Chemical Physics Letters ( IF 2.8 ) Pub Date : 2020-10-19 , DOI: 10.1016/j.cplett.2020.138098 Yunlong Gao , A. Ligia Focsan , Lowell D. Kispert
|
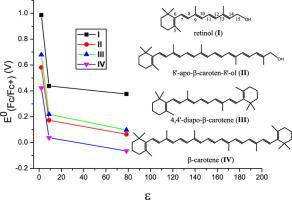
Four density functionals and the C-PCM solvation model were used in the DFT calculations of the oxidation potentials of four carotenoids in cyclohexane, dichloromethane and water. The values obtained using M06-2X+D3 density functional fit the experimental data the best. The calculated oxidation potentials of the carotenoids decrease with increasing the polarities of the solvents, the difference being as large as 0.6 V. These behaviors are independent of the symmetries and chain lengths of the carotenoids. The results of this study are very important in the design of supramolecular carotenoid complexes because antioxidant activities the carotenoids are oxidation potential dependent.
中文翻译:

环境极性对类胡萝卜素抗氧化活性的影响
DFT计算中使用了四种密度泛函和C-PCM溶剂化模型对四种类胡萝卜素在环己烷,二氯甲烷和水中的氧化电位进行了DFT计算。使用M06-2X + D3密度泛函获得的值最适合实验数据。计算出的类胡萝卜素氧化电位随溶剂极性的增加而降低,差异最大为0.6V。这些行为与类胡萝卜素的对称性和链长无关。这项研究的结果在超分子类胡萝卜素复合物的设计中非常重要,因为类胡萝卜素的抗氧化活性与氧化电位有关。

























 京公网安备 11010802027423号
京公网安备 11010802027423号