Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Insight into the Critical Role of Exchange Current Density on Electrodeposition Behavior of Lithium Metal
Advanced Science ( IF 15.1 ) Pub Date : 2021-01-06 , DOI: 10.1002/advs.202003301 Yangyang Liu 1 , Xieyu Xu 2 , Matthew Sadd 3 , Olesya O Kapitanova 4 , Victor A Krivchenko 4 , Jun Ban 1 , Jialin Wang 1 , Xingxing Jiao 1 , Zhongxiao Song 1 , Jiangxuan Song 1 , Shizhao Xiong 3 , Aleksandar Matic 3
Advanced Science ( IF 15.1 ) Pub Date : 2021-01-06 , DOI: 10.1002/advs.202003301 Yangyang Liu 1 , Xieyu Xu 2 , Matthew Sadd 3 , Olesya O Kapitanova 4 , Victor A Krivchenko 4 , Jun Ban 1 , Jialin Wang 1 , Xingxing Jiao 1 , Zhongxiao Song 1 , Jiangxuan Song 1 , Shizhao Xiong 3 , Aleksandar Matic 3
Affiliation
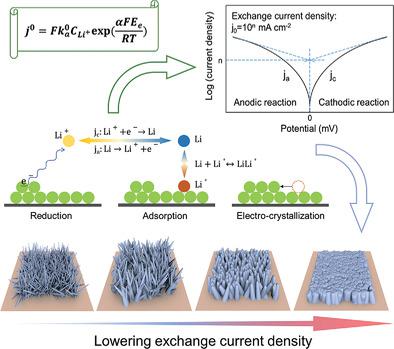
|
Due to an ultrahigh theoretical specific capacity of 3860 mAh g−1, lithium (Li) is regarded as the ultimate anode for high‐energy‐density batteries. However, the practical application of Li metal anode is hindered by safety concerns and low Coulombic efficiency both of which are resulted fromunavoidable dendrite growth during electrodeposition. This study focuses on a critical parameter for electrodeposition, the exchange current density, which has attracted only little attention in research on Li metal batteries. A phase‐field model is presented to show the effect of exchange current density on electrodeposition behavior of Li. The results show that a uniform distribution of cathodic current density, hence uniform electrodeposition, on electrode is obtained with lower exchange current density. Furthermore, it is demonstrated that lower exchange current density contributes to form a larger critical radius of nucleation in the initial electrocrystallization that results in a dense deposition of Li, which is a foundation for improved Coulombic efficiency and dendrite‐free morphology. The findings not only pave the way to practical rechargeable Li metal batteries but can also be translated to the design of stable metal anodes, e.g., for sodium (Na), magnesium (Mg), and zinc (Zn) batteries.
中文翻译:

深入了解交换电流密度对锂金属电沉积行为的关键作用
由于3860 mAh g -1的超高理论比容量,锂(Li)被认为是高能量密度电池的终极负极。然而,锂金属负极的实际应用受到安全问题和低库仑效率的阻碍,这两者都是由于电沉积过程中不可避免的枝晶生长造成的。本研究重点关注电沉积的一个关键参数——交换电流密度,该参数在锂金属电池的研究中很少引起关注。提出了一个相场模型来显示交换电流密度对锂电沉积行为的影响。结果表明,以较低的交换电流密度获得了电极上阴极电流密度的均匀分布,从而获得了均匀的电沉积。此外,研究表明,较低的交换电流密度有助于在初始电结晶中形成更大的临界成核半径,从而导致锂的致密沉积,这是提高库仑效率和无枝晶形态的基础。这些发现不仅为实用的可充电锂金属电池铺平了道路,而且还可以转化为稳定金属阳极的设计,例如钠(Na)、镁(Mg)和锌(Zn)电池。
更新日期:2021-03-03
中文翻译:

深入了解交换电流密度对锂金属电沉积行为的关键作用
由于3860 mAh g -1的超高理论比容量,锂(Li)被认为是高能量密度电池的终极负极。然而,锂金属负极的实际应用受到安全问题和低库仑效率的阻碍,这两者都是由于电沉积过程中不可避免的枝晶生长造成的。本研究重点关注电沉积的一个关键参数——交换电流密度,该参数在锂金属电池的研究中很少引起关注。提出了一个相场模型来显示交换电流密度对锂电沉积行为的影响。结果表明,以较低的交换电流密度获得了电极上阴极电流密度的均匀分布,从而获得了均匀的电沉积。此外,研究表明,较低的交换电流密度有助于在初始电结晶中形成更大的临界成核半径,从而导致锂的致密沉积,这是提高库仑效率和无枝晶形态的基础。这些发现不仅为实用的可充电锂金属电池铺平了道路,而且还可以转化为稳定金属阳极的设计,例如钠(Na)、镁(Mg)和锌(Zn)电池。

























 京公网安备 11010802027423号
京公网安备 11010802027423号