Chemosphere ( IF 8.8 ) Pub Date : 2021-06-04 , DOI: 10.1016/j.chemosphere.2021.131093 Yi Zhao 1 , Meixia Fan 1 , Wenlin Zhou 1 , Yanwei Li 2 , Yan Wang 1 , Zongming Xiu 3 , Baoyu Gao 1
|
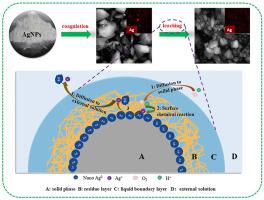
Sludge generated in wastewater treatment facilities is an integral part for the introduction of silver nanoparticles (AgNPs) to the terrestrial environment, which would cause some adverse ecosystem responses. The understanding of silver release process from the sludge is important to evaluate their risks. In this study, the amount and speciation of the released silver were investigated by taking the sludge generated by wastewater coagulation with AgNPs added (denoted as sludgeC-AgNPs) an example, and kinetic analysis and density functional theory (DFT) calculations were first used to explore the controlling steps and pathways about the silver release. The results showed that sludgeC-AgNPs could release the dissolved silver and the colloidal silver. Beside Cl−, Ca2+ in the leaching solution could enhance the silver release of sludgeC-AgNPs, especially for the colloidal silver. The released colloidal silver restricted in size from 40 nm to 100 nm with irregular shape. Although the oxidative dissolution of Ag0 was the origin of the silver release pathways from the sludgeC-AgNPs, the silver diffusion was the controlling step due to the spontaneous binding between silver and the hydrolysates of polyaluminium chloride in sludgeC-AgNPs. However, Ca2+ in the leaching solution could occupy the binding site of silver on sludgeC-AgNPs, which would increase the diffusion rate of silver over the oxidative rate of Ag0. With this condition, the controlling step of silver release from sludgeC-AgNPs turned to the oxidative dissolution of Ag0. Our findings are important to assess the fate of AgNPs in wastewater treatment as well as sludge applications.
中文翻译:

掺入银纳米颗粒的废水混凝产生的污泥中银释放的形态、控制步骤和途径
废水处理设施中产生的污泥是将银纳米粒子 (AgNPs) 引入陆地环境的一个组成部分,这会导致一些不利的生态系统反应。了解污泥中银的释放过程对于评估其风险很重要。本研究以添加AgNPs的废水混凝产生的污泥(表示为污泥C-AgNPs)为例,首先利用动力学分析和密度泛函理论(DFT)计算来研究银的释放量和形态探索银释放的控制步骤和途径。结果表明污泥C-AgNPs可以释放溶解的银和胶体银。在 Cl 旁边-,浸出液中的Ca 2+可以增强污泥C-AgNPs的银释放,特别是对于胶体银。释放的胶体银大小限制在 40 nm 到 100 nm 之间,形状不规则。虽然 Ag 0的氧化溶解是污泥C-AgNPs银释放途径的起源,但由于银与污泥C-AgNPs中聚合氯化铝的水解产物之间的自发结合,银扩散是控制步骤。然而,浸出液中的Ca 2+可以占据污泥C-AgNPs上银的结合位点,这将增加银的扩散速度而不是银的氧化速度。0 . 在此条件下,污泥C-AgNPs银释放的控制步骤转向Ag 0的氧化溶解。我们的发现对于评估 AgNPs 在废水处理和污泥应用中的命运很重要。

























 京公网安备 11010802027423号
京公网安备 11010802027423号