Journal of Molecular Graphics and Modelling ( IF 2.9 ) Pub Date : 2021-03-08 , DOI: 10.1016/j.jmgm.2021.107896 Nasir Shahzad 1 , Basharat Khan 2
|
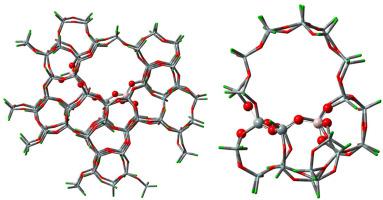
Nowadays, theoretical calculations are playing a pivotal role in understanding the underlying reaction mechanism and at the same time used for exploration of the zeolite-based catalysts. For such systems, an inconsistency is seen in formulating the active site and choosing the size of the zeolite framework. Herein, we have formulated the standard theoretical model for the MFI-based catalytic systems considering the most recent experimental findings.[1] The formulated model is used for the explanation of the mechanism of acetic acid formation by the co-activation of CO2 and CH4 over the MFI based catalyst, which excellently justifies the experimental results. The following conclusions are drawn from the work, a sufficiently large zeolite framework is critical to validate the experimental findings. The extended zeolite framework is crucial for accurate optimization of the active site, drawing the plausible mechanisms and predicting the correct rate-limiting step of the reaction. Moreover, the neighbor Al–Al atoms must be kept away from each other by at-least two Si-centers to be consistent with the experiment.
中文翻译:

通过CO 2和CH 4的共活化,了解MFI框架对直接形成CH 3 COOH的机理和动力学的影响
如今,理论计算在理解潜在的反应机理以及同时用于探索基于沸石的催化剂方面起着举足轻重的作用。对于这样的系统,在配制活性部位和选择沸石骨架的尺寸时看到不一致的地方。在此,我们考虑到最新的实验结果,为基于MFI的催化系统制定了标准理论模型。[1] 所建立的模型用于解释CO 2和CH 4的共活化形成乙酸的机理。在MFI基催化剂上,这很好地证明了实验结果的合理性。从工作中得出以下结论,足够大的沸石骨架对于验证实验结果至关重要。扩展的沸石骨架对于精确优化活性位点,绘制合理的机理并预测反应的正确限速步骤至关重要。此外,与实验相一致,相邻的Al-Al原子必须至少保持两个Si中心彼此远离。



























 京公网安备 11010802027423号
京公网安备 11010802027423号