International Journal for Parasitology: Drugs and Drug Resistance ( IF 4 ) Pub Date : 2020-10-29 , DOI: 10.1016/j.ijpddr.2020.10.008 Brad E Sleebs 1 , Kate E Jarman 1 , Sonja Frolich 2 , Wilson Wong 1 , Julie Healer 1 , Weiwen Dai 3 , Isabelle S Lucet 1 , Danny W Wilson 2 , Alan F Cowman 1
|
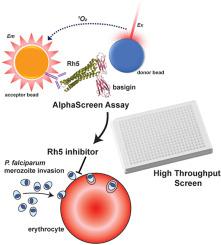
The P. falciparum parasite, responsible for the disease in humans known as malaria, must invade erythrocytes to provide an environment for self-replication and survival. For invasion to occur, the parasite must engage several ligands on the host erythrocyte surface to enable adhesion, tight junction formation and entry. Critical interactions include binding of erythrocyte binding-like ligands and reticulocyte binding-like homologues (Rhs) to the surface of the host erythrocyte. The reticulocyte binding-like homologue 5 (Rh5) is the only member of this family that is essential for invasion and it binds to the basigin host receptor. The essential nature of Rh5 makes it an important vaccine target, however to date, Rh5 has not been targeted by small molecule intervention. Here, we describe the development of a high-throughput screening assay to identify small molecules which interfere with the Rh5-basigin interaction. To validate the utility of this assay we screened a known drug library and the Medicines for Malaria Box and demonstrated the reproducibility and robustness of the assay for high-throughput screening purposes. The screen of the known drug library identified the known leukotriene antagonist, pranlukast. We used pranlukast as a model inhibitor in a post screening evaluation cascade. We procured and synthesised analogues of pranlukast to assist in the hit confirmation process and show which structural moieties of pranlukast attenuate the Rh5 – basigin interaction. Evaluation of pranlukast analogues against P. falciparum in a viability assay and a schizont rupture assay show the parasite activity was not consistent with the biochemical inhibition of Rh5, questioning the developability of pranlukast as an antimalarial. The high-throughput assay developed from this work has the capacity to screen large collections of small molecules to discover inhibitors of P. falciparum Rh5 for future development of invasion inhibitory antimalarials.
中文翻译:

用于鉴定恶性疟原虫网织红细胞结合样同源物 5 蛋白小分子抑制剂的高通量筛选试验的开发和应用
该恶性疟原虫导致人类疟疾的寄生虫必须侵入红细胞以提供自我复制和生存的环境。为了发生入侵,寄生虫必须与宿主红细胞表面的几种配体结合,以实现粘附、紧密连接的形成和进入。关键的相互作用包括红细胞结合样配体和网织红细胞结合样同源物 (Rhs) 与宿主红细胞表面的结合。网织红细胞结合样同源物 5 (Rh5) 是该家族中唯一对入侵至关重要的成员,它与 basigin 宿主受体结合。Rh5 的本质特性使其成为重要的疫苗靶点,但迄今为止,Rh5 尚未成为小分子干预的目标。在这里,我们描述了一种高通量筛选试验的发展,以识别干扰 Rh5-basigin 相互作用的小分子。为了验证该检测的实用性,我们筛选了一个已知的药物库和疟疾药物盒,并证明了该检测在高通量筛选方面的可重复性和稳健性。已知药物库的筛选鉴定了已知的白三烯拮抗剂普仑司特。我们在筛选后评估级联中使用普仑司特作为模型抑制剂。我们采购并合成了普仑司特的类似物,以协助命中确认过程并显示普仑司特的哪些结构部分减弱了 Rh5 - basigin 相互作用。普鲁司特类似物的评估 为了验证该检测的实用性,我们筛选了一个已知的药物库和疟疾药物盒,并证明了该检测在高通量筛选方面的可重复性和稳健性。已知药物库的筛选鉴定了已知的白三烯拮抗剂普仑司特。我们在筛选后评估级联中使用普仑司特作为模型抑制剂。我们采购并合成了普仑司特的类似物,以协助命中确认过程并显示普仑司特的哪些结构部分减弱了 Rh5 - basigin 相互作用。普鲁司特类似物的评估 为了验证该检测的实用性,我们筛选了一个已知的药物库和疟疾药物盒,并证明了该检测在高通量筛选方面的可重复性和稳健性。已知药物库的筛选鉴定了已知的白三烯拮抗剂普仑司特。我们在筛选后评估级联中使用普仑司特作为模型抑制剂。我们采购并合成了普仑司特的类似物,以协助命中确认过程并显示普仑司特的哪些结构部分减弱了 Rh5 - basigin 相互作用。普鲁司特类似物的评估 普鲁司特。我们在筛选后评估级联中使用普仑司特作为模型抑制剂。我们采购并合成了普仑司特的类似物,以协助命中确认过程并显示普仑司特的哪些结构部分减弱了 Rh5 - basigin 相互作用。普鲁司特类似物的评估 普鲁司特。我们在筛选后评估级联中使用普仑司特作为模型抑制剂。我们采购并合成了普仑司特的类似物,以协助命中确认过程并显示普仑司特的哪些结构部分减弱了 Rh5 - basigin 相互作用。普鲁司特类似物的评估恶性疟原虫在活力测定和裂殖体破裂测定中显示寄生虫活性与 Rh5 的生化抑制不一致,质疑 pranlukast 作为抗疟药的可开发性。根据这项工作开发的高通量检测能够筛选大量小分子,以发现恶性疟原虫Rh5 的抑制剂,以用于未来开发入侵抑制性抗疟药。

























 京公网安备 11010802027423号
京公网安备 11010802027423号