当前位置:
X-MOL 学术
›
Angew. Chem. Int. Ed.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Formic Acid‐Assisted Selective Hydrogenolysis of 5‐Hydroxymethylfurfural to 2,5‐Dimethylfuran over Bifunctional Pd Nanoparticles Supported on N‐Doped Mesoporous Carbon
Angewandte Chemie International Edition ( IF 16.6 ) Pub Date : 2020-12-07 , DOI: 10.1002/anie.202012816 Bin Hu 1, 2 , Lisa Warczinski 3 , Xiaoyu Li 1 , Mohong Lu 4 , Johannes Bitzer 1 , Markus Heidelmann 5 , Till Eckhard 1 , Qi Fu 1 , Jonas Schulwitz 1 , Mariia Merko 1 , Mingshi Li 4 , Wolfgang Kleist 1 , Christof Hättig 3 , Martin Muhler 1, 2 , Baoxiang Peng 1, 2
Angewandte Chemie International Edition ( IF 16.6 ) Pub Date : 2020-12-07 , DOI: 10.1002/anie.202012816 Bin Hu 1, 2 , Lisa Warczinski 3 , Xiaoyu Li 1 , Mohong Lu 4 , Johannes Bitzer 1 , Markus Heidelmann 5 , Till Eckhard 1 , Qi Fu 1 , Jonas Schulwitz 1 , Mariia Merko 1 , Mingshi Li 4 , Wolfgang Kleist 1 , Christof Hättig 3 , Martin Muhler 1, 2 , Baoxiang Peng 1, 2
Affiliation
|
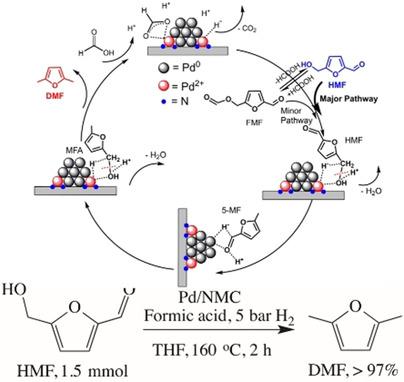
Biomass‐derived 5‐hydroxymethylfurfural (HMF) is regarded as one of the most promising platform chemicals to produce 2,5‐dimethylfuran (DMF) as a potential liquid transportation fuel. Pd nanoparticles supported on N‐containing and N‐free mesoporous carbon materials were prepared, characterized, and applied in the hydrogenolysis of HMF to DMF under mild reaction conditions. Quantitative conversion of HMF to DMF was achieved in the presence of formic acid (FA) and H2 over Pd/NMC within 2 h. The reaction mechanism, especially the multiple roles of FA, was explored through a detailed comparative study by varying hydrogen source, additive, and substrate as well as by applying in situ ATR‐IR spectroscopy. The major role of FA is to shift the dominant reaction pathway from the hydrogenation of the aldehyde group to the hydrogenolysis of the hydroxymethyl group via the protonation by FA at the C‐OH group, lowering the activation barrier of the C−O bond cleavage and thus significantly enhancing the reaction rate. XPS results and DFT calculations revealed that Pd2+ species interacting with pyridine‐like N atoms significantly enhance the selective hydrogenolysis of the C−OH bond in the presence of FA due to their high ability for the activation of FA and the stabilization of H−.
中文翻译:

N掺杂介孔碳负载的双功能Pd纳米颗粒上甲酸辅助的5-羟基甲基糠醛选择性氢解为2,5-二甲基呋喃
生物质衍生的5-羟甲基糠醛(HMF)被认为是最有前途的平台化学品之一,可以生产2,5-二甲基呋喃(DMF)作为潜在的液体运输燃料。制备,表征了负载在含N和无N介孔碳材料上的Pd纳米颗粒,并在温和的反应条件下将其用于HMF氢解为DMF。在甲酸(FA)和H 2的存在下,HMF定量转化为DMF在2小时内通过Pd / NMC处理。通过详细的比较研究,通过改变氢源,添加剂和底物以及应用原位ATR-IR光谱,探索了反应机理,尤其是FA的多种作用。FA的主要作用是通过FA在C-OH基上的质子化作用,将主要的反应路径从醛基的氢化转变为羟甲基的氢解,从而降低了C-O键断裂的活化能,从而大大提高了反应速度。XPS结果和DFT计算表明,与FA类似的Pd 2+物种与吡啶样N原子相互作用可显着增强C-OH键的选择性氢解,因为它们具有激活FA和稳定H的能力。-。
更新日期:2020-12-07
中文翻译:

N掺杂介孔碳负载的双功能Pd纳米颗粒上甲酸辅助的5-羟基甲基糠醛选择性氢解为2,5-二甲基呋喃
生物质衍生的5-羟甲基糠醛(HMF)被认为是最有前途的平台化学品之一,可以生产2,5-二甲基呋喃(DMF)作为潜在的液体运输燃料。制备,表征了负载在含N和无N介孔碳材料上的Pd纳米颗粒,并在温和的反应条件下将其用于HMF氢解为DMF。在甲酸(FA)和H 2的存在下,HMF定量转化为DMF在2小时内通过Pd / NMC处理。通过详细的比较研究,通过改变氢源,添加剂和底物以及应用原位ATR-IR光谱,探索了反应机理,尤其是FA的多种作用。FA的主要作用是通过FA在C-OH基上的质子化作用,将主要的反应路径从醛基的氢化转变为羟甲基的氢解,从而降低了C-O键断裂的活化能,从而大大提高了反应速度。XPS结果和DFT计算表明,与FA类似的Pd 2+物种与吡啶样N原子相互作用可显着增强C-OH键的选择性氢解,因为它们具有激活FA和稳定H的能力。-。

























 京公网安备 11010802027423号
京公网安备 11010802027423号