Organic Materials Pub Date : 2020-09-30 , DOI: 10.1055/s-0040-1715564 Yubin Fu 1 , Junzhi Liu 2 , Zhongbin Wu 3 , Jan J. Weigand 4 , Xinliang Feng 1
|
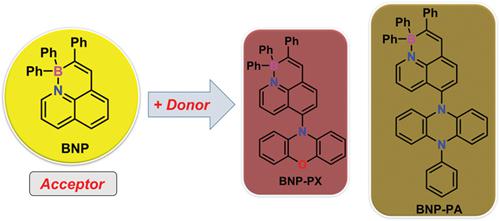
Organoboron compounds provide a new line to tune the electronic structures of π-conjugated molecules, which is critical to the development of new organic semiconductor materials. In this work, we demonstrate the synthesis of two novel boron–nitrogen (B−N) coordinated phenalene complexes (BNP-PX and BNP-PA) by employing BN phenalene (BNP) as the acceptor unit and phenoxazine/phenylphenazine groups as the donors. Based on single-crystal X-ray analysis, both BNP-PX and BNP-PA possess highly twisted conformations with the dihedral angles of 76.6 ° and 70.5 °, respectively. The photophysical properties of BNP-PX and BNP-PA are elucidated through UV-vis absorption, fluorescence spectroscopy, and theoretical calculations. In addition, BNP-PX exhibits a large Stokes shift (8,033 cm−1) and excellent aggregated-induced emission behavior. The red organic light-emitting diode device was fabricated based on compound BNP-PX, manifesting its promising application in organic optoelectronic devices.
中文翻译:

AIE-活性B–N-配苯酚配合物的合成与表征
有机硼化合物为调节π共轭分子的电子结构提供了一条新路线,这对于开发新型有机半导体材料至关重要。在这项工作中,我们通过使用BN苯并(BNP)作为受体单元和吩恶嗪/苯基吩嗪基团来证明两个新型的硼-氮(B-N)配位的苯并(BNP-PX和BNP-PA)配合物的合成。根据单晶X射线分析,BNP-PX和BNP-PA都具有高度扭曲的构型,二面角分别为76.6°和70.5°。BNP-PX和BNP-PA的光物理性质通过紫外可见吸收,荧光光谱和理论计算来阐明。另外,BNP-PX显示出大的斯托克斯位移(8,033cm -1)和优异的聚集诱导的发射行为。基于化合物BNP-PX制备了红色有机发光二极管器件,显示了其在有机光电器件中的应用前景。



























 京公网安备 11010802027423号
京公网安备 11010802027423号