当前位置:
X-MOL 学术
›
Org. Chem. Front.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Thermodynamic and kinetic studies of hydride transfer from Hantzsch ester under the promotion of organic bases
Organic Chemistry Frontiers ( IF 5.4 ) Pub Date : 2021-1-4 , DOI: 10.1039/d0qo01478h Zhen Li 1, 2, 3, 4, 5 , Jin-Dong Yang 1, 2, 3, 4, 5 , Jin-Pei Cheng 1, 2, 3, 4, 5
Organic Chemistry Frontiers ( IF 5.4 ) Pub Date : 2021-1-4 , DOI: 10.1039/d0qo01478h Zhen Li 1, 2, 3, 4, 5 , Jin-Dong Yang 1, 2, 3, 4, 5 , Jin-Pei Cheng 1, 2, 3, 4, 5
Affiliation
|
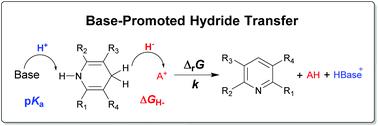
Base-promoted hydride transfer (BPHyT), a strategy for the upconversion of hydride donors, was studied here in a three-component system, composed of Hantzsch ester (HEH), acridinium derivatives and organic bases. Based on the thermodynamic parameters of hydricity and pKa, we proposed a thermodynamic cycle to evaluate the apparent hydricity of HEH/base combinations, as well as the overall driving force of BPHyT. Brønsted-type linear analysis indicated that the base used in BPHyT is much more effective to regulate the reaction kinetics, compared to conventional Brønsted acid or base catalysis. Structure–reactivity relationships showed that the hydride acceptor and the base contribute equally to regulate the kinetics of BPHyT. Kinetic isotope effects suggested that the hydride transfer is involved in the rate-determining step. Reductions of the polar C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds by HEH/base combinations were performed to confirm the feasibility of applying BPHyT in organic synthesis.
C bonds by HEH/base combinations were performed to confirm the feasibility of applying BPHyT in organic synthesis.
中文翻译:

在有机碱促进下从Hantzsch酯转移氢化物的热力学和动力学研究
在由Hantzsch酯(HEH),a啶衍生物和有机碱组成的三组分体系中,研究了碱促进的氢化物转移(BPHyT),一种氢化物供体的上转换策略。基于水力和p K a的热力学参数,我们提出了一个热力学循环来评价HEH的表观水力。/ base组合,以及BPHyT的整体驱动力。布朗斯台德型线性分析表明,与传统的布朗斯台德酸或碱催化相比,BPHyT中使用的碱能更有效地调节反应动力学。结构-反应关系表明,氢化物受体和碱同样可调节BPHyT的动力学。动力学同位素效应表明,氢化物转移参与了速率确定步骤。![[双键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e001.gif) 通过HEH /碱组合还原极性C C键,以证实在有机合成中应用BPHyT的可行性。
通过HEH /碱组合还原极性C C键,以证实在有机合成中应用BPHyT的可行性。
更新日期:2021-01-13
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds by HEH/base combinations were performed to confirm the feasibility of applying BPHyT in organic synthesis.
C bonds by HEH/base combinations were performed to confirm the feasibility of applying BPHyT in organic synthesis.
中文翻译:

在有机碱促进下从Hantzsch酯转移氢化物的热力学和动力学研究
在由Hantzsch酯(HEH),a啶衍生物和有机碱组成的三组分体系中,研究了碱促进的氢化物转移(BPHyT),一种氢化物供体的上转换策略。基于水力和p K a的热力学参数,我们提出了一个热力学循环来评价HEH的表观水力。/ base组合,以及BPHyT的整体驱动力。布朗斯台德型线性分析表明,与传统的布朗斯台德酸或碱催化相比,BPHyT中使用的碱能更有效地调节反应动力学。结构-反应关系表明,氢化物受体和碱同样可调节BPHyT的动力学。动力学同位素效应表明,氢化物转移参与了速率确定步骤。
![[双键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e001.gif) 通过HEH /碱组合还原极性C C键,以证实在有机合成中应用BPHyT的可行性。
通过HEH /碱组合还原极性C C键,以证实在有机合成中应用BPHyT的可行性。

























 京公网安备 11010802027423号
京公网安备 11010802027423号