Environmental Research ( IF 8.3 ) Pub Date : 2021-01-06 , DOI: 10.1016/j.envres.2020.110678 You-Sheng Liu , Yu-Xiao Cheng , Dan Wu , Quan-Le Chen , Guang-Guo Ying
|
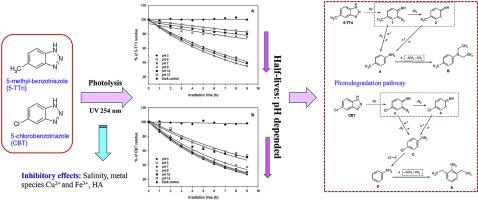
5-methylbenzotriazole (5-TTri) and 5-chlorobenzotriazole (CBT) are two benzotriazole derivatives widely used in various industrial and domestic applications. This paper reports on the photochemical behaviour of 5-TTri and CBT in aqueous solutions under UV radiation at 254 nm and the influences of pH, salinity, metal species and humic acid (HA) on their photo-transformation processes. The photolysis of 5-TTri and CBT under the exposure to UV light were found to follow the first-order reaction kinetic in all cases with half-lives ranging from 7.1 h to 24.3 h for 5-TTri and 5.1 h–20.5 h for CBT in various aqueous solutions containing metal ions and HA. The photolysis rates for both 5-TTri and CBT were strongly dependent on the solution pH value, and decreased with increasing solution pH. Salinity, metal species Cu2+ and Fe3+, and especially HA had inhibitory effects on the photolysis of 5-TTri and CBT under UV light irradiation at 254 nm. We proposed the tentative photo transformation schemes for both 5-TTri and CBT, which involved two photoproducts (4-methylaniline and N, N-diethylaniline- p-toluidine) and three photoproducts (4-chloroaniline, Aniline and 2,6-diethylaniline), respectively, via N–N and N–NH bond scission and dechlorination process.
中文翻译:

紫外线辐射5-甲基苯并三唑和5-氯苯并三唑的光转化:pH,盐度,金属种类和腐殖酸的影响
5-甲基苯并三唑(5-TTri)和5-氯苯并三唑(CBT)是广泛用于各种工业和家庭应用的两种苯并三唑衍生物。本文报道了5-TTri和CBT在254 nm紫外线下在水溶液中的光化学行为,以及pH,盐度,金属种类和腐殖酸(HA)对其光转化过程的影响。在所有情况下,对5-TTri和CBT的光解反应均遵循一级反应动力学,其中5-TTri的半衰期为7.1 h至24.3 h,CBT的半衰期为7.1 h至20.5 h在含有金属离子和HA的各种水溶液中 5-TTri和CBT的光解速率都强烈依赖于溶液的pH值,并且随着溶液pH值的增加而降低。盐度,金属种类Cu 2+Fe 3+,特别是HA对254 nm紫外线照射下5-TTri和CBT的光解有抑制作用。我们提出了针对5-TTri和CBT的暂定光转换方案,其中涉及两个光产物(4-甲基苯胺和N,N-二乙基苯胺-对甲苯胺)和三个光产物(4-氯苯胺,苯胺和2,6-二乙基苯胺)分别通过N–N和N–NH键断裂和脱氯过程。


























 京公网安备 11010802027423号
京公网安备 11010802027423号