当前位置:
X-MOL 学术
›
J. Mater. Chem. B
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Tumor microenvironment-responsive multifunctional nanoplatform based on MnFe2O4-PEG for enhanced magnetic resonance imaging-guided hypoxic cancer radiotherapy
Journal of Materials Chemistry B ( IF 7 ) Pub Date : 2021-1-15 , DOI: 10.1039/d0tb02631j Zhenhu He 1, 2, 3, 4, 5 , Haixiong Yan 1, 2, 3, 4, 5 , Wenbin Zeng 3, 4, 5, 6 , Kai Yang 5, 7, 8, 9 , Pengfei Rong 1, 2, 3, 4, 5
Journal of Materials Chemistry B ( IF 7 ) Pub Date : 2021-1-15 , DOI: 10.1039/d0tb02631j Zhenhu He 1, 2, 3, 4, 5 , Haixiong Yan 1, 2, 3, 4, 5 , Wenbin Zeng 3, 4, 5, 6 , Kai Yang 5, 7, 8, 9 , Pengfei Rong 1, 2, 3, 4, 5
Affiliation
|
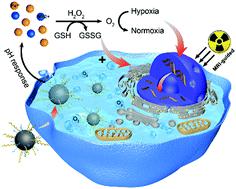
Radiotherapy occupies an essential position in curing and palliating a wide range of solid tumors based on DNA damage responses to eradicate cancer cells. However, the tumor microenvironment generally exhibits the characteristics of hypoxia and glutathione overexpression, which play a critical role in radioresistance, to prevent irreparable breaks to DNA and necrocytosis of cancer cells. Herein, polyethylene glycol (PEG) functionalized manganese ferrite nanoparticles (MnFe2O4-PEG) are designed to enable self-sufficiency of oxygen by continuously catalyzing the decomposition of endogenous hydrogen peroxide. Simultaneously, the nano-platform can consume GSH to reduce the loss of reactive oxygen species in radiotherapy and achieve better therapeutic effects at the cellular and animal levels. In addition, the MnFe2O4-PEG could act as an optimal T1- and T2-weighted contrast medium for tumor-specific magnetic resonance imaging. This work proposes a systematically administered radiosensitizer that can selectively reside in tumor sites via the enhanced permeability and retention effect to relieve hypoxia and reduce GSH concentration, combined with dual-mode magnetic resonance imaging, achieving precise and effective image-guided tumor therapy.
中文翻译:

基于MnFe2O4-PEG的肿瘤微环境响应多功能纳米平台,用于增强磁共振成像引导的缺氧癌放疗
放射疗法在根除癌细胞的DNA损伤反应中,在治愈和减轻各种实体瘤中占有重要地位。然而,肿瘤微环境通常表现出低氧和谷胱甘肽过表达的特征,其在放射抗性中起关键作用,以防止DNA的不可修复的断裂和癌细胞的坏死作用。本文中,聚乙二醇(PEG)功能化的锰铁氧体纳米颗粒(MnFe 2 O 4-PEG)被设计为通过连续催化内源性过氧化氢的分解来实现氧的自给。同时,纳米平台可以消耗GSH来减少放射治疗中活性氧的损失,并在细胞和动物水平上获得更好的治疗效果。此外,MnFe 2 O 4 -PEG可以作为肿瘤特异性磁共振成像的最佳T 1和T 2加权造影剂。这项工作提出了一种系统给药的放射增敏剂,它可以通过 增强的通透性和保留效果,以减轻缺氧并降低GSH浓度,结合双模磁共振成像,实现精确有效的图像引导肿瘤治疗。
更新日期:2021-01-21
中文翻译:

基于MnFe2O4-PEG的肿瘤微环境响应多功能纳米平台,用于增强磁共振成像引导的缺氧癌放疗
放射疗法在根除癌细胞的DNA损伤反应中,在治愈和减轻各种实体瘤中占有重要地位。然而,肿瘤微环境通常表现出低氧和谷胱甘肽过表达的特征,其在放射抗性中起关键作用,以防止DNA的不可修复的断裂和癌细胞的坏死作用。本文中,聚乙二醇(PEG)功能化的锰铁氧体纳米颗粒(MnFe 2 O 4-PEG)被设计为通过连续催化内源性过氧化氢的分解来实现氧的自给。同时,纳米平台可以消耗GSH来减少放射治疗中活性氧的损失,并在细胞和动物水平上获得更好的治疗效果。此外,MnFe 2 O 4 -PEG可以作为肿瘤特异性磁共振成像的最佳T 1和T 2加权造影剂。这项工作提出了一种系统给药的放射增敏剂,它可以通过 增强的通透性和保留效果,以减轻缺氧并降低GSH浓度,结合双模磁共振成像,实现精确有效的图像引导肿瘤治疗。

























 京公网安备 11010802027423号
京公网安备 11010802027423号