Organic Materials Pub Date : 2020-10-28 , DOI: 10.1055/s-0040-1715900 Christopher Schilling 1 , Finn Schulz 1 , Andreas Köhn 2 , Sabine Laschat 1
|
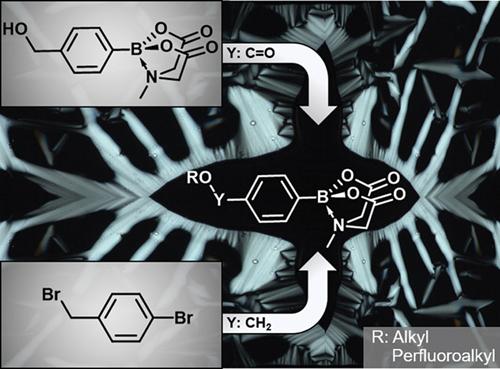
Two series of N-methyliminodiacetic acid (MIDA) boronates were prepared and their mesomorphic properties were investigated. MIDA-substituted benzoic acid esters were synthesized via the Mitsunobu reaction. The second series of MIDA benzyl ether derivatives was prepared via Williamson etherification and subsequent borylation. Both series exhibit smectic A (SmA) phases. In the case of MIDA boronate esters, a substitution with perfluorinated side chains led to increased transition temperatures and broadening of the SmA phases. The phase geometries of the mesophases were determined by X-ray diffraction. Quantum-chemical calculations provided further insight into the packing model.
中文翻译:

液晶苯甲酸酯MIDA硼酸酯:合成和同构性质
制备了两个系列的N-甲基亚氨基二乙酸(MIDA)硼酸盐,并研究了它们的介晶性质。通过Mitsunobu反应合成了MIDA取代的苯甲酸酯。第二系列的MIDA苄基醚衍生物是通过Williamson醚化和随后的硼化反应制得的。两个系列均表现出近晶A(SmA)相。在MIDA硼酸酯的情况下,用全氟侧链取代会导致转变温度升高和SmA相变宽。中间相的相几何形状通过X射线衍射确定。量子化学计算提供了对堆积模型的进一步了解。


























 京公网安备 11010802027423号
京公网安备 11010802027423号