当前位置:
X-MOL 学术
›
J. Nat. Prod.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Structure–Activity Relationships for the Marine Natural Product Sintokamides: Androgen Receptor N-Terminus Antagonists of Interest for Treatment of Metastatic Castration-Resistant Prostate Cancer
Journal of Natural Products ( IF 5.1 ) Pub Date : 2020-10-30 , DOI: 10.1021/acs.jnatprod.0c00921 Luping Yan , Carmen A Banuelos 1 , Nasrin R Mawji 1 , Brian O Patrick , Marianne D Sadar 1, 2 , Raymond J Andersen
Journal of Natural Products ( IF 5.1 ) Pub Date : 2020-10-30 , DOI: 10.1021/acs.jnatprod.0c00921 Luping Yan , Carmen A Banuelos 1 , Nasrin R Mawji 1 , Brian O Patrick , Marianne D Sadar 1, 2 , Raymond J Andersen
Affiliation
|
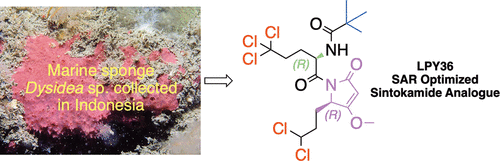
Synthetic analogues of the marine natural product sintokamides have been prepared in order to investigate the structure–activity relationships for the androgen receptor N-terminal domain (AR NTD) antagonist activity of the sintokamide scaffold. An in vitro LNCaP cell-based transcriptional activity assay with an androgen-driven luciferase (Luc) reporter was used to monitor the potency of analogues. The data have shown that the chlorine atoms on the leucine side chains are essential for potent activity. Analogues missing the nonchlorinated methyl groups of the leucine side chains (C-1 and C-17) are just as active and in some cases more active than the natural products. Analogues with the natural R configuration at C-10 and the unnatural R configuration at C-4 are most potent. Replacing the natural propionamide N-terminus cap with the more sterically hindered pivaloylamide N-terminus cap leads to enhanced potency. The tetramic acid fragment and the methyl ether on the tetramic acid fragment are essential for activity. The SAR optimized analogue 76 is more selective, easier to synthesize, more potent, and presumed to be more resistant to proteolysis than the natural sintokamides.
中文翻译:

海洋天然产物 Sintokamides 的结构-活性关系:用于治疗转移性去势抵抗性前列腺癌的雄激素受体 N 末端拮抗剂
制备了海洋天然产物辛托卡酰胺的合成类似物,以研究辛托卡酰胺支架的雄激素受体 N 末端结构域 (AR NTD) 拮抗剂活性的结构-活性关系。使用雄激素驱动的荧光素酶 (Luc) 报告基因进行体外LNCaP 细胞转录活性测定,以监测类似物的效力。数据表明亮氨酸侧链上的氯原子对于有效活性至关重要。缺少亮氨酸侧链非氯化甲基的类似物(C-1 和 C-17)与天然产物一样具有活性,并且在某些情况下活性更高。C-10 处具有天然R构型和C-4 处非天然R构型的类似物是最有效的。用空间位阻更大的新戊酰酰胺 N 端帽替换天然丙酰胺 N 端帽可增强效力。特特拉姆酸片段和特特拉姆酸片段上的甲基醚对于活性是必需的。SAR 优化的类似物76比天然 Sintokamides 更具选择性、更容易合成、更有效,并且推测更能抵抗蛋白水解。
更新日期:2020-10-30
中文翻译:

海洋天然产物 Sintokamides 的结构-活性关系:用于治疗转移性去势抵抗性前列腺癌的雄激素受体 N 末端拮抗剂
制备了海洋天然产物辛托卡酰胺的合成类似物,以研究辛托卡酰胺支架的雄激素受体 N 末端结构域 (AR NTD) 拮抗剂活性的结构-活性关系。使用雄激素驱动的荧光素酶 (Luc) 报告基因进行体外LNCaP 细胞转录活性测定,以监测类似物的效力。数据表明亮氨酸侧链上的氯原子对于有效活性至关重要。缺少亮氨酸侧链非氯化甲基的类似物(C-1 和 C-17)与天然产物一样具有活性,并且在某些情况下活性更高。C-10 处具有天然R构型和C-4 处非天然R构型的类似物是最有效的。用空间位阻更大的新戊酰酰胺 N 端帽替换天然丙酰胺 N 端帽可增强效力。特特拉姆酸片段和特特拉姆酸片段上的甲基醚对于活性是必需的。SAR 优化的类似物76比天然 Sintokamides 更具选择性、更容易合成、更有效,并且推测更能抵抗蛋白水解。

























 京公网安备 11010802027423号
京公网安备 11010802027423号