当前位置:
X-MOL 学术
›
Org. Biomol. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Novel aryltriazole acyclic C-azanucleosides as anticancer candidates
Organic & Biomolecular Chemistry ( IF 3.2 ) Pub Date : 2020-11-17 , DOI: 10.1039/d0ob02164d Yanhua Zhang 1 , Yun Lin , Qianqian Hou , Xi Liu , Sabrina Pricl , Ling Peng , Yi Xia
Organic & Biomolecular Chemistry ( IF 3.2 ) Pub Date : 2020-11-17 , DOI: 10.1039/d0ob02164d Yanhua Zhang 1 , Yun Lin , Qianqian Hou , Xi Liu , Sabrina Pricl , Ling Peng , Yi Xia
Affiliation
|
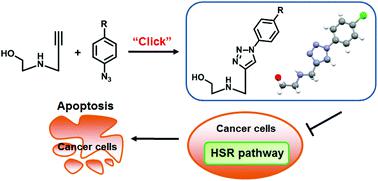
Nucleoside analogues represent an important class of drug candidates. With the aim of searching for novel bioactive nucleosides, we developed an efficient synthetic way to construct a series of aryl 1,2,3-triazole acyclic C-azanucleosides via Huisgen 1,3-dipolar cycloaddition. The aryl 1,2,3-triazole motifs within these azanucleosides showed coplanar features, suggesting they could act as surrogates for large planar aromatic systems or nucleobases. Moreover, several aryltriazole acyclic C-azanucleosides bearing long alkyl chains exhibited potent antiproliferative activity against various cancer cell lines via induction of apoptosis. Most interestingly, the lead compound significantly down-regulated the key proteins involved in the heat shock response pathway, representing the first anticancer acyclic azanucleoside with such a mode of action. These novel aryl 1,2,3-triazole cyclic C-azanucleosides therefore serve as promising paradigms for further exploring anticancer drug candidates.
中文翻译:

新型芳基三唑无环 C-氮杂核苷作为抗癌候选者
核苷类似物代表一类重要的候选药物。为了寻找新的生物活性核苷,我们开发了一种通过Huisgen 1,3-偶极环加成构建一系列芳基1,2,3-三唑无环C-氮杂核苷的有效合成方法。这些氮杂核苷中的芳基 1,2,3-三唑基序显示出共面特征,表明它们可以作为大型平面芳香系统或核碱基的替代物。此外,几种带有长烷基链的芳基三唑无环C-氮杂核苷通过以下途径对各种癌细胞系表现出有效的抗增殖活性诱导细胞凋亡。最有趣的是,先导化合物显着下调了参与热休克反应途径的关键蛋白,代表了第一个具有这种作用方式的抗癌无环氮杂核苷。因此,这些新型芳基 1,2,3-三唑环状C-氮杂核苷可作为进一步探索抗癌候选药物的有希望的范例。
更新日期:2020-11-25
中文翻译:

新型芳基三唑无环 C-氮杂核苷作为抗癌候选者
核苷类似物代表一类重要的候选药物。为了寻找新的生物活性核苷,我们开发了一种通过Huisgen 1,3-偶极环加成构建一系列芳基1,2,3-三唑无环C-氮杂核苷的有效合成方法。这些氮杂核苷中的芳基 1,2,3-三唑基序显示出共面特征,表明它们可以作为大型平面芳香系统或核碱基的替代物。此外,几种带有长烷基链的芳基三唑无环C-氮杂核苷通过以下途径对各种癌细胞系表现出有效的抗增殖活性诱导细胞凋亡。最有趣的是,先导化合物显着下调了参与热休克反应途径的关键蛋白,代表了第一个具有这种作用方式的抗癌无环氮杂核苷。因此,这些新型芳基 1,2,3-三唑环状C-氮杂核苷可作为进一步探索抗癌候选药物的有希望的范例。

























 京公网安备 11010802027423号
京公网安备 11010802027423号