当前位置:
X-MOL 学术
›
J. Phys. Chem. C
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Impact of π-Conjugated Linkers on the Effective Exciton Binding Energy of Diketopyrrolopyrrole–Dithienopyrrole Copolymers
The Journal of Physical Chemistry C ( IF 3.7 ) Pub Date : 2020-12-02 , DOI: 10.1021/acs.jpcc.0c08768 Pieter J. Leenaers 1 , Arthur J. L. A. Maufort 1 , Martijn M. Wienk 1 , René A. J. Janssen 1, 2
The Journal of Physical Chemistry C ( IF 3.7 ) Pub Date : 2020-12-02 , DOI: 10.1021/acs.jpcc.0c08768 Pieter J. Leenaers 1 , Arthur J. L. A. Maufort 1 , Martijn M. Wienk 1 , René A. J. Janssen 1, 2
Affiliation
|
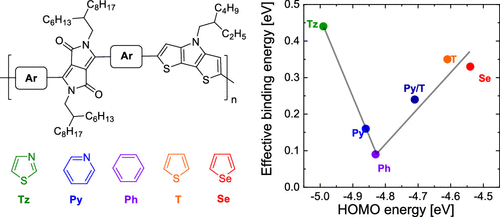
The effect of the nature of the π-conjugated linker that is positioned between electron-deficient 2,5-dihydropyrrolo[3,4-c]pyrrole-1,4-dione (DPP) and electron-rich dithieno[3,2-b:2′,3′-d]pyrrole (DTP) units in alternating DPP–DTP copolymers on the optical and electrochemical band gaps and the effective exciton binding energy is investigated for six different aromatic linkers. The optical band gap is related to the electron-donating properties of DTP and the electron-withdrawing properties of DPP but likewise strongly affected by the nature of the linker and varies between 1.13 and 1.80 eV for the six different linkers. The lowest optical band gaps are found for linkers that either raise the highest occupied molecular orbital or lower the lowest unoccupied molecular orbital most, while the highest optical band gap is found for phenyl linkers that have neither strong donating nor strong accepting properties. Along with the optical band gap, the electrochemical band gap also changes, but to a lesser extent from 1.46 to 1.89 eV. The effective exciton binding energy (Eb), defined as the difference between the electrochemical and optical band gaps, decreases with an increasing band gap and reaches a minimum of 0.09 eV for the copolymer with the highest band gap, that is, with phenyl linkers. The reduction in Eb with an increasing band gap is tentatively explained by a reduced electronic interaction between the DTP and DPP units when the HOMO localizes on DTP and the LUMO localizes on DPP. Support for this explanation is found in the molar absorption coefficient of the copolymers, which shows an overall decreasing trend with decreasing Eb.
中文翻译:

π共轭连接基对二酮吡咯并吡咯-二硫代吡咯共聚物有效激子结合能的影响
位于缺电子的2,5-二氢吡咯并[3,4- c ]吡咯-1,4-二酮(DPP)和富电子的二噻吩并[3,2-]之间的π共轭连接基的性质的影响b:2′,3′- d研究了六种不同的芳族连接基在光学和电化学带隙上交替的DPP-DTP共聚物中的]吡咯(DTP)单元和有效激子结合能。光学带隙与DTP的供电子特性和DPP的吸电子特性有关,但同样受到连接子性质的强烈影响,并且对于六个不同的连接子在1.13和1.80 eV之间变化。对于提高最高占据分子轨道或降低最低未占据分子轨道的连接子,发现了最低的光学带隙,而对于既没有强供体也没有强接受性的苯基连接器发现了最高的光学带隙。电化学带隙与光学带隙一起也发生变化,但幅度较小,从1.46到1.89 eV。E b,定义为电化学带隙与光学带隙之间的差,随着带隙的增加而减小,并且对于具有最高带隙的共聚物,即具有苯基接头的共聚物,其最小达到0.09 eV。在还原ë b随着增加的带隙暂定由DTP和DPP单元之间的减小的电子交互说明的,当HOMO本地化上DTP和LUMO本地化上DPP。在共聚物的摩尔吸收系数中找到了支持该解释的证据,该摩尔吸收系数显示出随着E b降低而整体降低的趋势。
更新日期:2020-12-17
中文翻译:

π共轭连接基对二酮吡咯并吡咯-二硫代吡咯共聚物有效激子结合能的影响
位于缺电子的2,5-二氢吡咯并[3,4- c ]吡咯-1,4-二酮(DPP)和富电子的二噻吩并[3,2-]之间的π共轭连接基的性质的影响b:2′,3′- d研究了六种不同的芳族连接基在光学和电化学带隙上交替的DPP-DTP共聚物中的]吡咯(DTP)单元和有效激子结合能。光学带隙与DTP的供电子特性和DPP的吸电子特性有关,但同样受到连接子性质的强烈影响,并且对于六个不同的连接子在1.13和1.80 eV之间变化。对于提高最高占据分子轨道或降低最低未占据分子轨道的连接子,发现了最低的光学带隙,而对于既没有强供体也没有强接受性的苯基连接器发现了最高的光学带隙。电化学带隙与光学带隙一起也发生变化,但幅度较小,从1.46到1.89 eV。E b,定义为电化学带隙与光学带隙之间的差,随着带隙的增加而减小,并且对于具有最高带隙的共聚物,即具有苯基接头的共聚物,其最小达到0.09 eV。在还原ë b随着增加的带隙暂定由DTP和DPP单元之间的减小的电子交互说明的,当HOMO本地化上DTP和LUMO本地化上DPP。在共聚物的摩尔吸收系数中找到了支持该解释的证据,该摩尔吸收系数显示出随着E b降低而整体降低的趋势。

























 京公网安备 11010802027423号
京公网安备 11010802027423号