Chemosphere ( IF 8.8 ) Pub Date : 2021-02-22 , DOI: 10.1016/j.chemosphere.2021.130020 Sayed Mukit Hossain , Heeju Park , Hui-Ju Kang , Jong Seok Mun , Leonard Tijing , Inkyu Rhee , Jong-Ho Kim , Young-Si Jun , Ho Kyong Shon
|
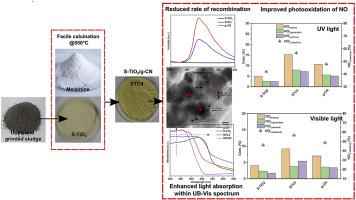
In this study, sludges generated from Ti-based flocculation of dye wastewater were used to retrieve photoactive titania (S–TiO2). It was heterojunctioned with graphitic carbon nitride (g-CN) to augment photoactivity under UV/visible light irradiance. Later the as-prepared samples were utilized to remove nitrogen oxides (NOx) in the atmospheric condition through photocatalysis. Heterojunction between S–TiO2 and g-CN was prepared through facile calcination (@550 °C) of S–TiO2 and melamine mix. Advanced sample characterization was carried out and documented extensively. Successful heterojunction was confirmed from the assessment of morphological and optical attributes of the samples. Finally, the prepared samples’ level of photoactivity was assessed through photooxidation of NOx under both UV and visible light irradiance. Enhanced photoactivity was observed in the prepared samples irrespective of the light types. After 1 h of UV/visible light-based photooxidation, the best sample STC4 was found to remove 15.18% and 9.16% of atmospheric NO, respectively. In STC4, the mixing ratio of S–TiO2, to melamine was maintained as 1:3. Moreover, the optical bandgap of STC4 was found as 2.65 eV, where for S–TiO2, it was 2.83 eV. Hence, the restrained rate of photogenerated charge recombination and tailored energy bandgap of the as-prepared samples were the primary factors for enhancing photoactivity.
中文翻译:

合成和NO X的锐钛矿的去除性能S-的TiO 2 /克- CN异质结从染料废水污泥形成
在这项研究中,从染料废水的钛基絮凝中产生的污泥被用于回收光活性二氧化钛(S–TiO 2)。它与石墨碳氮化物(g-CN)异质连接,以增强UV /可见光辐照下的光活性。随后,将所制备的样品用于在大气条件下通过光催化去除氮氧化物(NO x)。S-的TiO之间异质结2和g-CN通过容易煅烧制备(@ 550℃)的S-的TiO 2和三聚氰胺混合。进行了高级样品表征,并进行了广泛记录。通过评估样品的形态和光学属性,证实了成功的异质结。最后,通过在紫外线和可见光照射下对NO x进行光氧化来评估所制备样品的光活性水平。不管光的类型如何,在制备的样品中观察到增强的光活性。经过基于紫外线/可见光的光氧化1小时后,发现最佳样品STC4分别去除了大气NO的15.18%和9.16%。在STC4中,S-TiO 2与三聚氰胺的混合比保持为1:3。此外,发现STC4的光学带隙为2.65 eV,其中S–TiO 2为,它是2.83 eV。因此,所制备样品的光生电荷复合的抑制速率和定制的能带隙是增强光活性的主要因素。


























 京公网安备 11010802027423号
京公网安备 11010802027423号