当前位置:
X-MOL 学术
›
Asian J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Ring Distortion Diversity-Oriented Approach to Fully Substituted Furoxans and Isoxazoles
Asian Journal of Organic Chemistry ( IF 2.7 ) Pub Date : 2021-08-16 , DOI: 10.1002/ajoc.202100475 Daniil A. Chaplygin 1 , Yaroslav K. Gorbunov 1 , Leonid Fershtat 2
Asian Journal of Organic Chemistry ( IF 2.7 ) Pub Date : 2021-08-16 , DOI: 10.1002/ajoc.202100475 Daniil A. Chaplygin 1 , Yaroslav K. Gorbunov 1 , Leonid Fershtat 2
Affiliation
|
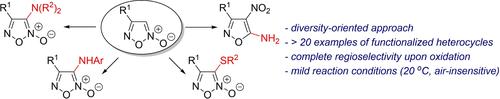
Furoxan as precursor: An operationally simple assembly of pharmacologically oriented fully substituted furoxans and isoxazoles was achieved. The presented protocol is based on a one-pot ring cleavage/nucleophilic addition/oxidation cascade of readily available monosubstituted furoxans. Synthesized disubstituted furoxans revealed an ability to release NO which indicate their strong potential as drug candidates for various biomedical applications.

中文翻译:

环畸变多样性导向方法完全取代呋喃和异恶唑
Furoxan 作为前体:实现了药理学取向的完全取代的呋喃和异恶唑的操作简单组装。所提出的协议基于一锅环裂解/亲核加成/氧化级联的现成的单取代呋喃。合成的二取代呋喃烷显示出释放 NO 的能力,这表明它们作为各种生物医学应用的候选药物具有强大的潜力。

更新日期:2021-10-19

中文翻译:

环畸变多样性导向方法完全取代呋喃和异恶唑
Furoxan 作为前体:实现了药理学取向的完全取代的呋喃和异恶唑的操作简单组装。所提出的协议基于一锅环裂解/亲核加成/氧化级联的现成的单取代呋喃。合成的二取代呋喃烷显示出释放 NO 的能力,这表明它们作为各种生物医学应用的候选药物具有强大的潜力。



























 京公网安备 11010802027423号
京公网安备 11010802027423号