Journal of Hazardous Materials ( IF 13.6 ) Pub Date : 2020-11-17 , DOI: 10.1016/j.jhazmat.2020.124446 Ling-Xi Zhao , Hua Xiao , Ming-Hui Li , Meng Xie , Na Li , Ru-Song Zhao
|
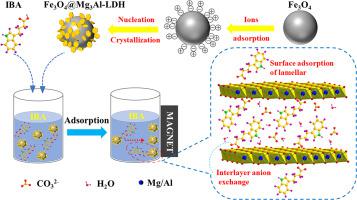
The magnetic layered double hydroxide-based materials (MLDHs) with the metal composition of Mg(II)Al(III) were synthesized by different conditions as the adsorbent for removal of a phytohormone, indole-3-butyric acid (IBA). The morphological characteristics of MLDHs were studied through various characterization methods such as XRD, SEM, TEM, FTIR, BET, Zeta-potential and VSM. The adsorption results showed that the adsorption capacity of MLDH-1 synthesized by co-precipitation method with ammonia as the base source was the best (maximum 522.6 mg/g). The extent of adsorption in the pH range of 3.0–9.0 was observed to be no noticeable change. From the economical point of view, 1.0 g/L MLDH-1 composites were selected as optimum parameter. For a given adsorbent concentration (Cs), its kinetics and adsorption isotherm followed the pseudo-second-order and Liu isotherm model, respectively. The adsorbed sample can be easily magnetically separated and regenerated with NaNO3. The adsorption process was spontaneous and exothermic, including two path stages: surface adsorption of lamellar and interlayer anion exchange. The research makes a contribution to evaluating the ability of MLDHs in IBA removal and helping the public to understand the mechanism of adsorption process.
中文翻译:

磁性层状双氢氧化物基吸附剂有效去除水溶液中的吲哚-3-丁酸
在不同条件下合成了金属组成为Mg(II)Al(III)的磁性层状双氢氧化物基材料(MLDHs)作为去除植物激素吲哚-3-丁酸(IBA)的吸附剂。通过XRD,SEM,TEM,FTIR,BET,Zeta电位和VSM等多种表征方法研究了MLDHs的形态特征。吸附结果表明,以氨为基础共沉淀法合成的MLDH-1的吸附能力最好(最大522.6 mg / g)。在3.0-9.0的pH范围内,吸附程度没有明显变化。从经济角度考虑,选择1.0 g / L MLDH-1复合材料作为最佳参数。对于给定的吸附剂浓度(C s),其动力学和吸附等温线分别遵循伪二级和Liu等温线模型。吸附的样品可以很容易地磁分离并用NaNO 3再生。吸附过程是自发且放热的,包括两个路径阶段:层状表面吸附和层间阴离子交换。该研究为评估MLDHs去除IBA的能力做出了贡献,并有助于公众了解吸附过程的机理。

























 京公网安备 11010802027423号
京公网安备 11010802027423号