Computational and Structural Biotechnology Journal ( IF 6 ) Pub Date : 2020-12-23 , DOI: 10.1016/j.csbj.2020.12.023 Jessica Duprez 1, 2 , Kristen Kalbfleisch 3 , Sasmit Deshmukh 4 , Jessie Payne 1 , Manjit Haer 1 , Wayne Williams 1 , Ibrahim Durowoju 1 , Marina Kirkitadze 1
|
Purpose
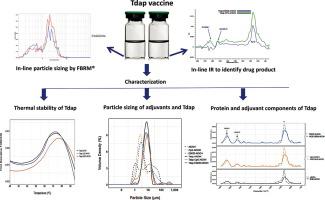
The goal of this study was to characterize an acellular pertussis vaccine (Tdap) containing genetically modified pertussis toxin (gdPT) and TLR agonist adsorbed to AlOOH adjuvant.
Methods
Several analytical tools including nanoDSF, FTIR, and LD were used to examine the conformation of novel gdPT and the composition of AlOOH adjuvant formulations adsorbed to pertussis vaccine.
Results
DLS particle size results were 9.3 nm and 320 nm for gdPT. For pertussis toxoid (PT), the DLS particle size results were larger at ~440 nm. After adsorption to AlOOH, which was driven by the protein antigen, the size distribution ranged from 3.5 to 22 µm. Two thermal transitions were observed by DSC for gdPT at 70 °C and 102 °C. The main thermal transition was confirmed to be at 72 °C by nanoDSF. All three vaccine formulations showed one thermal transition: Tdap-AlOOH had a thermal transition of 74.6 °C, Tdap-E6020-AlOOH had a thermal transition at 74.2 °C, and Tdap-CpG-AlOOH had a thermal transition at 77.0 °C. Analysis of pertussis toxin (PTx) and gdPT was also performed by FTIR spectroscopy for the purpose of comparison. The second derivative of the FTIR spectra showed an additional feature for PTx at 1685 cm−1 compared to gdPT. The antigen’s amide I and II regions were largely unchanged after adsorption to AlOOH adjuvant as shown by FTIR, suggesting that there were no significant changes in the secondary structure.
Conclusion
gdPT conformation was successfully characterized using an array of analytical methods. All three Tdap formulations have similar thermal stability as shown by nanoDSF, similar size distribution as shown by LD, and similar overall secondary structure as shown by FTIR. In-line particle sizing and IR can be used as in-process characterization tools to monitor consistency of adsorbed vaccine and to confirm product identity.
中文翻译:

羟基铝吸附百日咳疫苗的结构和成分分析
目的
本研究的目的是表征一种含有转基因百日咳毒素 (gdPT) 和吸附在 AlOOH 佐剂上的 TLR 激动剂的无细胞百日咳疫苗 (Tdap)。
方法
包括 nanoDSF、FTIR 和 LD 在内的几种分析工具用于检查新型 gdPT 的构象和吸附到百日咳疫苗的 AlOOH 佐剂制剂的组成。
结果
对于 gdPT,DLS 粒径结果为 9.3 nm 和 320 nm。对于百日咳类毒素 (PT),DLS 粒径结果在 ~440 nm 处较大。吸附到由蛋白质抗原驱动的 AlOOH 后,尺寸分布范围为 3.5 至 22 µm。DSC 在 70 °C 和 102 °C 下观察到 gdPT 的两个热转变。通过 nanoDSF 确认主要热转变为 72 °C。所有三种疫苗制剂都显示出一个热转变:Tdap-AlOOH 的热转变为 74.6 °C,Tdap-E6020-AlOOH 的热转变为 74.2 °C,Tdap-CpG-AlOOH 的热转变为 77.0 °C。百日咳毒素 (PTx) 和 gdPT 的分析也通过 FTIR 光谱进行,以进行比较。FTIR 光谱的二阶导数在 1685 cm -1处显示了 PTx 的附加特征与 gdPT 相比。如FTIR所示,抗原的酰胺I和II区域在吸附到AlOOH佐剂后基本没有变化,表明二级结构没有显着变化。
结论
使用一系列分析方法成功地表征了 gdPT 构象。如 nanoDSF 所示,所有三种 Tdap 配方具有相似的热稳定性,如 LD 所示的相似尺寸分布,以及如 FTIR 所示的相似的整体二级结构。在线粒度测定和 IR 可用作过程中表征工具,以监测吸附疫苗的一致性并确认产品身份。

























 京公网安备 11010802027423号
京公网安备 11010802027423号