 2021-11-16 , DOI: 10.1016/j.apcatb.2021.120919 Yi Zeng 1 , Zhiyuan Tang 2 , Xingyang Wu 1 , Anhua Huang 1 , Xin Luo 2 , Guo Qin Xu 3 , Yongfa Zhu 4 , Song Ling Wang 1
|
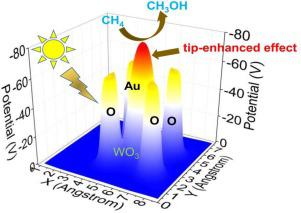
Atomic-scale metals as active center have been widely investigated for efficient photocatalysis. Understanding the specific electronic structure of atomic-scale center is of profound fundamental importance for superior catalytic performance. Here, we report an atomically dispersed gold on tungsten trioxide (Au1/WO3) catalyst for photocatalytic oxidation of methane toward value-added methanol. The Au1 species reveal a specific tip-enhanced local electrons field which favors the C-H dehydrogenation of methane and thus form methanol (up to 589 µmol g−1 h−1). Both experimental and theoretical results demonstrate such tip-enhanced effect enhance the catalytic activity of methane oxidation. The theoretical calculations further reveal a lower adsorption energy of product methanol on Au1, in contrast to Au particles, which suppresses the overoxidation of methanol, and thus promotes its selectivity. Establishing the relationship between electronic density and catalytic activity may create a platform for designing efficient atomic-scale catalysts for C1 catalysis and green chemistry.
中文翻译:

三氧化钨负载原子金室温光催化氧化甲烷制甲醇
原子级金属作为活性中心已被广泛研究用于有效的光催化。了解原子级中心的特定电子结构对于卓越的催化性能具有深远的基础意义。在这里,我们报道了一种原子分散的金在三氧化钨 (Au 1 /WO 3 ) 催化剂上,用于将甲烷光催化氧化为增值甲醇。Au 1物种揭示了一个特定的尖端增强的局部电子场,有利于甲烷的 CH 脱氢并因此形成甲醇(高达 589 µmol g -1 h -1)。实验和理论结果都表明这种尖端增强效应增强了甲烷氧化的催化活性。理论计算进一步揭示了产物甲醇在Au 1上的较低吸附能,与Au颗粒相比,其抑制了甲醇的过氧化,从而提高了其选择性。建立电子密度和催化活性之间的关系可以为设计用于 C1 催化和绿色化学的高效原子级催化剂创造一个平台。

























 京公网安备 11010802027423号
京公网安备 11010802027423号