Journal of Photochemistry and Photobiology A: Chemistry ( IF 4.3 ) Pub Date : 2020-12-03 , DOI: 10.1016/j.jphotochem.2020.113004 Zahra Abbasi , Elisa I. García-López , Giuseppe Marcì , Abdolhadi Farrokhnia , Morteza Zargar Shoushtari
|
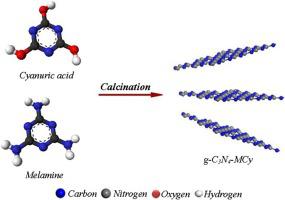
A graphitic carbon nitride (g-C3N4-MCy) photocatalyst was prepared by thermal condensation of melamine and cyanuric acid with equal proportions. The photocatalyst was characterized by Fourier transform infrared spectroscopy (FTIR), Specific surface area (SSA), X-ray diffraction (XRD), Field Emission Scanning Electron Microscope (FESEM), Transmission electron microscope (TEM), and DRS–UV–Vis spectroscopic methods. We studied the photo-degradation kinetics of 4-nitrophenol using a g-C3N4-MCy. Four parameters were applied including initial concentration of 4-nitrophenol, amount of catalyst, dissolved oxygen content, and photon flux. The reaction rate constant was shown to be correlated with the 4-nitrophenol concentration and also with a direct increase in photon flux, and dissolved oxygen concentration. The following rate of photo-degradation for 4-nitrophenol was achieved by kinetic modeling:
中文翻译:

gC 3 N 4 -MCy光催化降解4-硝基苯酚:机理研究和动力学模型
通过使三聚氰胺和氰尿酸等比例热缩合,制得石墨氮化碳(gC 3 N 4 -MCy)光催化剂。通过傅里叶变换红外光谱(FTIR),比表面积(SSA),X射线衍射(XRD),场发射扫描电子显微镜(FESEM),透射电子显微镜(TEM)和DRS–UV–Vis对光催化剂进行了表征。光谱方法。我们使用gC 3 N 4研究了4-硝基苯酚的光降解动力学-MCy。应用了四个参数,包括4-硝基苯酚的初始浓度,催化剂量,溶解氧含量和光子通量。已显示反应速率常数与4-硝基苯酚浓度相关,并且还与光子通量和溶解氧浓度的直接增加相关。通过动力学建模,获得了以下4-硝基苯酚的光降解速率:



























 京公网安备 11010802027423号
京公网安备 11010802027423号