当前位置:
X-MOL 学术
›
J. Chem. Eng. Data
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Solid–Liquid Equilibrium and Thermodynamic Properties of 6-Bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3]pyrimidin-7(8H)-one in 10 Pure Solvents and 3 Different Miscible Systems at T = (278.15–323.15) K
Journal of Chemical & Engineering Data ( IF 2.6 ) Pub Date : 2021-07-21 , DOI: 10.1021/acs.jced.1c00362 Hanhan Xu 1 , Guoxing Hu 1 , Wenge Yang 1 , Wei Sun 1 , Weiliang Shen 1 , Yonghong Hu 1
Journal of Chemical & Engineering Data ( IF 2.6 ) Pub Date : 2021-07-21 , DOI: 10.1021/acs.jced.1c00362 Hanhan Xu 1 , Guoxing Hu 1 , Wenge Yang 1 , Wei Sun 1 , Weiliang Shen 1 , Yonghong Hu 1
Affiliation
|
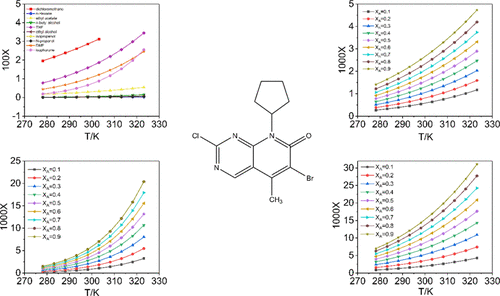
6-Bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3]pyrimidin-7(8H)-one is a key intermediate in the synthesis of Ibrance. We used high-performance liquid chromatography to analyze the solubility of 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3]pyrimidin-7(8H)-one in 10 single solvents [dichloromethane, n-hexane, ethyl acetate, n-butyl alcohol, tetrahydrofuran (THF), ethyl alcohol, isopropanol, 1-propanol, dimethylformamide, and isophorone] and 3 binary solvents (ethyl acetate + ethyl alcohol, isophorone + isopropanol, and THF + ethyl alcohol) at atmospheric pressure from 278.15 to 323.15 K. The results show that temperature and the solubility of 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3]pyrimidin-7(8H)-one in single solvents and binary solvents are positively correlated. The solubility in pure solvents is fitted by the modified Apelblat model and the Buchwski–Ksiazaczak λh model, and the solubility data in the binary solvent are correlated by the CNIBS/R–K model and the Jouyban–Acree model. The good agreement between the calculated value of the equation and the experimental result is shown by the results of equation fitting. By comparing the average value of relative average deviation and root-mean-square deviation of the equations, we can see that the modified Apelblat model has the best correlation effect in the single solution systems and the CNIBS/R–K model has the best correlation effect in the miscible systems. The KAT-LSER model is used to explain the competition between solute–solvent interactions and solvent–solvent interactions in pure solvents.
中文翻译:

6-Bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3]pyrimidin-7(8H)-one 在 10 种纯溶剂和 3 种不同混溶体系中的固液平衡和热力学性质在 T = (278.15 –323.15) K
6-Bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3]pyrimidin-7(8 H )-one 是合成 Ibrance 的关键中间体。我们使用高效液相色谱法分析了 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3]pyrimidin-7(8 H )-one 在 10 种单一溶剂中的溶解度[二氯甲烷, n -己烷、乙酸乙酯、正丁醇、四氢呋喃 (THF)、乙醇、异丙醇、1-丙醇、二甲基甲酰胺和异佛尔酮]和 3 种二元溶剂(乙酸乙酯 + 乙醇、异佛尔酮 + 异丙醇和 THF + 乙醇)在大气压下从 278.15 到 323.15 K。结果表明 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3]pyrimidin-7(8 H)-1 在单一溶剂和二元溶剂中呈正相关。在纯溶剂中的溶解度由修正的 Apelblat 模型和 Buchwski-Ksiazaczak λ h拟合模型,二元溶剂中的溶解度数据通过 CNIBS/R-K 模型和 Jouyban-Acree 模型相关联。方程拟合结果表明方程计算值与实验结果吻合良好。通过比较方程的相对平均偏差和均方根偏差的平均值,可以看出修正的Apelblat模型在单解系统中的相关效果最好,而CNIBS/R-K模型的相关效果最好在混溶系统中的作用。KAT-LSER 模型用于解释纯溶剂中溶质-溶剂相互作用和溶剂-溶剂相互作用之间的竞争。
更新日期:2021-08-12
中文翻译:

6-Bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3]pyrimidin-7(8H)-one 在 10 种纯溶剂和 3 种不同混溶体系中的固液平衡和热力学性质在 T = (278.15 –323.15) K
6-Bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3]pyrimidin-7(8 H )-one 是合成 Ibrance 的关键中间体。我们使用高效液相色谱法分析了 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3]pyrimidin-7(8 H )-one 在 10 种单一溶剂中的溶解度[二氯甲烷, n -己烷、乙酸乙酯、正丁醇、四氢呋喃 (THF)、乙醇、异丙醇、1-丙醇、二甲基甲酰胺和异佛尔酮]和 3 种二元溶剂(乙酸乙酯 + 乙醇、异佛尔酮 + 异丙醇和 THF + 乙醇)在大气压下从 278.15 到 323.15 K。结果表明 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3]pyrimidin-7(8 H)-1 在单一溶剂和二元溶剂中呈正相关。在纯溶剂中的溶解度由修正的 Apelblat 模型和 Buchwski-Ksiazaczak λ h拟合模型,二元溶剂中的溶解度数据通过 CNIBS/R-K 模型和 Jouyban-Acree 模型相关联。方程拟合结果表明方程计算值与实验结果吻合良好。通过比较方程的相对平均偏差和均方根偏差的平均值,可以看出修正的Apelblat模型在单解系统中的相关效果最好,而CNIBS/R-K模型的相关效果最好在混溶系统中的作用。KAT-LSER 模型用于解释纯溶剂中溶质-溶剂相互作用和溶剂-溶剂相互作用之间的竞争。

























 京公网安备 11010802027423号
京公网安备 11010802027423号