Ecotoxicology and Environmental Safety ( IF 6.8 ) Pub Date : 2021-09-20 , DOI: 10.1016/j.ecoenv.2021.112802 Shiyao Zhang 1 , Mengyi Xu 1 , Ziyue Shen 1 , Changrui Shang 1 , Wenxiang Zhang 1 , Siyu Chen 1 , Chang Liu 2
|
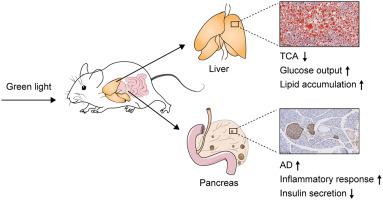
The increased incidence of metabolic syndrome (MetS) has been demonstrated to be closely associated with external environments, such as unhealthy ambient light exposure. Of note, spectral distribution of the light functions as a critical determinant of light’s pathophysiological effects. However, the effects of the lighting spectrum on metabolic homeostasis and the specific target organs remain elusive. To address this concern, we in this study high-fat diet (HFD)-fed obese mice with different spectra of the light, and divided them into white light (WL)-treated group, green light (GL)-treated group and blue light (BL)-treated group. We found that compared with BL- or WL-treated obese mice, animals exposed to GL showed worsened metabolic status, including increased body weight gain, impaired glucose tolerance/insulin sensitivity, increased levels of serum lipids, and decreased levels of serum insulin. At the organ level, GL exposure particularly exacerbated hepatic lipid accumulation and enlarged the islet volume. Taking advantages of metabolomics and transcriptomics analyses, we screened out taurocholic acid (TCA) and adenosine (AD) as two promising metabolites mediating the deleterious effects of GL on the liver and islets, respectively. In detail, GL aggravates HFD-induced lipid synthesis and gluconeogenesis in the liver via the reduction of TCA, while triggering inflammation and cellular dysfunction in islets via the induction of AD. Collectively, our findings confirmed that GL and the HFD have a synergistic effect in the induction of metabolic disorders.
Data availability
All data supported the paper are present in the paper and/or the Supplementary Materials. The original datasets are also available from the corresponding author upon request.
中文翻译:

绿光照射加重雄性小鼠高脂饮食诱导的肝脂肪变性和胰腺功能障碍
代谢综合征 (MetS) 的发病率增加已被证明与外部环境密切相关,例如不健康的环境光暴露。值得注意的是,光的光谱分布是光的病理生理效应的关键决定因素。然而,光谱对代谢稳态和特定靶器官的影响仍然难以捉摸。为了解决这个问题,我们在这项研究中用不同光谱的高脂肪饮食 (HFD) 喂养肥胖小鼠,并将它们分为白光 (WL) 治疗组、绿光 (GL) 治疗组和蓝光治疗组。光 (BL) 处理组。我们发现,与 BL 或 WL 处理的肥胖小鼠相比,暴露于 GL 的动物表现出更差的代谢状态,包括体重增加增加、葡萄糖耐量/胰岛素敏感性受损、血脂水平升高,血清胰岛素水平降低。在器官水平,GL 暴露尤其加剧了肝脏脂质积累并扩大了胰岛体积。利用代谢组学和转录组学分析,我们筛选出牛磺胆酸 (TCA) 和腺苷 (AD) 作为两种有前景的代谢物,分别介导 GL 对肝脏和胰岛的有害影响。详细地说,GL 加剧了 HFD 诱导的肝脏脂质合成和糖异生 我们筛选出牛磺胆酸 (TCA) 和腺苷 (AD) 作为两种有希望的代谢物,分别介导 GL 对肝脏和胰岛的有害影响。详细地说,GL 加剧了 HFD 诱导的肝脏脂质合成和糖异生 我们筛选出牛磺胆酸 (TCA) 和腺苷 (AD) 作为两种有希望的代谢物,分别介导 GL 对肝脏和胰岛的有害影响。详细地说,GL 加剧了 HFD 诱导的肝脏脂质合成和糖异生通过减少 TCA,同时通过诱导 AD引发胰岛炎症和细胞功能障碍。总的来说,我们的研究结果证实 GL 和 HFD 在诱导代谢紊乱方面具有协同作用。
数据可用性
支持论文的所有数据都存在于论文和/或补充材料中。原始数据集也可应要求从相应的作者处获得。


























 京公网安备 11010802027423号
京公网安备 11010802027423号